Labetalol
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ləˈbɛtəlɔːl/ |
| Trade names | Normodyne, Trandate, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685034 |
| License data | |
| Pregnancy category |
|
intravenous | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 25% |
| Protein binding | 50% |
| Metabolism | Liver pass metabolism, |
| Elimination half-life | Tablet: 6–8 hours; IV: 5.5 hours |
| Excretion | Excreted in urine, not removed by hemodialysis |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Labetalol is a medication used to treat
Common side effects include
Labetalol was patented in 1966 and came into medical use in 1977.
Medical uses
Labetalol is effective in the management of
It has a particular indication in the treatment of
It is also used as an alternative in the treatment of severe hypertension.[7]
Special populations
Pregnancy: studies in lab animals showed no harm to the baby. However, a comparable well-controlled study has not been performed in pregnant women.[9]
Nursing: breast milk has been shown to contain small amounts of labetalol (0.004% original dose). Prescribers should be cautious in the use of labetalol for nursing mothers.[9]
Pediatric: no studies have established safety or usefulness in this population.[9]
Geriatric: the elderly are more likely to experience dizziness when taking labetalol. Labetalol should be dosed with caution in the elderly and counseled on this side effect.[9]
Side effects
Common
- Neurologic: headache (2%), dizziness (11%)[9]
- Gastrointestinal: nausea (6%), dyspepsia (3%)[9]
- Cholinergic: nasal congestion (3%), ejaculation failure (2%)[9]
- Respiratory: dyspnea (2%)[9]
- Other:
Low blood pressure with standing is more severe and more common with IV formulation (58% vs 1%[9]) and is often the reason larger doses of the oral formulation cannot be used.[10]
Rare
- Fever[9]
- Muscle cramps[9]
- Dry eyes[9]
- Heart block[9]
- Hyperkalemia[9]
- Hepatotoxicity[9]
- Drug eruption similar to lichen planus[11]
- respiratory distress[9]
Contraindications
Labetalol is contraindicated in people with overt cardiac failure, greater-than-first-degree
Pharmacology
Mechanism of action
Labetalol is a
Labetalol is about
Labetalol's dual α- and β-adrenergic antagonism has different physiological effects in short- and long-term situations. In short-term, acute situations, labetalol decreases blood pressure by decreasing
Labetalol possesses significant intrinsic sympathomimetic activity.
Similar to local anesthetics and sodium channel blocking antiarrhythmics, labetalol also has membrane stabilizing activity.[16][21] By decreasing sodium entry, labetalol decreases action potential firing and thus has local anesthetic activity.[22]
Physiological action
The physiological effects of labetalol when administered acutely (intravenously) are not predictable solely by their receptor blocking effect, i.e. blocking β1-adrenergic receptors should decrease heart rate, but labetalol does not. When labetalol is given in acute situations, it decreases the peripheral vascular resistance and systemic blood pressure while having little effect on the heart rate, cardiac output and stroke volume, despite its α1-, β1- and β2-adrenergic receptor blocking mechanism.[17][18] These effects are mainly seen when the person is in the upright position.[20]
Long term labetalol use also has different effects from other beta blockers. Other beta blockers, such as propranolol, persistently reduce cardiac output during exercise. The peripheral vascular resistance decreases when labetalol is first administered. Continuous labetalol use further decreases peripheral vascular resistance. However, during exercise, cardiac output remains the same due to a compensatory mechanism that increases stroke volume. Thus, labetalol is able to reduce heart rate during exercise while maintaining cardiac output by the increase in stroke volume.[18]
Pharmacokinetics
Labetalol is classified as a beta blocker with low
Chemistry
The minimum requirement for
Labetalol has two
| Stereoisomers of labetalol | |
|---|---|
 (R,R)-Labetalol CAS number: 75659-07-3 |
 (S,S)-Labetalol CAS number: 83167-24-2 |
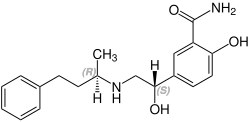 (R,S)-Labetalol CAS number: 83167-32-2 |
 (S,R)-Labetalol CAS number: 83167-31-1 |
Labetalol acts by blocking α- and β-adrenergic receptors, resulting in decreased
The β:α antagonism of labetalol is approximately 3:1.[27][28]
It is chemically designated in International Union of Pure and Applied Chemistry (IUPAC) nomenclature as 2-hydroxy-5-[1-hydroxy-2-[(1-methyl-3-phenylpropyl)amino]ethyl]benzamide monohydrochloride.[26][29]
History
Labetalol was the first drug created that combined both α- and β-adrenergic receptor blocking properties. It was created to potentially fix the compensatory reflex issue that occurred when blocking a single receptor subtype, i.e. vasoconstriction after blocking β-adrenergic receptors or tachycardia after blocking α-adrenergic receptors. Because the reflex from blocking the single receptor subtypes acted to prevent the lowering of blood pressure, it was postulated that weak blocking of both α- and β-adrenergic receptors could work together to decrease blood pressure.[15][18]
References
- ^ a b c d e f "Labetalol Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ^ ISBN 9780857113382.
- ^ "Labetalol Use During Pregnancy". Drugs.com. Retrieved 11 March 2019.
- ISBN 9783527607495.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Labetalol - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ ISBN 978-1-60913-713-7.
- PMID 23962474.
- ^ a b c d e f g h i j k l m n o p q "Trandate" (PDF). Prometheus Laboratories Inc. November 2010. Retrieved 3 November 2015.
- ^ "Labetalol hydrochloride" (PDF). Hospira. May 2015. Archived from the original (PDF) on 4 March 2016. Retrieved 3 November 2015.
- ISBN 978-1-4160-2999-1.
- ^ a b c d "Labetalol [package insert]. Spring Valley, NY: Par Pharmaceutical; 2011" (PDF). Archived from the original (PDF) on 10 December 2015. Retrieved 3 November 2015.
- ^ PMID 33572109.
- ^ ISBN 978-0-07-176401-8.
- ^ ISBN 978-0-444-90469-0.
- ^ ISBN 978-0781737623.
- ^ S2CID 20410587.
- ^ S2CID 46974416.
- PMID 2464093.
- ^ S2CID 46986875.
- ISBN 978-0-7817-8899-1.
- ISBN 978-81-312-1620-0.
- ISBN 978-3-642-74209-5.
- ^ a b Mehta A (2007). "Adrenergics and Cholinergic their Biosynthesis, Metabolism and Structure Activity Relationships". Medicinal Chemistry of the Peripheral Nervous System. PharmaXChange.info. Archived from the original on 4 November 2010. Retrieved 16 October 2010.
- PMID 1687367.
- ^ ISBN 978-0-07-176401-8.
- ISBN 978-0-07-145153-6.
- PMID 9968.
- ^ "labetalol | C19H24N2O3". PubChem. U.S. National Library of Medicine. Retrieved 4 November 2015.
