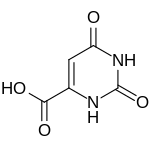
Orotic acid
 | |
| Clinical data | |
|---|---|
| Other names | uracil-6-carboxylic acid |
| AHFS/Drugs.com | International Drug Names |
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Orotic acid (vitamin B complex and was called vitamin B13, but it is now known that it is not a vitamin.
The compound is synthesized in the body via a
dietary supplements (to increase their bioavailability), most commonly for lithium orotate
.
Synthesis
ribose sugar, whereas purine synthesis happens by building the base directly on the sugar.[3]
Chemistry
Orotic acid is a
anion, is able to bind to metals. Lithium orotate, for example, has been investigated for use in treating alcoholism,[4][5] and complexes of cobalt, manganese, nickel, and zinc are known.[6] The pentahydrate nickel orotate coordination complex converts into a polymeric trihydrate upon heating in water at 100 °C.[6][7][8] Crystals of the trihydrate can be obtained by hydrothermal treatment of nickel(II) acetate and orotic acid. When the reactions are run with bidentate nitrogen ligands such as 2,2'-bipyridine
present, other solids can be obtained.
Pathology
A buildup of orotic acid can lead to
urea cycle disorder
.
In
tricarboxylic acid cycle (TCA) decreasing adenosine triphosphate
(ATP) production.
Orotic aciduria is a cause of megaloblastic anaemia.
Biochemistry
Orotic acid is a precursor to a RNA base, uracil. [10] The breast milk of smokers has a higher concentration of orotic acid than that of a non smoking woman. It is reasoned that the smoking causes the pyrimidine biosynthesis process in the mother to be altered thus causing the orotic acid concentration to increase.[11]
A modified orotic acid (5-fluoroorotic acid) is toxic to yeast. The mutant yeasts which are resistant to 5-fluoroorotic acid require a supply of uracil.[12]
See also
- Magnesium orotate
- Pyrimidine biosynthesis
References
- ^ "Orotic acid". Merriam-Webster.com Dictionary. Retrieved 2023-02-07.
- PMID 10727948.
- ^ Harvey D, Ferrier D, eds. (2008). Biochemistry (PDF) (5th ed.). Lippincott, Williams & Wilkins. p. 302.
- .
- PMID 3718672.
- ^ .
- doi:10.5517/cc6cgmm.
- .
- S2CID 25297304.
- .
- S2CID 29181790.
- S2CID 84498320.
Further reading
- Greenbaum SB (1954). "Potential Metabolic Antagonists of Orotic Acid: 6-Uracilsulfonamide and 6-Uracil Methyl Sulfone". Journal of the American Chemical Society. 76 (23): 6052–6054. .
External links
Wikimedia Commons has media related to Orotic acid.
- Orotic+Acid at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
