Transition metal indenyl complex
In
Preparation and structure
Indene is deprotonated by
- C9H8 + BuLi → LiC9H7 + BuH
The resulting lithium indenide can be used to prepare indenyl complexes by salt metathesis reactions of metal halides.[3] When the metal halide is easily reduced, the trimethylstannylindenyl can be used as a source of indenyl anion:
- Me3SnC9H7 + TiCl4 → Me3SnCl + C9H7TiCl3
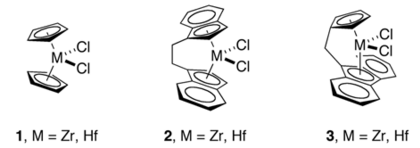
The M-C distances in indenyl complexes are comparable to those in cyclopentadienyl complexes. For the metallocenes M(Ind)2, ring slipping is evident for the case of M = Co and especially Ni, but not for M = Fe.[4] A number of chelating or ansa-bis(indenyl complexes are known, such as those derived from 2,2'-bis(2-indenyl) biphenyl
-
side view of (indenyl)2ZrMe2
-
Top view of (indenyl)2ZrMe2
-
View of (indenyl)2ZrMe2 down C2 symmetry axis.[5]
Indenyl effect
The indenyl effect refers to an explanation for the enhanced rates of substitution exhibited by η5-indenyl complexes vs the related η5-cyclopentadienyl complexes.[6][7][8][9][10][11]
Kinetic data support two proposed mechanisms for associative ligand substitution. The first mechanism, proposed by Hart-Davis and Mawby, is a concerted attack by the nucleophile and η5 to η3 transition followed by loss of a ligand and a η3 to η5 transition.
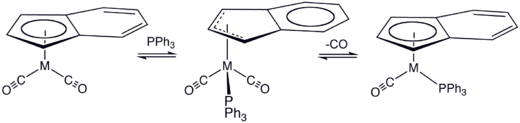
In a mechanism proposed by
η5 to η3 rearrangement in other ligands
Indenyl like effects are also observed in a number of non indenyl substituted metal complexes. In

Veiros conducted a study comparing the rate of substitution on [(η5-X)Mn(CO)3] where X is cyclopentadienyl, indenyl, fluorenyl, cyclohexadienyl, and 1-hydronaphthalene. Unsurprisingly, it was found that the ease of η5 to η3 haptotropic shift correlated to the strength of the Mn-X bond.[10]
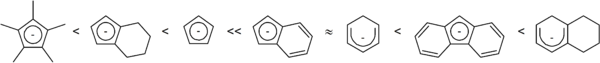
History
The indenyl analogues of
The cobalt derivative is a poorer reductant than cobaltocene.The indenyl effect was discovered by Hart-Davis and Mawby in 1969 through studies on the conversion of (η5-C9H7)Mo(CO)3CH3 to the phosphine-substituted acetyl complex, which follows bimolecular kinetics. This rate law was attributed to the haptotropic rearrangement of the indenyl ligand from η5 to η3. The corresponding reaction of tributylphosphine with (η5-C5H5)Mo(CO)3CH3 was 10 x slower.[13] The term indenyl effect was coined by Fred Basolo.
Subsequent work by Hart-Davis, Mawby, and White compared CO substitution by phosphines in Mo(η5-C9H7)(CO)3X and Mo(η5-C5H5)(CO)3X (X = Cl, Br, I) and found the cyclopentadienyl compounds to substitute by an SN1 pathway and the indenyl compounds to substitute by both SN1 and SN2 pathways. Mawby and Jones later studied the rate of CO substitution with P(OEt)3 with Fe(η5-C9H7)(CO)2I and Fe(η5-C5H5)(CO)2I and found that both occur by an SN1 pathway with the indenyl substitution occurring about 575 times faster.
Work in the early 1980s by Basolo found the SN2 replacement of CO in Rh(η5-C9H7)(CO)2 to be 108 times faster than in Rh(η5-C5H5)(CO)2. Shortly afterwards, Basolo tested the effect of the indene ligand on Mn(η5-C9H7)(CO)3, the cyclopentadienyl analogue of which having been shown to be inert to CO substitution. Mn(η5-C9H7)(CO)3 did undergo CO loss and was found to substitute via an SN2 mechanism.
References
- .
- )
- ^ .
- .
- .
- doi:10.1016/S0020-1693(00)93323-1.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
- .
- ^ .
- PMID 11857701.
- doi:10.1039/A908934I.
- .

![View of (indenyl)2ZrMe2 down C2 symmetry axis.[5]](http://upload.wikimedia.org/wikipedia/commons/thumb/a/ae/INDMZRdownC2.png/106px-INDMZRdownC2.png)