Transition metal carbene complex
A transition metal carbene complex is an
Classification
Metal carbene complexes are often classified into two types. The Fischer carbenes, named after
Fischer carbenes
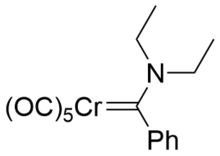
The common features of Fisher carbenes are:[6]
- low oxidation state metal center
- middle and late transition metals Fe(0), Mo(0), Cr(0)
- π-acceptor metal ligands
- π-donor aminogroups.
Examples include (CO)5W=COMePh and (OC)5Cr=C(NR2)Ph.

Fisher carbene complexes are related to the singlet form of carbenes, where both electrons occupy the same sp2 orbital at the carbon. This lone pair donates to a metal-based empty d orbital, forming a σ bond. π-backbonding from a filled metal d orbital to the empty p orbital of the carbon atom is possible. However this interaction is generally weak since the alpha donor atoms also donate to this orbital. As such, fisher carbenes are characterized as having partial double bond character. The major resonance structures of Fisher carbenes put the negative charge on the metal centre, and the positive on the carbon atom, making it electrophilic.[6]
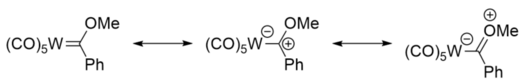
Fischer carbenes can be likened to ketones, with the carbene carbon atom being electrophilic, like the carbonyl carbon atom of a ketone. This can be seen from the resonance structures, where there is a significant contribution from the structure bearing a positive carbon centre.[6] Like ketones, Fischer carbene species can undergo aldol-like reactions. The hydrogen atoms attached to the carbon atom α to the carbene carbon atom are acidic, and can be deprotonated by a base such as n-butyllithium, to give a nucleophile, which can undergo further reaction.[7]
Schrock carbenes

Schrock carbenes do not have π-accepting ligands on the metal centre, and are typically found with:[6]
- high oxidation state metal center
- early transition metals Ti(IV), Ta(V)
- σ-donor and sometimes π-donor metal ligands
- hydrogen and alkyl substituents on carbenoid carbon.
Examples include ((CH3)3CCH2)Ta=CHC(CH3)3[9] and Os(PPh3)2(NO)Cl(=CH2).[10]

Bonding in such complexes can be viewed as the coupling of a triplet state metal and triplet carbene, forming a true double bond. Both the metal and carbon atom donate 2 electrons, one to each bond. Since there is no donation to the carbene atom from adjacent groups, the extent of pi backbonding is much greater, giving a strong double bond. These bonds are weakly polarized towards carbon and therefore the carbene atom is a nucleophile. Furthermore, the major resonance structures of Schrock carbene put the negative charge on the carbon atom, making it nucleophilic.[6] Complexes with the methylidene ligand (=CH2) are the simplest Schrock-type carbenes.

N-Heterocyclic carbenes
Bimetallic carbene complexes
An early example of this bonding mode was provided by [C5Me5Mn(CO)2]2(μ−CO) prepared from diazomethane:
- 2 C5Me5Mn(CO)2(thf) + CH2N2 → [C5Me5Mn(CO)2]2(μ−CH2] + N2 + 2 thf
Another example of this family of compounds is
Application of Metal Carbenes
Metal carbene complexes have applications in hetereogeneous and homogeneous catalysis, and as reagents for organic reactions.
Catalysis

The dominant application of metal carbenes involves none of the above classes of compounds, but rather
A variety of homogeneous carbene catalysts, especially the
Stoichiometric reactions
Homogeneous Schrock-type carbene complexes such as Tebbe's reagent can be used for the olefination of carbonyls, replacing the oxygen atom with a methylidene group. The nucleophilic carbon atom behaves similarly to the carbon atom of the phosphorus ylide in the Wittig reaction, attacking the electrophilic carbonyl atom of a ketone, followed by elimination of a metal oxide.[1]

In the nucleophilic abstraction reaction, a methyl group can be abstracted from the donating group of a Fischer carbene, making it a strong nucleophile for further reaction.[6]

Diazo compounds like

Wulff-Dötz Reaction
Fischer carbenes are used with alkynes as the starting reagents for the Wulff–Dötz reaction, forming phenols.[20]

History
The first metal carbene complex to have been reported was Chugaev's red salt, first synthesized as early as 1925, although it was never identified to be a carbene complex.[21] The characterization of (CO)5W(COCH3(Ph)) in the 1960s is often cited as the starting point of the area and Ernst Otto Fischer, for this and other achievements in organometallic chemistry, was awarded the 1973 Nobel Prize in Chemistry.[22] In 1968, Hans-Werner Wanzlick and Karl Öfele separately reported metal-bonded N-heterocyclic carbenes.[6][23][24] The synthesis and characterization of ((CH3)3CCH2)Ta=CHC(CH3)3 by Richard R. Schrock in 1974 marked the first metal alkylidene complex.[9] In 1991, Anthony J. Arduengo synthesized and crystallized the first persistent carbene, an NHC with large adamantane alkyl groups, accelerating the field of N-heterocarbene ligands to its current use.[5][6]
See also
References
- ^ ISBN 978-3-527-29390-2.
- ^ ISSN 0009-2665.
- ^ ISSN 0570-0833.
- ^ .
- ^ .
- ^ ISSN 0010-8545.
- ISBN 978-0-471-66256-3.
- hdl:1808/17672.
- ^ ISSN 0002-7863.
- ISSN 0002-7863.
- PMID 18398856.
- PMID 19900893.
- .
- PMID 25621143.
- .
- .
- .
- ISSN 1381-1169.
- ISSN 1460-4744.
- ISBN 978-0-471-26418-7.
- ^ ISSN 1433-7851.
- ISSN 0570-0833.
- ISSN 0570-0833.
- ISSN 0022-328X.
