Phenoxy herbicide

Phenoxy herbicides (or "phenoxies") are two families of chemicals that have been developed as commercially important herbicides, widely used in agriculture. They share the part structure of phenoxyacetic acid.
Auxins
The first group to be discovered act by mimicking the auxin growth hormone
(corn), they selectively kill broad-leaf weeds, leaving the crops relatively unaffected.-
IAA
-
MCPA
-
2,4-D
-
2,4,5-T
Introduced in 1946, these
-
Mecoprop
-
(2R)-Dichlorprop
-
Fenoprop
-
2,4-DB
-
MCPB
Other members of this group include 4-(2,4-dichlorophenoxy)butyric acid (

The use of herbicides in US agriculture is mapped by the US Geological Survey. As of 2019[update], 2,4-D was the most used of the auxins. 45,000,000 pounds (20,000,000 kg) were sprayed that year,
ACCase inhibitors
In the 1970s, agrochemical companies were working to develop new herbicides to be complementary to the auxins. The aim was to find materials which would selectively control grass weeds in broad-leaf crops such as cotton and soybean.

Diclofop: X=CH, R1=R2=Cl
Chlorazifop: X=N, R1=R2=Cl
Fluazifop: X=N, R1=CF3, R2=H
Haloxyfop: X=N, R1=CF3, R2=Cl
In 1973,
This group of herbicides acts by inhibiting plant
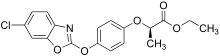

Salts and esters of this class of herbicide are active owing to their ability to metabolise to the corresponding parent acid. For example, fenoxaprop-P ethyl
Resistance
Cummins et al., 1999, 2009, and 2013 find that
References
- PMID 19823992.
- S2CID 85637273.
- ISBN 978-0-08-095168-3.
- ISBN 9780120007547.
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for 2,4-D, 2019". Retrieved 2021-12-27.
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for MCPA, 2018". Retrieved 2021-12-27.
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for Dicamba, 2019". Retrieved 2021-12-27.
- ^ Gray, Bryce (2016-11-09). "EPA approves Monsanto's less-volatile form of dicamba herbicide". St. Louis Post-Dispatch. Retrieved 2021-12-27.
- ^ a b Evans, D. (1992). "Designing more efficient herbicides" (PDF). Proceeding of the First International Weed Control Congress , Melbourne. pp. 37–38. Retrieved 2021-02-27.
- ^ "Aryloxyphenoxypropionic herbicides". BCPC. Retrieved 2022-10-06.
- PMID 2902848.
- S2CID 27124700.
- PMID 12859251.
- ISBN 9783527339471.
- ^ Pesticide Properties Database. "Fenoxaprop-P-ethyl". University of Hertfordshire. Retrieved 2021-03-02.
- ^ Pesticide Properties Database. "Quizalofop-P-ethyl". University of Hertfordshire. Retrieved 2021-03-02.
- ^ Pesticide Properties Database. "Cyhalofop-butyl". University of Hertfordshire. Retrieved 2022-10-06.
- ^ Pesticide Properties Database. "Fluazifop-P-butyl". University of Hertfordshire. Retrieved 2021-03-02.
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for Fluazifop, 2018". Retrieved 2021-12-27.
- S2CID 246978319.







