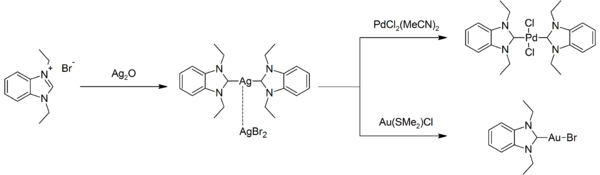Organosilver chemistry

Organosilver chemistry is the study of
The first attempts in organosilver were recorded by Buckton in 1859
Alkyl, alkenyl, aryl derivatives
Phenylsilver can be obtained by reaction of silver nitrate with an trialkylphenyllead or diphenylzinc:[6]
- Ph2Zn + AgNO3 → PhAg + "PhZnNO3"
Like all silver complexes, organosilver compounds have coordination numbers ≥2. For example,
- AgCl + (CH3)3C6H2MgBr → 1/4 [(CH3)3C6H2Ag]4 + MgClBr
A variety of organosilver compounds include phosphorus ylides. A simple example is the pentafluorophenylsilver complex of methylenetriphenylphosphorane:[8]
- AgC6F5 + Ph3P=CH2 → Ph3P=CH2−AgC6F5
Alkenylsilver compounds are also more stable than their alkylsilver counterparts. Vinylsilver can be obtained by reaction of silver nitrate with tetravinyllead:[9]
- AgNO3 + (CH2=CH)4Pb → (CH2=CH)Ag + (CH2=CH)3PbNO3
Fluoroalkyl and fluoroalkenyl derivatives
Following established trends, perfluorinated alkyl and alkenyl derivatives of silver exhibit significant thermal stability. An alkenyl derivatives are generated by the addition of silver fluoride to
- AgF + CF2=CF(CF3) → AgCF(CF3)2
Organosilver compounds usually have the oxidation state +1. A notable exception is Ag(CF3)4−.
Carbene and CO complexes
Silver forms relatively fragile complexes with CO, including [Ag(CO)n]+ (n = 1, 2, 3).[12] The green, planar, paramagnetic Ag(CO)3 is stable at 6–15 K and dimerizes at 25–30 K, probably by forming Ag–Ag bonds. Additionally, the silver carbonyl [Ag(CO)] [B(OTeF5)4] is known.
Silver-
Alkene complexes
Like other heavy d10 metal ions, Ag+ has a pronounced affinity for alkenes. The ability of silver to form
Catalysis
In catalysis silver is active as
Further reading
- W.A. Herrmann, ed. (1999). Synthetic Methods of Organometallic and Inorganic Chemistry. Vol. 5, Copper, Silver, Gold, Zinc, Cadmium, and Mercury. Stuttgart: Thieme. ISBN 3-13-103061-5.
- Christoph Elschenbroich (2006). Organometallics (3 ed.). Weinheim: Wiley-VCH. ISBN 3-527-29390-6.
- The Chemistry of Organic Derivatives of Gold and Silver. Edited by Saul Patai and Zvi Rappoport Copyright 1999 John Wiley & Sons, Ltd. ISBN 0-471-98164-8
References
- ISBN 9780470597521.
- .
- .
- .
- ^ Greenwood and Earnshaw, pp. 1199–200
- .
- .
- .
- .
- .
- .
- doi:10.1039/A908459B.
- .
- S2CID 94984101.
- ^ Boryana Nikolova-Damyanova. "Principles of Silver Ion Complexation with Double Bonds".
- .