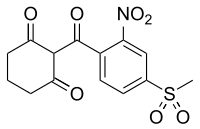
Mesotrione
| |
| Names | |
|---|---|
| IUPAC name
2-(4-mesyl-2-nitrobenzoyl)cyclohexane-1,3-dione
| |
| Preferred IUPAC name
2-[4-(Methylsulfonyl)-2-nitrobenzoyl]cyclohexane-1,3-dione | |
| Other names
ZA1296
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.111.661 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C14H13NO7S | |
| Molar mass | 339.32 g·mol−1 |
| Appearance | Yellow to tan coloured solid |
| Density | 1.49 g/cm3 |
| Melting point | 165.3 °C (329.5 °F; 438.4 K) |
| 1500 mg/L (20 °C) | |
| log P | 0.11 |
| Acidity (pKa) | 3.12 |
| Hazards[2] | |
| GHS labelling: | |

| |
| Warning | |
| H410 | |
| P273, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mesotrione is a
History
The invention of the triketone class of herbicides had its beginnings in an observation in 1977 of
The triketone herbicides were found to be effective on a wide range of commercially-important weed species and to have both pre- and post-emergence activity. Mesotrione was chosen for development (by Zeneca Agrochemicals under the code number ZA1296) because it controls a wide range of broad-leaved weeds that compete with maize and can also suppress some annual grass weeds that may be present in the crop. It achieves this selectivity and lack of damage to the crop owing to its greater potency on the target enzyme found in dicotyledons than monocotyledons[9] and because maize can metabolise the compound in the dione-containing ring.[6]

Synthesis
The synthesis of mesotrione[9] was first disclosed in patents[10] filed by ICI, who had acquired Stauffer in 1987.[11] 1,3-Cyclohexanedione is first reacted with the acid chloride of 4-(methylsulfonyl)-2-nitrobenzoic acid under conditions in which the enolic hydroxyl group of the diketone reacts to form the benzoylated derivative. In a separate step, this is rearranged to mesotrione using a catalytic amount of cyanide ion derived from acetone cyanohydrin.
Mechanism of action
Mesotrione inhibits the enzyme
Formulations
Mesotrione is made available to end-users only in formulated products. These use non-powdery material with reduced or no use of hazardous solvents, for example suspension concentrates. The herbicide is compatible with other compounds that may be mixed by the farmer to extend control to the grass weeds which mesotrione itself does not kill.[13]
Usage

Mesotrione is a systemic pre- and post-emergence herbicide for the selective contact and residual control of broadleaf weeds in field corn, seed corn, yellow popcorn and sweet corn.
The estimated annual use of mesotrione in US agriculture is mapped by the US Geological Service and shows an increasing trend from its introduction in 2001 to 2018, the latest date for which figures are available and now reaching 4,500,000 pounds (2,000,000 kg). As would be expected for a compound used almost exclusively in maize, the heaviest use is in the
Human safety
The
Effects on the environment
Mesotrione is very toxic to aquatic life with long lasting effects.[14] Its ecotoxicology is summarised in the Pesticide Properties database.[1]
Resistance management
Reports of individual weed species, for example Amaranthus tuberculatus, becoming resistant to mesotrione[19] are monitored by manufacturers, regulatory bodies such as the EPA and the Herbicides Resistance Action Committee (HRAC).[20] In some cases, the risks of resistance developing can be reduced by using a mixture of two or more herbicides which each have activity on relevant weeds but with unrelated mechanisms of action. HRAC assigns active ingredients into classes so as to facilitate this.
Brands
By international convention and in many countries the law, pesticide labels are required to include the common name of the active ingredients. These names are not the exclusive property of the holder of any patent or trademark and as such they are the easiest way for non-experts to refer to individual chemicals. Companies selling pesticides normally do so using a brand name or wordmark which allows them to distinguish their product from competitor products having the same active ingredient. In many cases, this branding is country and formulation-specific so there can be multiple brand names for a given active ingredient. Mesotrione may be pre-mixed with other herbicides to provide more complete weed control. For example, Acuron is the name used by Syngenta for a mixture containing bicyclopyrone, atrazine and S-metolachlor in addition to mesotrione. Brand names for mesotrione include Callisto, Instigate, Meristo, Resicore and Tenacity. Suppliers and brand names in the United States are listed in the National Pesticide Information Retrieval System.[21]
See also
- Nitisinone (orfadin)
References
- ^ a b c d Pesticide Properties Database. "Mesotrione". University of Hertfordshire.
- ^ PubChem Database. "Mesotrione".
- ^ PMID 15581571. Archived from the original(PDF) on 2014-03-03.
- ^ a b Uttley, Nigel (June 3, 2011). "Product Profile: Mesotrione". AgriBusiness global. Retrieved 2020-03-28.
- .
- ^ PMID 19349184.
- ISBN 978-94-010-5817-9.
- ^ Derek Cornes (2005). "Callisto: a very successful maize herbicide inspired by allelochemistry". Fourth World Congress on Alleopathy. The Regional Institute Ltd. Retrieved March 28, 2020.
- ^ PMID 11455642.
- ^ US patent 5006158, Carter, C.G.; Lee, D.L. & Michaely, W.J. et al., "Certain 2-(2-substituted benzoyl)-1,3-cyclohexanediones", issued 1991-04-09, assigned to ICI Americas Inc
- ^ Hicks, Jonathan P. (June 6, 1987). "Imperial set to buy Stauffer". The New York Times. Retrieved 2020-03-28.
- ^ "Tenacity Herbicide". Syngenta.
- ^ a b c d Syngenta US. "Callisto herbicide US label".
- ^ a b c Syngenta UK. "Callisto UK label" (PDF).
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for mesotrione, 2018". Retrieved 2022-01-17.
- ]
- ^ Low, K.; Tasheva, M. "Mesotrione" (pdf).
- ^ FAO / WHO. "Mesotrione".
- PMID 28662111.
- ^ "Herbicides Resistance Action Committee website".
- ^ NPIRS Public. "Search Federal Pesticide Products".
Further reading
- Singh, H. P.; Batish, Daizy R.; Wikidata Q111370060.
- Dayan, Franck E.; Owens, Daniel K.; Duke, Stephen O. (2012). "Rationale for a natural products approach to herbicide discovery" (PDF). Pest Management Science. 68 (4): 519–528. PMID 22232033.
External links
- Mesotrione in the Pesticide Properties DataBase (PPDB)




