Transition metal chloride complex

In chemistry, a transition metal chloride complex is a coordination complex that consists of a transition metal coordinated to one or more chloride ligand. The class of complexes is extensive.[1]
Bonding
Halides are X-type
Homoleptic metal halide complexes are known with several stoichiometries, but the main ones are the hexahalometallates and the tetrahalometallates. The hexahalides adopt
Due to the presence of filled pπ orbitals, halide ligands on transition metals are able to reinforce
Homoleptic complexes
Homoleptic complexes (complexes with only chloride ligands) are often common reagents. Almost all examples are anions.
1st row
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| TiCl4 | colourless | (t2g)0 | tetrahedral | ||
| [Ti2Cl9]− | white/colourless | d0d0 | face-sharing bioctahedron | Ti-Cl(terminal) = 2.23 Å, 2.45 (terminal) (N(PCl3)2)+ salt)[4] | |
| [Ti2Cl9]3- | orange | (t2g)1(t2g)1 | face-sharing bioctahedron | Ti-Ti =3.22 Å Ti-C1(terminal) = 2.32-2.35 Å, Ti-Cl(bridge) = 2.42-2.55 Å ((NEt4+)3)3 salt)[5] | |
| [Ti2Cl10]2− | colourless | d0d0 | 
|
bioctahedral | |
| [Ti3Cl12]3- | green | (t2g)1(t2g)1(t2g)1 | face-sharing trioctahedron | Ti-Ti = 3.19, 3.10 Å (terminal) Ti-C1(terminal) = 2.36 Å (terminal), Ti-Cl(bridge) = 2.50 Å ((PPh4+)3)3 salt)[6] | |
| [TiCl6]2− | yellow | d0 | 
|
octahedral | PPh4+ salt Ti-Cl = 2.33 Å[7] |
| VCl4 | red | (t2g)1 | tetrahedral | V1−Cl = 2.29 Å | |
V2Cl10
|
violet | (t2g)0 | 
|
edge-shared bioctahedron | V1−Cl(bridging) = 2.48 Å V1−Cl(terminal) = 2.16-2.21 Å[8] |
| [VCl6]2- | red | (t2g)1 | 
|
octahedral | V1−Cl = 2.29 Å[9] |
| [CrCl6]3− | pink[3] | (t2g)3 | 
|
octahedral[10][3] | |
| [Cr2Cl9]3− | red | (d3)2 | face-sharing bioctahedron | Cr-Cl(terminal) = 2.31 Å, 2.42 (terminal) (Et2NH2+ salt)[11] | |
| [MnCl4]2−[12] | pale pink to while | (eg)2(t2g)3 | tetrahedral | Mn-Cl bond length = 2.3731-2.3830 Å[13] | |
| [MnCl6]2− | dark red | (t2g)3(eg)1 | 
|
octahedral | Mn-Cl distance = 2.28 Å K+ salt[14]) salt is isostructural with K2PtCl6 |
| [MnCl6]3− | brown[3] | (t2g)3(eg)1 | 
|
octahedral[10][3] | |
| [Mn2Cl6]2− | yellow-green | (eg)2(t2g)3 | 
|
bitetrahedral | Mn-Cl(terminal) bond length = 2.24 Å Mn-Cl(terminal) bond length = 2.39 Å[15] (PPN+)2 salt |
| [Mn3Cl12]6− | pink | (t2g)3(eg)2 | cofacial trioctahedron | Mn-Cl distance = --- Å [(C(NH2)3]+6 salt[16]
| |
| [FeCl4]2−[12] | cream | (eg)3(t2g)3 | tetrahedral((Et4N+)2 salt)[12] | ||
| [FeCl4]− | (eg)2(t2g)3 | tetrahedral | Fe-Cl bond length = 2.19 Å[17] | ||
| [FeCl6]3− | orange | (t2g)3(eg)2 | 
|
