Organocadmium chemistry
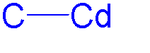
Organocadmium chemistry describes the physical properties, synthesis, reactions, and use of organocadmium compounds, which are
The simplest organocadmium compound is
Synthesis
Dimethylcadmium and diethylcadmium were reported in 1917 by Erich Krause. In general, they are prepared by transmetalation or by an exchange reaction between an alkylating agent and a cadmium salt.[3]
According to one procedure, diethylcadmium is produced by the reaction of
Fluoroalkyl and alkenyl derivatives
Following established trends, perfluorinated alkyl and alkenyl derivatives of cadmium exhibit improved thermal stability. The alkenyl derivatives are generated by the addition of iodotrifluoroethylene to cadmium metal.[4]
Reactions
Organocadmium compounds are less
Another synthetic use of an organocadmium is the reaction of diisoamylcadmium with β-carbomethoxypropionyl chloride to methyl 4-keto-7-methyloctanoate without reacting further with the ketone group or the ester group.[7]
This selectivity is observed provided that the reaction is carried out salt free.[8] When the cadmium reagent is generated in situ from a cadmium salt, the halide generates a more nucleophilic organocadmium reagent, an ate complex. The same salt effect can be observed with organozinc compounds.
Dimethylcadmium has been used to synthesize colloidal nanocrystals of II-VI materials such as cadmium selenide. Its toxic and volatile nature has led researchers to look elsewhere for cadmium precursors such as cadmium oxide.[9]
Toxicity
Cadmium compounds are toxic. Dimethylcadmium is toxic to the kidney, the liver, the central nervous system, and the respiratory organs when inhaled.[10] Cadmium compounds in general are considered to be carcinogen to humans by the IARC.[11]
References
- ISBN 3-13-103061-5
- PMID 27457504.
- .
- .
- ISBN 9780471264187.
- .
- doi:10.15227/orgsyn.028.0075.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - .
- PMID 11273619.
- PMID 1916391.
- ISBN 9789283201359.

