Transition metal carbyne complex
Transition metal carbyne complexes are
Synthesis
Transition metal carbyne complexes are most common for the early transition metals, especially niobium, tantalum, molybdenum, tungsten, and rhenium. They can also have low-valence metals as well as high-valence metals.

The first Fischer carbyne complex was reported in 1973.[4] Two years later in 1975, the first "Schrock carbyne" was reported.[5]
Many high-valent carbyne complexes have since been prepared, often by
Bridging alkylidyne ligands in cluster compounds
Some metal carbynes
Several cluster-bound carbyne complexes are known, typically with
- 2 HCBr3 + 9⁄2 Co2(CO)8 → 2 HCCo3(CO)9 + 18 CO + 3 CoBr2
Structure
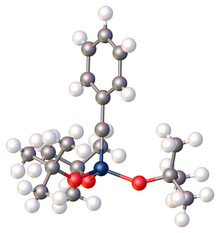
Monomeric metal carbyne complexes exhibit fairly linear M–C–R linkages according to
Metal carbyne complexes also exhibit a large trans effect, where the ligand opposite the carbyne is typically labile.
Reactions and applications
Hexa(tert-butoxy)ditungsten(III) is a catalyst for alkyne metathesis.[11] The catalytic cycle involves an carbyne intermediate.[12]
Some carbyne complexes react with electrophiles at C-carbyne followed by association of the anion. The net reaction gives a transition metal carbene complex:
- LnM≡CR + HX → Ln(X)M=CHR
These complexes can also undergo
In some carbyne complexes, coupling of the carbyne ligand to a
Main group analogue
A sulfur-based main group analog of a carbyne complex has been prepared by Seppalt and coworkers.[14] The compound, trifluoro(2,2,2-trifluoroethylidyne)-λ6-sulfurane, F3C–C≡SF3, prepared by dehydrofluorination of F3C–CH=SF4 or F3C–CH2–SF5, is an unstable gas that readily undergoes dimerization to form trans-(CF3)(SF3)C=C(CF3)(SF3) at above –50 °C.
Further reading
- Mayr, A.; Bastos, C. M. (1992). Coupling Reactions of Terminal Two-Faced π Ligands and Related Cleavage Reactions. Progress in Inorganic Chemistry. Vol. 40. pp. 1–98. )
References
- PMID 35793547.
- )
- ^ ISBN 978-3-527-29390-2.
- .
- .
- )
- .
- ISBN 9780199342679.
- ISBN 9781118138076.
- ISBN 9789401047289.
- PMID 34519486.
- .
- .
- ISSN 0002-7863.