octahedral[3] | |
| [Fe2Cl6]2− | pale yellow | (eg)2(t2g)3 | 
|
bitetrahedral | Fe-Cl(terminal) bond length = 2.24 Å Fe-Cl(terminal) bond length = 2.39 Å[15] (PPN+)2 salt |
| [CoCl4]2−[12] | blue[12] | (eg)4(t2g)3 | tetrahedral | ||
| [Co2Cl6]2− | blue[15] | (eg)4(t2g)3 | 
|
bitetrahedral | Mn-Cl(terminal) bond length = 2.24 Å Co-Cl(terminal) bond length = 2.35 Å[15] (PPN+)2 salt |
| [NiCl4]2−[12] | blue[12] | (eg)4(t2g)4 | tetrahedral | Ni-Cl bond length = 2.28 Å (Et4N+)2 salt[18] | |
| [Ni3Cl12]6− | orange[19] | (t2g)6(eg)2 | confacial trioctahedral | ((Me2NH2+)2)8 salt double salt with two Cl− Ni-Cl bond length = 2.36-2.38 Å[19] | |
| [CuCl4]2−[12] | orange[20] yellow (flattened tetrahedral)[21] green (square planar)[22] |
(t2g)6(eg)3 | flattened tetrahedral or square planar[23][24] |
Cu-Cl bond length = 2.24 Å | |
| [Cu2Cl6]2− | red | [(t2g)6(eg)3]2 | edge-shared bis(square planar)[25] | Cu-Cl(terminal) = 2.24 Å Cu-Cl(bridging) = 2.31 Å | |
| [ZnCl4]2− | white/colorless | d10 | tetrahedral |
2nd row
Some homoleptic complexes of the second row transition metals feature metal-metal bonds.
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| [ZrCl6]2− | yellow | d0 | 
|
octahedral | Zr-Cl distance = 2.460 Å (Me4N+)2 salt[27] |
| [Zr2Cl10]2− | colorless | (d0)2 | 
|
edge-shared bioctahedral | Zr-Cl = 2.36 Å (terminal), 2.43 Å (bridging) N(PCl3)2)+ salt[4] |
Nb2Cl10
|
yellow | (d0)2 | 
|
edge-shared bioctahedral [Nb2Cl10] | 3.99 Å[28] |
| [NbCl6]− | yellow | d0 | 
|
octahedral | Nb-Cl = 2.34 Å N(PCl3)2)+ salt[4] |
| [Nb6Cl18]2− | black | (d2)4(d3)2 (14 cluster electrons) | 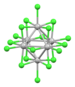
|
cluster Nb---Nb bonding | Nb-Cl = 2.92 Å (K+)2 salt[29] |
MoCl6
|
black | d0 | 
|
octahedron | Mo−Cl = 2.28 -2.31 Å[8] |
| [MoCl6]2− | yellow | (t2g)2 | 
|
octahedron | Mo−Cl = 2.37, 2.38, 2.27 Å[30] |
| [MoCl6]3− | pink | (t2g)3 | 
|
octahedral | |
| [Mo2Cl8]4− | purple[31] | 2(d4) | 
|
Mo-Mo quadruple bond | |
| [Mo2Cl9]3− | 2(d3) | face-shared bioctahedral | Mo-Mo (triple) bond length = 2.65 Å Mo-Cl (terminal) bond length = 2.38 Å Mo-Cl (bridging) bond length = 2.49 Å[32][33] | ||
| Mo2Cl10 | green | (d1)2 | 
|
edge-sharing bioctahedra[34] | |
| [Mo2Cl10]2− | (d2)2 | 
|
edge-sharing bioctahedra[35] | ||
| [Mo5Cl13]2− | brown[31] | d2d2d2d2d3 | 
|
incomplete octahedron[36] | |
| [Mo6Cl14]2− | yellow | d4 | 
|
octahedral cluster | (4-HOPyH+)2 salt[37] |
| [TcCl6]2− | yellow | (t2g)3 | 
|
octahedron | Tc-Cl = 2.35 Å for As(C6H5)4+ salt[38] |
| [Tc2Cl8]2− | green | (t2g)4 | 
|
Tc-Tc quadruple bond | Tc-Tc = 2.16, Tc-Cl = 2.34 Å for NBu4+ salt[39] |
