Organoruthenium chemistry
Organoruthenium chemistry is the
In its organometallic compounds, ruthenium is known to adopt oxidation states from -2 ([Ru(CO)4]2−) to +6 ([RuN(Me)4]−). Most common are those in the 2+ oxidation state, as illustrated below.
-
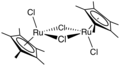
(cymene)ruthenium dichloride dimer
Ligands
As with other late transition metals, ruthenium binds more favorably with soft ligands.[3] The most important ligands for ruthenium are:
- halides, especially chloride.
- phosphines, especially triphenylphosphine.
- N-heterocyclic carbenes(NHCs).
- cyclopentadienyl ligands.
- various arenes and dienes
- carbon monoxide.
- hydride, notably in the Shvo catalyst.
- metal carbenes, notably in the Grubbs catalyst.
Phosphine ligands
While monodentate phosphine ligands such as
N-Heterocyclic carbene ligands
NHC ligands have become very common in organoruthenium complexes.[8][9] NHC ligands can be prepared with precise steric and electronic parameters, and can be chiral for use in asymmetric catalysis.[10] NHCs, as strongly donating L-type ligands, are often used to replace phosphine ligands. A notable example is 2nd generation Grubbs catalyst, in which a phosphine of the 1st generation catalyst is replaced by an NHC.
Cyclopentadienyl ligands
The parent compound
Chloro(cyclopentadienyl)bis(triphenylphosphine)ruthenium features a reactive chloro group, which is readily substituted by organic substrates.
Arene and alkene ligands
One example of an Ru-arene complex is
Multinuclear organo-ruthenium complexes have been investigated for anti-cancer properties. The compounds studied include di-, tri-, and tetra-nuclear complexes and tetrara-, hexa-, and octa- metalla-cages.[2]
Carbonyls
The main ruthenium carbonyl is
- Ru3(CO)12 + 3 CO 3 Ru(CO)5
Carbonylation of ruthenium trichloride gives a series of Ru(II) chlorocarbonyls. These are the precursors to Ru3(CO)12.
Organoosmium compounds
In the same
The 5d-orbitals in Os are higher in energy that the 4d-orbitals in Ru. Thus,
Important compounds, at least for academic studies, are the carbonyls such as triosmium dodecacarbonyl and decacarbonyldihydridotriosmium. The phosphine complexes are analogous to those or ruthenium, but hydride derivatives, e.g. OsHCl(CO)(PPh3)3, tend to be more stable.[19]
References
- ^ Synthesis of Organometallic Compounds: A Practical Guide Sanshiro Komiya Ed. S. Komiya, M. Hurano 1997
- ^ PMID 29394025.
- .
- ^ Example: Organic Syntheses, Coll. Vol. 10, p.276 (2004); Vol. 77, p.1 (2000). Link
- ^ Example: Organic Syntheses, Organic Syntheses, Coll. Vol. 9, p.589 (1998); Vol. 71, p.1 (1993). Link
- ^ Example: Organic Syntheses, Coll. Vol. 9, p.169 (1998); Vol. 72, p.74 (1995). Link
- ^ Example: Organic Syntheses, Vol. 81, p.178 (2005). Link
- PMID 19449832.
- PMID 19534492.
- PMID 21235210.
- PMID 20095576.
- ^ Organic Syntheses, Organic Syntheses, Vol. 82, p.10 (2005).Link
- ^ Example: Organic Syntheses, Organic Syntheses, Vol. 82, p.188 (2005). Link
- .
- PMID 33809231.
- PMID 29714397.
- ISBN 978-0-12-812838-1.
- PMID 37195088.
- ^ Perry, Paxtan (2022). "The Synthesis and Analysis of Triosmium Carbonyl Clusters with Potential Biological Activity" (PDF). Drew University. Retrieved 2024-05-08.