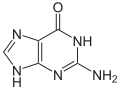
Hypoxanthine

| |
| Names | |
|---|---|
| Preferred IUPAC name
1,9-Dihydro-6H-purin-6-one | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.000.634 |
IUPHAR/BPS |
|
| KEGG | |
| MeSH | Hypoxanthine |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H4N4O | |
| Molar mass | 136.112 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Hypoxanthine is a naturally occurring
tRNA in the form of its nucleoside inosine. It has a tautomer known as 6-hydroxypurine. Hypoxanthine is a necessary additive in certain cells, bacteria, and parasite cultures as a substrate and nitrogen source. For example,[1][2] it is commonly a required reagent in malaria parasite cultures, since Plasmodium falciparum
requires a source of hypoxanthine for nucleic acid synthesis and energy metabolism.
In August 2011, a report, based on
organic molecules, including the DNA and RNA components adenine and guanine, may have been formed extraterrestrially in outer space.[3][4][5]
The Pheretima aspergillum worm, used in Chinese medicine preparations, contains hypoxanthine.[6]
Reactions
It is one of the products of the action of
xanthine oxidoreductase
.
IMP in nucleotide salvage
.
Hypoxanthine is also a spontaneous
DNA transcription/replication, as it base pairs with cytosine
. Hypoxanthine is removed from DNA by base excision repair, initiated by N-methylpurine glycosylase (MPG), also known as alkyl adenine glycosylase (Aag).
[7]
Additional images
References
- ^ "Estimation of Plasmodium falciparum drug susceptibility by the 3H-hypoxanthine uptake inhibition assay" (PDF). Worldwide Antimalarial Resistance Network. Archived (PDF) from the original on 2022-10-09. Retrieved 2017-01-20.
- PMID 11132385.
- PMID 21836052.
- ^ Steigerwald, John (8 August 2011). "NASA Researchers: DNA Building Blocks Can Be Made in Space". NASA. Retrieved 2011-08-10.
- ^ ScienceDaily Staff (9 August 2011). "DNA Building Blocks Can Be Made in Space, NASA Evidence Suggests". ScienceDaily. Retrieved 2011-08-09.
- ^ The Pharmacology of Chinese Herbs, Second Edition By Kee C. Huang
- PMID 19219989.
External links
- Hypoxanthine at the U.S. National Library of Medicine Medical Subject Headings (MeSH)