Plethodontidae
| Lungless salamander Temporal range:
| |
|---|---|

| |
| California slender salamander (Batrachoseps attenuatus) | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Class: | Amphibia |
| Order: | Urodela |
| Suborder: | Salamandroidea |
| Family: | Plethodontidae Gray, 1850 |
| Subgroups | |
| |
| |
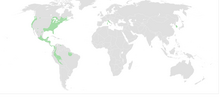
| Native distribution of plethodontids (in green) | |
Plethodontidae, or lungless salamanders, are a family of salamanders.[1][2] With over 500 species, lungless salamanders are by far the largest family of salamanders in terms of their diversity. Most species are native to the Western Hemisphere, from British Columbia to Brazil. Only two extant genera occur in the Eastern Hemisphere: Speleomantes (native to Sardinia and mainland Europe south of the Alps) and Karsenia (native to South Korea).[3]
Biology
Adult lungless salamanders have four limbs, with four toes on the fore limbs, and usually with five on the hind limbs. Within many species, mating and reproduction occur solely on land. Accordingly, many species also lack an aquatic larval stage, a phenomenon known as direct development in which the offspring hatch as fully-formed, miniature adults. Direct development is correlated with changes in the developmental characteristics of plethodontids compared to other families of salamanders including increases in egg size and duration of embryonic development. Additionally, the evolutionary loss of the aquatic larval stage is related to a diminishing dependence on aquatic habitats for reproduction. The lift of this constraint allowed widespread colonization and diversification within a broad number of terrestrial habitats which is a testament to the high success and proliferation of Plethodontidae.[4]
Despite the absence of lungs, some can grow rather large. The largest species of lungless salamanders, Bell's false brook salamander, can reach lengths of 36 cm (14 in).[5]
Many species have a
Measured in individual numbers, they are very successful animals where they occur. In some places, they make up the dominant biomass of vertebrates.
Courtship and mating
Plethodontids exhibit highly stereotyped and complex mating behaviors and courtship rituals that are not present in any other salamander family. Mating behavior tends to be uniform among all plethodontids and typically involves a tail-straddle walk in which the female orients her head at the base of the male's tail while also straddling the tail with her body. The male will twist his body around and deposit a sperm capsule, known as the spermatophore, on the substrate in front of the female's snout. As the male leads the female over the spermatophore with his tail, the female lowers her cloaca onto the spermatophore and lodges the sperm mass inside while leaving the base of the spermatophore on the ground.[8]
Within many species of plethodontidae, the courtship ritual is often accompanied by transfer of male pheromones during the tail-straddling walk. During the breeding period, males will grow enlarged anterior teeth used to scratch the female's skin on her head as a part of the courtship ritual. Subsequently, the male will rub pheromones onto the abraded spot which are secreted from a pad of tissue called the mental gland located underneath the male's chin.[8][9][10][11]
Courtship pheromones greatly increase male mating success for a variety of reasons. Overall, the pheromone secretions increase female receptivity to courtship and sperm transfer. This not only increases the likelihood of successful mating with a specific female, but also shortens the duration of courtship which is important because it minimizes the chance of the male being interrupted by other competing males.[12]
In scientific literature discussing the variations between the mental glands of plethodontid salamanders, it was discovered that male plethodontids had minor variations in height and diameter of the simple tubular glands, and major variation was found in the diameter of the secretory granules. This is attributed to the fact that males can mate throughout all months of the year, while females oviposit seasonally.
Respiration
A number of features distinguish the plethodontids from other salamanders. Most significantly, they lack lungs, conducting respiration through their skin, and the tissues lining their mouths.[3] Some species of cave salamanders are neotenic, and keep their larval gills even as adults. Gills are absent in all other adult plethodontids.[13] Plethodontids possess costal grooves on the trunk of their bodies. These help keep the skin moist via water transport over the surface of the body.[14]
Plethodontid salamanders are almost entirely reliant on cutaneous respiration.[15] Approximately 83%–93% of oxygen uptake is through this method.[16] Plethodontid salamander respiration rates are constrained by their SA:V, and higher SA:Vs are correlated to warmer, wetter climates.[17]