| [RuCl6]2− | brown | (t2g)4 | 
|
octahedral | (EtPPh3+)2 salt[40] |
| [Ru2Cl9]3− | red | [(t2g)5]2 | cofacial bioctahedral | Ru-Ru bond length = 2.71 Å; Ru-Cl(terminal) = 2.35 Å, Ru-Cl(bridging) = 2.36 Å ((Et4N)+)3 salt[41] | |
| [Ru3Cl12]4− | green | (d5)2(d6) | cofacial trioctahedral | Ru-Ru bond lengths = 2.86 Å Ru-Cl bond lengths = 2.37-2.39 Å (Et4N+)2(H7O3+)2 salt[42] | |
| [RhCl6]3− | red | (t2g)6 | 
|
octahedral | H2N+(CH2CH2NH3+)2 salt)[43] |
| [Rh2Cl9]3− | red-brown | (t2g)6 | octahedral | Rh-Cl(terminal) = 2.30 Å, Rh-Cl(terminal) = 2.40 Å ((Me3CH2Ph)+)3 salt)[32] | |
| [PdCl4]2− | brown | d8 | square planar | ||
| [Pd2Cl6]2−[44] | red ((Et4N+)2 salt) | d8 | square planar | ||
| [Pd3Cl8]2−[45] | orange brown ((Bu4N+)2 salt) | d8 | square planar | ||
| [PdCl6]2− | brown | d6 | 
|
octahedral | Pd(IV) |
| [Pd6Cl12] | yellow-brown | d8 | 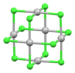
|
square planar[46] | |
| [AgCl2]− | white/colorless | d10 | linear | salt of [K(2.2.2-crypt)]+[47] | |
| [CdCl4]2− | white/colorless | d10 | tetrahedral | Et4N+ salt, Cd-Cl distance is 2.43 Å[26] | |
| [Cd2Cl6]2− | white/colorless | d10 | 
|
edge-shared bitetrahedron | (C6N3(4-C5H4N)33+ salt[48] |
| [Cd3Cl12]6− | white/colorless | d10 | octahedral (central Cd) pentacoordinate (terminal Cd's) cofactial trioctahedral |
(C6N3(4-C5H4N)33+ salt[48] (3,8-Diammonium-6-phenylphenanthridine3+)2[49] | |
| [Cd6Cl19]7− | white/colorless | d10 | 
|
octahedron of octahedra | 4,4'-(C6H3(2-Et)NH3+)2 salt[50] |
3rd row
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| [HfCl6]2− | white | d0 | 
|
octahedral | Hf-Cl distance = 2.448 A ((Me4N+)2 salt)[27] |
| [Hf2Cl10]2− | colorless/white | d0 | 
|
edge-shared bioctahedral[51] | |
| [Hf2Cl9]− | colorless/white | (d0)2 | face-shared bioctahedral[52] | ||
| [TaCl5] | white | d0 | 
|
edge-shared bioctahedral | |
| [TaCl6]− | white/colourless | d0 | 
|
octahedral | Ta-Cl = 2.34 Å (N(PCl3)2)+ salt)[4] |
| [Ta6Cl18]2- | green | d0 | 
|
octahedral | Ta-Ta = 2.34 Å (H+2 salt hexahydrate[53] |
| WCl6 | blue | d0 | 
|
octahedral | 2.24–2.26 Å[54] |
| [WCl6]2− | (t2g)2 | 
|
octahedral | W-Cl distances range from 2.34 to 2.37 Å (PPh4+ salt)[55] | |
| [WCl6]− | (t2g)1 | 
|
octahedral | W-Cl distance = 2.32 Å (Et4N+ salt)[56] | |
| W2Cl10 | black[57] | (t2g1)2 | 
|
bioctahedral | W-W distance = 3.814 Å[58] |
| [W2Cl8]4− | blue | 2(d4) | 
|
W-W quadruple bond | dW-W = 2.259 Å [Na(tmeda)+]4 salt[59] |
| [W2Cl9]2− | d3d2 | face-sharing bioctahedral | W-W distance = 2.54 Å W-Cl(terminal) = 2.36 Å, W-Cl(bridge) = 2.45 Å ((PPN+)2 salt)[60] | ||
