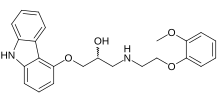
Carvedilol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Coreg, others |
| Other names | BM-14190 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697042 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 25–35% |
| Protein binding | 98% |
| Metabolism | Liver (CYP2D6, CYP2C9) |
| Elimination half-life | 7–10 hours |
| Excretion | Urine (16%), feces (60%) |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Carvedilol, sold under the brand name Coreg among others, is a
Common
Carvedilol was patented in 1978 and approved for medical use in the United States in 1995.
Medical uses
Carvedilol is indicated in the management of
Carvedilol reduces the risk of death, hospitalizations, and recurring heart attacks for patients with reduced heart function following a heart attack.[11][12] Carvedilol has also been proven to reduce death and hospitalization in patients with severe heart failure.[13]
In practice, carvedilol has been used in the treatment of uncomplicated hypertension, yet studies suggest it has relatively ineffective blood pressure-lowering effects when compared with other blood pressure-lowering therapies or other beta blockers.[14]
Carvedilol has also shown efficacy in the prevention of bleeding from esophageal varices in patients with cirrhosis.[15]
Available forms
Carvedilol is available in the following forms:
Contraindications
Carvedilol should not be used in patients with bronchial asthma or bronchospastic conditions due to increased risk of bronchoconstriction.
Side effects
The most common side effects (>10% incidence) of carvedilol include:[16]
- Dizziness
- Fatigue
- Low blood pressure
- Diarrhea
- Weakness
- Slowed heart rate
- Weight gain
- Erectile dysfunction
Carvedilol is not recommended for people with uncontrolled bronchospastic disease (e.g. current asthma symptoms) as it can block receptors that assist in opening the airways.[16]
Carvedilol may mask symptoms of low blood sugar,[16] resulting in hypoglycemia unawareness. This is termed beta blocker induced hypoglycemia unawareness.
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Action |
|---|---|---|
| 5-HT1A | 3.4 | Antagonist |
| 5-HT2 | 207 | Antagonist |
| D2 | 213 | Antagonist |
| α1 | 3.4 | Antagonist |
α2
|
2,168 | Antagonist |
β1
|
0.24–0.43 | Antagonist |
β2
|
0.19–0.25 | Antagonist |
2
|
? | Antagonist[23] |
Carvedilol is both a
Using rat proteins, carvedilol has shown affinity for a variety of targets including the β1-adrenergic receptor (Ki = 0.24–0.43 nM), β2-adrenergic receptor (Ki = 0.19–0.25 nM), α1-adrenergic receptor (Ki = 3.4 nM),
Carvedilol reversibly binds to β-adrenergic receptors on cardiac
Pharmacokinetics
Carvedilol is about 25% to 35%
The majority of carvedilol is
The compound is
The mean
References
- ^ a b c d e f g h i "Carvedilol Monograph for Professionals". Drugs.com. AHFS. Retrieved 24 December 2018.
- ^ "Carvedilol Use During Pregnancy". Drugs.com. Retrieved 24 December 2018.
- ISBN 9780857113382.
- ISBN 9783527607495.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Carvedilol - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 23741058.
- ISBN 9780071604055.
- PMID 26131706.
- S2CID 1840228.
- PMID 25862157.
- PMID 29040525.
- PMID 26306578.
- PMID 23250049.
- ^ a b c d e f g h i "Coreg - Food and Drug Administration" (PDF).
- ^ "Drug Approval Package". www.accessdata.fda.gov. Retrieved 5 November 2015.
- PMID 28126029.
- PMID 12490274.
- PMID 28213164.
- PMID 30372514.
- S2CID 23494154.
- S2CID 22480332.
- ^ "Carvedilol | Ligand page | IUPHAR/BPS Guide to PHARMACOLOGY".
- PMID 2462161.
- PMID 30998954.
- PMID 17297209. Archived from the original(PDF) on 4 March 2016. Retrieved 12 November 2015.
- S2CID 2901620.
- PMID 20579773.
- PMID 15951832.
Further reading
- Chakraborty S, Shukla D, Mishra B, Singh S (February 2010). "Clinical updates on carvedilol: a first choice beta-blocker in the treatment of cardiovascular diseases". Expert Opinion on Drug Metabolism & Toxicology. 6 (2): 237–50. S2CID 25670550.
- Dean L (2018). "Carvedilol Therapy and CYP2D6 Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 30067327. Bookshelf ID: NBK518573.
