Organonickel chemistry

Organonickel chemistry is a branch of organometallic chemistry that deals with organic compounds featuring nickel-carbon bonds.[1][2] They are used as a catalyst, as a building block in organic chemistry and in chemical vapor deposition. Organonickel compounds are also short-lived intermediates in organic reactions. The first organonickel compound was nickel tetracarbonyl Ni(CO)4, reported in 1890 and quickly applied in the Mond process for nickel purification. Organonickel complexes are prominent in numerous industrial processes including carbonylations, hydrocyanation, and the Shell higher olefin process.[3][4]
Classes of compounds
Alkyl and aryl complexes
A popular reagent is Ni(CH3)2(tetramethylethylenediamine).[5]
Many alkyl and aryl complexes are known with the formula NiR(X)L2. Examples include [(dppf)Ni(cinnamyl)Cl)], trans-(PCy2Ph)2Ni(o-tolyl)Cl]], (dppf)Ni(o-tolyl)Cl]], (TMEDA)Ni(o-tolyl)Cl, and (TMEDA)NiMe2.

Nickel compounds of the type NiR2 also exist with just 12 valence electrons. In solution however solvent always interact with the metal atom increasing the electron count. One 12 VE compound is di(mesityl)nickel prepared from (allyl)2Ni2Br2 and the corresponding Grignard reagent.
- (allyl)2Ni2Br2 + 4 C6H2Me3MgBr → 2 (allyl)MgBr + 2 MgBr2 + 2 (C6H2Me3)2Ni
Alkene complexes
Many complexes exist of nickel coordinated to an
Allyl complexes
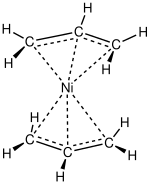
Nickel forms several simple
Cyclopentadienyl complexes
Nickelocene NiCp2 with +2 Ni oxidation state and 20 valence electrons is the main metallocene of nickel. It can be oxidized by one electron. The corresponding palladocene and platinocene are unknown. From nickelocene, many derivatives are generated, e.g. CpNiLCl, CpNiNO, and Cp2Ni2(CO)3.
Carbene complexes
Nickel forms carbene complexes, formally featuring C=Ni double bonds.[9]
Reactions
Alkene/alkyne oligomerizations
Nickel compounds catalyze the
One practical implementation of alkyne oligomerization is the
This is a formal [2+2+2+2]cycloaddition. The oligomerization of butadiene with ethylene to trans-1,4-hexadiene was an industrial process at one time.
Formal [2+2+2]cycloadditions also take place in
In the
The formation of organonickel compounds in this type of reaction is not always obvious but in a carefully designed experiment two such intermediates are formed quantitatively:[11][12]
It is noted in one study
The selected reaction conditions also minimize the amount formed of competing [2+2+2]cycloaddition product to the corresponding substituted arene.
Coupling reactions
Nickel compounds cause the
Ni carbonylation
Ni catalyzes the addition of carbon monoxide to alkenes and alkynes. The industrial production of acrylic acid at one time consisted of combining acetylene, carbon monoxide and water at 40-55 atm and 160-200 °C with nickel(II) bromide and a copper halide.
See also
- Nickel(IV) organometallic complex
- Nickel(II) precatalysts
- Lactate racemase
Further reading
- P.W. Jolly, G. Wilke, ed. (1974). The Organic Chemistry of Nickel Volume I: Organonickel Complexes. Academic Press. ISBN 9780123884015.
References
- ISBN 0-306-41199-7
- ISBN 0-08-044590-X
- .
- PMID 24828188.
- S2CID 224930545.
- PMID 25886092.
- S2CID 221124789.
- ^ Martin F. Semmelhack and Paul M. Helquist (1988). "Reaction of Aryl Halides with π-Allylnickel Halides: Methallylbenzene". Organic Syntheses. 52: 115; Collected Volumes, vol. 6, p. 161.
- S2CID 73515728.
- PMID 15886770.
- dihydropyridine