| [W2Cl9]3− | d3d3 | octahedral | W-Cl distance = 2.32 Å (Et4N+ salt)[60] | ||
| [W3Cl13]3− | d3,d3,d4 | 
|
[W3(μ3-Cl)(μ-Cl)3Cl9]3- | W-W distances = 2.84 Å[61] | |
| [W3Cl13]2− | d3,d4,d4 | 
|
[W3(μ3-Cl)(μ-Cl)3Cl9]2-[61] | W-W distances = 2.78 Å[61] | |
| [W6Cl14]2- | yellow[62] | (d4)6 | 
|
see Mo6Cl12 | |
| [ReCl6]− | red-brown | (t2g)2 | 
|
octahedral | Re-Cl distance = 2.24-2.31 Å (PPh4+ salt)[63] |
| [ReCl6] | (t2g)1 | 
|
octahedral | Re-Cl distance = 226.3(6) Å[8] | |
| [ReCl6]2− | green | (t2g)3 | 
|
octahedral | Re-Cl distance = 2.35-2.38 Å ((PPN+)2 salt)[64] |
| [Re2Cl9]2− | (t2g)3(t2g)4 | face-sharing bioctahedral | Re-Re distance = 2.48 Å Re-Cl distances = 2.42 Å (bridge), 2.33 Å (terminal) ((Et4N+)2 salt)[65] | ||
| [Re2Cl9]− | ((t2g)3)2 | face-sharing bioctahedral | Re-Re distance = 2.70 Å Re-Cl distances = 2.41 (bridge), 2.28 Å (terminal) (Bu4N+ salt)[65] | ||
| [OsCl6]− | dark green | (t2g)3 | 
|
octahedral | dOs-Cl = 2.30 Å for Et4N+[66] and Ph4P+[67] salts |
| [OsCl6]2− | yellow-orange | (t2g)4 | 
|
octahedral[67] | Os-Cl distance 2.33 Å |
| [Os2Cl8]2− | green | (d5)2 | 
|
square antiprism | dOs-Os = 2.182 Å, dOs-Cl = 2.32 Å (Bu4N+)2 salt[68] |
| [Os2Cl10]2− | green | (d4)2 | 
|
octahedral | dOs-Cl(terminal) = 2.30 Å dOs-Cl(bridging) = 2.42 Å (Et4N+)2 salt[66] |
| [IrCl6]3− | red | (t2g)6 | 
|
octahedral | Ir-Cl = 2.36 Å[69] |
| [IrCl6]2− | brown | (t2g)5 | 
|
octahedral | Ir-Cl = 2.33 Å[70] |
| [Ir2Cl9]3− | - | ((t2g)6)2 | bi-octahedral[71] | ||
| [PtCl4]2− | pink | d8 | square planar | ||
| [PtCl6]2− | yellow | d6 | 
|
octahedral | Pt-Cl distance = 2.32 Å Et4N+ salt, ((Me4N+)2 salt)[27] |
| [Pt2Cl9]− | red (Bu4N+ salt) | ((t2g)6)2 | octahedral | Pt-Clt and Pt-Clbridge = 2.25, 2.38 Å[72] | |
| [Pt2Cl10]2− | yellow-brown (PPN+ salt) | ((t2g)6)2 | 
|
edge-shared bioctahedral | Pt-Clt and Pt-Clbridge = 2.27, 2.37 Å[72] |
| [Pt6Cl12] | yellow-brown | (d8)6 | 
|
square planar | Pt-Cl = 2.31[73] |
| [AuCl2]− | white/colorless | d10 | linear | Au-Cl distances of 2.28 Å NEt4+ salt[74] | |
| Au4Cl8 | black | (d10)2(d8)2 | 
|
linear and square planar | rare example of mixed valence, molecular chloride[75] |
| [AuCl4]− | yellow | d8 | square planar | Au-Cl distances of 2.26 Å NBu4+ salt[76] | |
| [HgCl4]2− | white/colorless | d10 | tetrahedral | Hg-Cl distance is 2.46 Å[26] Et4N+ salt | |
| [Hg2Cl6]2− | white/colorless | d10 | 
|
edge-shared bitetrahedral | Hg-Cl distance is 2.46 Å[77] Bu4N+ salt |
Heteroleptic complexes
Heteroleptic complexes containing chloride are numerous. Most hydrated metal halides are members of this class.
