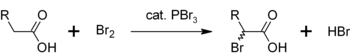Phosphorus tribromide

| |

| |

| |
| Names | |
|---|---|
| IUPAC name
Phosphorus tribromide
| |
| Other names
phosphorus(III) bromide,
phosphorous bromide, tribromophosphine | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.029.253 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| PBr3 | |
| Molar mass | 270.69 g/mol |
| Appearance | clear, colourless liquid |
| Density | 2.852 g/cm3 |
| Melting point | −41.5 °C (−42.7 °F; 231.7 K) |
| Boiling point | 173.2 °C (343.8 °F; 446.3 K) |
| rapid hydrolysis | |
Refractive index (nD)
|
1.697 |
| Viscosity | 0.001302 Pas |
| Structure | |
| trigonal pyramidal | |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H314, H335 | |
| P260, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
phosphorus trifluoride phosphorus trichloride phosphorus triiodide |
Other cations
|
nitrogen tribromide arsenic tribromide antimony tribromide |
Related compounds
|
phosphorus oxybromide
|
| Supplementary data page | |
| Phosphorus tribromide (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phosphorus tribromide is a colourless liquid with the formula
Preparation
PBr3 is prepared by treating
- P4 + 6 Br2 → 4 PBr3
Because the reaction is highly exothermic, it is often conducted in the presence of a diluent such as PBr3. Phosphorus tribromide is also generated in situ from
Reactions
Phosphorus tribromide, like
An important reaction of PBr3 is with
- PBr3 + 3 (CH3)2CHCH2OH → 3 (CH3)2CHCH2Br + HP(O)(OH)2
Several detailed procedures are available.[5][6] In some cases, triphenylphosphine/Br2 is superior to PBr3.[7]
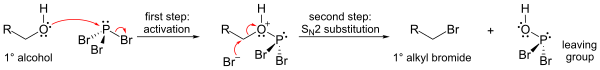
The mechanism for a primary alcohol involves formation of a phosphorous ester (to form a good leaving group), followed by an SN2 substitution.
Because of the SN2 substitution step, the reaction generally works well for primary and secondary alcohols, but fails for tertiary alcohols. If the reacting carbon centre is chiral, the reaction usually occurs with inversion of configuration at the carbon alpha to the alcohol, as is usual with an SN2 reaction.
In a similar reaction, PBr3 also converts carboxylic acids to acyl bromides:[8]
- PBr3 + 3 RCO2H → 3 RCOBr + HP(O)(OH)2
Applications
The main use for phosphorus tribromide is for conversion of primary or secondary
Another use for PBr3 is as a catalyst for the α-bromination of
On a commercial scale, phosphorus tribromide is used in the manufacture of
Phosphorus tribromide is used for doping in microelectronics.[12]
Precautions
PBr3 evolves corrosive HBr, which is toxic, and reacts violently with water and alcohols.
- PBr3 + 3 H2O → H3PO3 + 3 HBr
In reactions that produce phosphorous acid as a by-product, when working up by distillation be aware that this can decompose above about 160 °C to give phosphine which can cause explosions in contact with air.[9]
References
- ^ J. F. Gay, R. N. Maxson "Phosphorus(III) Bromide" Inorganic Syntheses, 1947, vol. 2, 147ff.
- .
- .
- .
- .
- .
- doi:10.15227/orgsyn.048.0051.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- ^ a b Harrison, G. C.; Diehl, H. (1955). "β-Ethoxyethyl Bromide". Organic Syntheses; Collected Volumes, vol. 3, p. 370.
- ^ Wade, L. G. Jr. (2005). Organic Chemistry (6th ed.). Upper Saddle River, NJ, USA: Pearson/Prentice Hall. p. 477.
- ^ Wade, L. G. Jr. (2005). Organic Chemistry (6th ed.). Upper Saddle River, NJ, USA: Pearson/Prentice Hall. p. 1051.
- ISSN 0021-8979.
Further reading
- ISBN 978-0-08-037941-8.
- Lide, D. R., ed. (1990). Handbook of Chemistry and Physics (71st ed.). Ann Arbor, MI: CRC Press. ISBN 978-0849304712.
- March, J. (1992). Advanced Organic Chemistry (4th ed.). New York: Wiley. p. 723. ISBN 978-0471601807.
- Stecher, P. G., ed. (1960). The Merck Index (7th ed.). Rahway, NJ, USA: Merck & Co.
- Holmes, R. R. (1960). "An Examination of the Basic Nature of the Trihalides of Phosphorus, Arsenic and Antimony". .