Plethodontids are constantly exposed to air or water, which allows for constant gas exchange that is not limited by ventilation.[15] Oxygen uptake is identical in water and air, assuming the partial pressure of oxygen is the same.[18] Oxygenated and non-oxygenated blood are mixed together in the venous system, which causes the partial pressure of oxygen within cardiac blood to typically be low.[15]
Plethodontids can tolerate
Plethodontids have been observed to develop rudimentary lungs as embryos.[19] The lung rudiment develops similarly to that of non-plethodontid salamanders for the first three weeks of development and then begins to regress through apoptosis.[19]

Chemoreception
Another distinctive feature is the presence of a vertical slit between the nostril and upper lip, known as the "nasolabial groove". The groove is lined with glands, and enhances the salamander's
Evolutionary history
Plethodontidae are estimated to have split from their sister group
Subfamilies and genera
The family Plethodontidae consists of two extant subfamilies and about 516[2] to 520[1] species divided among these genera, making up the majority of known salamander species:[2]
| Subfamily | Genus, scientific name, and author | Common name | Species |
|---|---|---|---|
| Hemidactyliinae Hallowell, 1856 | |||
| Aquiloeurycea Rovito, Parra-Olea, Recuero, and Wake, 2015 | 6 - 7
| ||
Batrachoseps Bonaparte, 1839 |
Slender salamanders | 23
| |
| Bolitoglossa Duméril, Bibron & Duméril, 1854 | Tropical climbing salamanders | 139 - 140
| |
| Bradytriton Wake & Elias, 1983 | Finca Chiblac salamander | 1
| |
| Chiropterotriton Taylor, 1944 | Splay-foot salamanders | 23
| |
| Cryptotriton García-París & Wake, 2000 | Hidden salamanders | 7
| |
| Dendrotriton Wake & Elias, 1983 | Bromeliad salamanders | 8
| |
Eurycea Rafinesque, 1822 |
North American brook salamanders | 34
| |
| Gyrinophilus Cope, 1869 | Spring salamanders | 4
| |
Hemidactylium Tschudi, 1838 |
Four-toed salamanders | 1
| |
| Isthmura Dubois and Raffaelli, 2012 | 7
| ||
| Ixalotriton Wake and Johnson, 1989 | Jumping salamanders | 2
| |
| Nototriton Wake & Elias, 1983 | Moss salamanders | 22
| |
| Nyctanolis Elias & Wake, 1983 | Long-limbed salamanders | 1
| |
| Oedipina Keferstein, 1868 | Worm salamanders | 40
| |
| Parvimolge Taylor, 1944 | Tropical dwarf salamanders | 1
| |
| Pseudoeurycea Taylor, 1944 | False brook salamanders | 41
| |
| Pseudotriton Tschudi, 1838 | Mud and red salamanders | 2 - 4
| |
| Stereochilus Cope, 1869 | Many-lined salamanders | 1
| |
| Thorius Cope, 1869 | Minute salamanders | 29
| |
| Urspelerpes Camp, Peterman, Milanovich, Lamb, Maerz, and Wake, 2009 | Patch-nosed salamanders | 1
| |
| Plethodontinae Gray, 1850 | |||
Aneides Baird, 1851 |
Climbing salamanders | 10
| |
| Desmognathus Baird, 1850 | Dusky salamanders | 39
| |
| Ensatina Gray, 1850 | Ensatinas | 1
| |
| Hydromantes Gistel, 1848 | Web-toed salamanders | 5
| |
Karsenia Min, Yang, Bonett, Vieites, Brandon & Wake, 2005 |
Korean crevice salamanders | 1
| |
Phaeognathus Highton, 1961 |
Red Hills salamanders | 1
| |
| Plethodon Tschudi, 1838 | Slimy and mountain salamanders | 58
| |
| Speleomantes Dubois, 1984 | European cave salamanders | 8
|
Following a major revision in 2006, the genus Haideotriton was found to be a synonym of
A single hemidactyliine (Palaeoplethodon) is known from Miocene fossil remains preserved in Dominican amber, marking the only record of salamanders in the Caribbean.[24]
Conservation status
| Status | Number of Species |
|---|---|
| Least Concern | 94 |
| Near Threatened | 39 |
| Vulnerable | 60 |
| Endangered | 88 |
| Critically Endangered | 68 |
| Extinct | 1 |
| Data Deficient | 40 |
References
- ^ . Retrieved 4 March 2024.
- ^ a b c "Plethodontidae". AmphibiaWeb. University of California, Berkeley. 2024. Retrieved 4 March 2024.
- ^ ISBN 0-12-178560-2.
- .
- ^ Bell's False Brook Salamander - World Land Trust
- ^ Hairston, N.G., Sr. 1987. Community ecology and salamander guilds. Cambridge University Press. Cambridge.
- ^ "Salamanders a more abundant food source in forest ecosystems than previously thought". ScienceDaily. 18 November 2014.
- ^ OCLC 427511083.
- PMID 30034038.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - .
- . Retrieved July 5, 2022.
- S2CID 53185123.
- ISBN 0253347327.
- ^ Doring, M. "Salamanders". gbif.org. Retrieved 26 April 2024.
- ^ PMID 4821655.
- .
- S2CID 209407983.
- ^ PMID 4821656.
- ^ PMID 35977024.
- ^ S2CID 87787255.
- PMID 26385618.
- .
- hdl:2246/5781.
- ^ Poinar Jr., G.; Wake, David B. (2015). "Palaeoplethodon hispaniolae gen. n., sp. n. (Amphibia: Caudata), a fossil salamander from the Caribbean" (PDF). Palaeodiversity. 8: 21–29.
- ^ IUCN RedList, (2020). Plethodontidae. Retrieved from https://www.iucnredlist.org/search?taxonomies=101246&searchType=species