Hydrates

As indicated in the table below, many hydrates of metal chlorides are molecular complexes.[78][79] These compounds are often important commercial sources of transition metal chlorides. Several hydrated metal chlorides are not molecular and thus are not included in this tabulation. For example the dihydrates of manganese(II) chloride, nickel(II) chloride, copper(II) chloride, iron(II) chloride, and cobalt(II) chloride are coordination polymers.
| Formula of hydrated metal halides |
Coordination sphere of the metal |
|---|---|
| TiCl3(H2O)6 | trans-[TiCl2(H2O)4]+[80] |
| VCl3(H2O)6 | trans-[VCl2(H2O)4]+[80] |
| CrCl3(H2O)6 | trans-[CrCl2(H2O)4]+ |
| CrCl3(H2O)6 | [CrCl(H2O)5]2+ |
| CrCl2(H2O)4 | trans-[CrCl2(H2O)4] |
| CrCl3(H2O)6 | [Cr(H2O)6]3+[81] |
| MnCl2(H2O)6 | trans-[MnCl2(H2O)4] |
| MnCl2(H2O)4 | cis-[MnCl2(H2O)4][82] |
| FeCl2(H2O)6 | trans-[FeCl2(H2O)4] |
| FeCl2(H2O)4 | trans-[FeCl2(H2O)4] |
| FeCl3(H2O)6 | one of four hydrates of ferric chloride,[83]
|
| FeCl3(H2O)2.5 | cis-[FeCl2(H2O)4]+[84] |
| CoCl2(H2O)6 | trans-[CoCl2(H2O)4] |
| CoCl2(H2O)4 | cis-[CoCl2(H2O)4] |
| NiCl2(H2O)6 | trans-[NiCl2(H2O)4] |
| NiCl2(H2O)4 | cis-[NiCl2(H2O)4] |
Adducts
Metal chlorides form adducts with ethers to give transition metal ether complexes.
References
- ISBN 978-0-08-037941-8.
- ISBN 978-1-891389-53-5.
- ^ .
- ^ PMID 12205363.
- .
- .
- .
- ^ PMID 23172658.
- PMID 15500362.
- ^ a b O. S. Filipenko, D. D. Makitova, O. N. Krasochka, V. I. Ponomarev, L. O. Atovmyan (1987). Koord. Khim. 13: 669.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - .
- ^ ISBN 978-0-470-13240-1.
- .
- .
- ^ .
- S2CID 141394650.
- PMID 24816016.
- .
- ^ PMID 19805875.
- .
- PMID 16866484.
- PMID 25643715.
- PMID 22968285.
- .
- .
- ^ .
- ^ .
- doi:10.1107/S0108270191000239.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- S2CID 201122319.
- ^ ISBN 978-0-470-13244-9.
- ^ .
- doi:10.1021/ic50101a027.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 95489209.
- .
- PMID 21743925.
- PMID 18613684.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- PMID 22906536.
- S2CID 105746627.
- ^ I. A. Efimenko, T. A. Balakaeva, A. P. Kurbakova, A. S. Kanishcheva, A. V. Chuvaev, V. M. Stepanovich, Yu. N. Mikhailov (1992). Zh. Neorg. Khim. (Russ. J. Inorg. Chem.). 37: 1312.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - .
- .
- .
- PMID 15345822.
- .
- .
- ^ S2CID 203568412.
- doi:10.1039/b812504j.
- PMID 17279266.
- .
- .
- .
- .
- .
- .
- .
- .
- .
- ^ .
- ^ .
- PMID 11670462.
- .
- .
- ^ PMID 11666851.
- ^ S2CID 95254820.
- ^ .
- .
- .
- S2CID 199574461.
- ^ Yellowlees, L.; Elliot, M.; Parsons, S.; Messenger, D. "MASNEA". Cambridge Crystallographic Database CCDC 278284.
- ^ PMID 18166044.
- .
- .
- .
- doi:10.1071/C97029.
- .
- .
- .
- ^ .
- ^ Andress, K.R.; Carpenter, C. "Kristallhydrate. II.Die Struktur von Chromchlorid- und Aluminiumchloridhexahydrat" Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie 1934, volume 87, p446-p463.
- .
- .
- S2CID 105925459.
