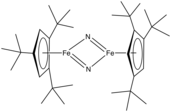
Metal nitrido complex
Metal nitrido complexes are
Structural trends
Mononuclear complexes feature terminal nitride ligands, typically with short M-N distances consistent with
- Example metal nitrido complexes
-
[OsNO3]−, isoelectronic with osmium tetroxide.
-
[MoNCl4]−, a square pyramidal Mo(VI) complex.
-
[W2(μ-N)Cl10]−, containing two W(VI) centres bridged by a nitrido ligand.
-
[Ir3N(SO4)6(H2O)3]4−, structurally related tobasic iron acetate.
-
A uranium nitrido complex.
Preparative routes
Metal nitrides are produced using a variety of nitrogen sources. The first example above is prepared from
- OsO4 + KNH2 → KOsO3N + H2O
Most commonly however, nitrido complexes are produced by decomposition of azido complexes.[6] The driving force for these reactions is the great stability of N2. Nitrogen trichloride is an effective reagent to give chloro-nitrido complexes. In some cases, even N2 and nitriles can serve as sources of nitride ligands.[7]
Reactions of nitrido ligands
The nitride ligand can be both electrophilic and nucleophilic.[8][9] Terminal nitrides of early metals tend to be basic and oxidizable, whereas nitrides of the later metals tend to be oxidizing and electrophilic. The former behavior is illustrated by their N-protonation and N-alkylation. Ru and Os nitrido complexes often add organophosphines to give iminophosphine derivatives containing the R3PN− ligand.
Interstitial nitrides
Owing to the ability of nitrido ligands to serve as a

See also
- Abiological nitrogen fixation
- Transition metal dinitrogen complex
General references
- ISBN 978-0-08-037941-8.
- .
- .
- PMID 22745250.
- LCCN 63-14307– via the Internet Archive.
- PMID 30155215.
- .
- doi:10.1039/a802635a.
- )
- )

![[OsNO3]−, isoelectronic with osmium tetroxide.](http://upload.wikimedia.org/wikipedia/commons/thumb/f/f8/OsNO3_anion.png/165px-OsNO3_anion.png)
![[MoNCl4]−, a square pyramidal Mo(VI) complex.](http://upload.wikimedia.org/wikipedia/commons/thumb/2/29/MoNCl4_anion.png/170px-MoNCl4_anion.png)
![[W2(μ-N)Cl10]−, containing two W(VI) centres bridged by a nitrido ligand.](http://upload.wikimedia.org/wikipedia/commons/thumb/8/82/W2NCl10_anion.png/170px-W2NCl10_anion.png)
![[Ir3N(SO4)6(H2O)3]4−, structurally related to basic iron acetate.](http://upload.wikimedia.org/wikipedia/commons/thumb/0/00/Ir3N%28SO4%296aq3.png/128px-Ir3N%28SO4%296aq3.png)