Organotechnetium chemistry
Organotechnetium chemistry is the science of describing the physical properties, synthesis, and reactions of organotechnetium compounds, which are
In general, organotechnetium compounds are not typically used in
Brief history
Technetium were first used as a radiopharmaceutical in 1961.
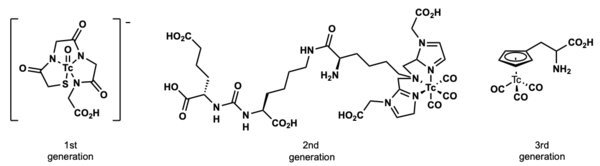
Examples
A vast majority of technetium compounds used in radiopharmaceutical imaging and diagnosis are inorganic coordination complexes. There are a number of “classical” organometallic organotechnetium compounds, specifically containing carbon-technetium bonds are in clinical use. These organotechnetium compounds are mostly seen as technetium tri-carbonyl compounds and technetium cyclopentadienyl compounds.

One of the most prominent radio pharmaceutical compounds in clinical use is Cardiolie®, also known as 99mTc-
The above
Synthesis
Radioactive 99mTc is obtained in the pertechnetate form in dilute aqueous solution from 99Mo/99mTc generators. Pertechnetate can then be made into more useful carbonyl and hydrate precursors for subsequent synthesis into technetate complexes.
As the starting radiometals are most available in aqueous solution due to method of isolation, the chemistry for synthesis of technetate compounds must be done in aqueous solution.
The study of technetium compounds is typically done in conjugation with rhenium as an isoelectronic and non-radioactive alternative to technetium.
Precursors
For 99mTc and 188Re, the synthesis of compounds start with pertechnetate or

Typical methods of organometallic compounds synthesis difficult to utilize. To be useful as a radiopharmaceutical, the reaction should be done in an aqueous saline solution that can be injected into the body intravenously.
Double Ligand Transfer
A double ligand transfer (DLT) reaction was developed by Martin Wenzel for synthesis of organotechnetium/organorhenium complexes.

Mechanism
This mechanism is proposed to proceed by ring slippage. First, reduction and carbonylation of the pertechnetate/perrhenate with CrCl3 and/or Cr(CO)6 to from the 6 coordinate intermediate. Subsequent reaction with the substituted ferrocene through ring-slipped, bridged intermediates then gives product. The transfer of the more electron deficient ring is favored by the stabilization of the transition state of η5- η3 ring slip of ferrocene.

Metal-Mediated Retro Diels-Alder
Aqueous synthesis enables development for medically relevant radiopharmaceuticals. First aqueous synthesis of fac-[99mTc(η5 -Cp-C(O)CH3)(CO)3] was described by the Alberto lab utilized a metal-mediated retro Diels-Alder to synthesize the organotechnetium complexes.[14]
Mechanism
In a step-wise manner, the carboxylate first coordinates to technetium followed by coordination to the adjacent cyclopentadiene (Path A). The reaction is thermodynamically driven, given a strong electronic interaction between [99mTc(CO)3]+ and the cyclopentadiene.

The favorable formation of the {(η5-Cp)Tc} as a driving force for formation of the product 2, prompted the use of the Diels-Ader dimer (HCp-COOH)2 (Thiele’s acid) as a precursor to the cyclopentadiene. Thermal cracking of 3 typically requires T >160 °C. Reaction of 3 and 1 at 95 °C for 30 min in buffer gave quantitative formation of 2. As no free HCp-COOH was observed, in situ retro Diels-Alder and subsequent entry into path A was excluded.
Examples
The metal-mediated retro Diels-Alder reaction suggests a general approach to [(Cp-R)99mTc(CO)3], enable access to a variety of R groups on the Cp ring.

With the development of this retro Diels-Alder method for synthesis of 99mTc and Re complexes in aqueous media by the Alberto lab, The labeling of biomolecules with piano-stool like complexes is now possible. Enabling access to the development of novel radiopharmaceuticals.
Reactivity
Technetium has been shown to react similarity to osmium. Able to catalyze a cis dihydroxylation.

References
- ^ Kowalsky RJ (2006). "Technetium Radiopharmaceutical Chemistry" (PDF). The University of New Mexico. Health Sciences Center, College of Pharmacy. pp. 1–77.
- ^ Ritter SK (April 2017). "Good reasons to study technetium chemistry". Chemical & Engineering News. 95 (15). Retrieved 2021-06-12.
- .
- PMID 21378873.
- ISSN 0276-7333.
- S2CID 19897272.
- PMID 15354226.
- OCLC 1079003267.
- S2CID 99371461.
- S2CID 219912503.
- PMID 660286.
- ISSN 0276-7333.
- ^ "John A. Katzenellenbogen | Chemistry at Illinois". chemistry.illinois.edu. Retrieved 2021-06-12.
- PMID 18186638.
