Solanine
| |

| |
| Names | |
|---|---|
| IUPAC name
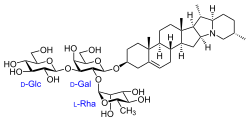
Solanid-5-en-3β-yl α-L-rhamnopyranosyl-(1→2)-[β-D-glucopyranosyl-(1→3)]-β-D-galactopyranoside
| |
| Systematic IUPAC name
(2S,3R,4R,5R,6S)-2-{[(2R,3R,4S,5S,6R)-5-Hydroxy-6-(hydroxymethyl)-2-{[(2S,4aR,4bS,6aS,6bR,7S,7aR,10S,12aS,13aS,13bS)-4a,6a,7,10-tetramethyl-2,3,4,4a,4b,5,6,6a,6b,7,7a,8,9,10,11,12a,13,13a,13b,14-icosahydro-1H-naphtho[2′,1′:4,5]indeno[1,2-b]indolizin-2-yl]oxy}-4-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-3-yl]oxy}-6-methyloxane-3,4,5-triol | |
| Other names
α-Solanine; Solanin; Solatunine
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.039.875 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C45H73NO15 | |
| Molar mass | 868.06 |
| Appearance | white crystalline solid |
| Melting point | 271 to 273 °C (520 to 523 °F; 544 to 546 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Solanine is a glycoalkaloid poison found in species of the nightshade family within the genus Solanum, such as the potato (Solanum tuberosum), the tomato (Solanum lycopersicum), and the eggplant (Solanum melongena). It can occur naturally in any part of the plant, including the leaves, fruit, and tubers. Solanine has pesticidal properties, and it is one of the plant's natural defenses. Solanine was first isolated in 1820 from the berries of the European black nightshade (Solanum nigrum), after which it was named.[1] It belongs to the chemical family of saponins.
Solanine poisoning
Symptoms
Solanine poisoning is primarily displayed by gastrointestinal and neurological disorders. Symptoms include
Ingestion of solanine in moderate amounts can cause death. One study suggests that doses of 2 to 5 mg/kg of body weight can cause toxic symptoms, and doses of 3 to 6 mg/kg of body weight can be fatal.[5]
Symptoms usually occur 8 to 12 hours after ingestion, but may occur as rapidly as 10 minutes after eating high-solanine foods.[citation needed]
Correlation with birth defects
Some studies show a correlation between the consumption of potatoes suffering from late blight (which increases solanine and other glycoalkaloid levels) and the incidence of spina bifida in humans.[citation needed] However, other studies have shown no correlation between potato consumption and the incidence of birth defects.[6]
Livestock poisoning
Livestock can also be susceptible to glycoalkaloids. High concentrations of solanine are necessary to cause death to mammals. The gastrointestinal tract cannot efficiently absorb solanine, which helps decrease its strength to the mammal body.[7] Livestock can hydrolyze solanine and excrete its contents to diminish its presence in the body.[7]
Mechanism of action
There are several proposed mechanisms of how solanine causes toxicity in humans, but the true mechanism of action is not well understood.
Biosynthesis

Solanine is a glycoalkaloid poison created by various plants in the genus Solanum, such as the potato plant. When the plant's stem, tubers, or leaves are exposed to sunlight, it stimulates the biosynthesis of solanine and other glycoalkaloids as a defense mechanism so it is not eaten.[12] It is therefore considered to be a natural pesticide.[citation needed]
Though the structures of the intermediates in this biosynthetic pathway are shown, many of the specific
Plants like the potato and tomato constantly synthesize low levels of glycoalkaloids like solanine. However, under stress, such as the presence of a
Safety
Suggested limits on consumption of solanine
Calculations have shown that 2 to 5 mg/kg of body weight is the likely toxic dose of glycoalkaloids like solanine in humans, with 3 to 6 mg/kg constituting the fatal dose.[20] Other studies have shown that symptoms of toxicity were observed with consumption of even 1 mg/kg.[11]
Proper storage of potatoes
Various storage conditions can have an impact on the level of solanine in potatoes. Glycoalkaloid levels increase when potatoes are exposed to light because light increases synthesis of glycoalkaloids like solanine.[18] Potatoes should therefore be stored in a dark place to avoid increased solanine synthesis. Potatoes that have turned green due to increased chlorophyll and photosynthesis are indicative of increased light exposure and are therefore associated with high levels of solanine.[20] Synthesis of solanine is also stimulated by mechanical injury because glycoalkaloids are synthesized at cut surfaces of potatoes.[18] Storage of potatoes for extended periods of time has also been associated with increased solanine content.[21] A study found that the solanine levels in Kurfi Jyoti and Kurfi Giriraj potatoes increase solanine levels by 0.232 mg/g and 0.252 mg/g respectively after being poorly stored in a heap.[22]
Effects of cooking on solanine levels
Most home processing methods like boiling, cooking, and frying potatoes have been shown to have minimal effects on solanine levels. For example,
The majority (30–80%) of the solanine in potatoes is found in the outer layer of the potato.
Recorded human poisonings
Though fatalities from solanine poisoning are rare, there have been several notable cases of human solanine poisonings. Between 1865 and 1983, there were around 2000 documented human cases of solanine poisoning, with most recovering fully and 30 deaths.[26] Because the symptoms are similar to those of food poisoning, it is possible that there are many undiagnosed cases of solanine toxicity.[27]
In 1899, 56 German soldiers fell ill due to solanine poisoning after consuming cooked potatoes containing 0.24 mg of solanine per gram of potato.
In Scotland in 1918, there were 61 cases of solanine poisoning after consumption of potatoes containing 0.41 mg of solanine per gram of potato, resulting in the death of a five-year old.[29]
A case report from 1925 reported that 7 family members who ate green potatoes fell ill from solanine poisoning two days later, resulting in the deaths of the 45-year-old mother and 16-year-old daughter. The other family members recovered fully.[19] In another case report from 1959, four members of a British family exhibited symptoms of solanine poisoning after eating jacket potatoes containing 0.5 mg of solanine per gram of potato.[citation needed]
There was a mass solanine poisoning incident in 1979 in the U.K., when 78 adolescent boys at a boarding school exhibited symptoms after eating potatoes that had been stored improperly over the summer.[30] Seventeen of them ended up hospitalized, but they all recovered. The potatoes were determined to have between 0.25 and 0.3 mg of solanine per gram of potato.[citation needed]
Another mass poisoning was reported in Canada in 1984, after 61 schoolchildren and teachers showed symptoms of solanine toxicity after consuming baked potatoes with 0.5 mg of solanine per gram of potato.[31]
In potatoes

Potatoes naturally produce solanine and chaconine, a related glycoalkaloid, as a defense mechanism against insects, disease, and herbivores. Potato leaves, stems, and shoots are naturally high in glycoalkaloids.[citation needed]
When potato tubers are exposed to light, they turn green and increase glycoalkaloid production. This is a natural defense to help prevent the uncovered tuber from being eaten. The green colour is from chlorophyll, and is itself harmless. However, it is an indication that increased level of solanine and chaconine may be present. In potato tubers, 30–80% of the solanine develops in and close to the skin, and some potato varieties have high levels of solanine.[citation needed]
Some potato diseases, such as late blight, can dramatically increase the levels of glycoalkaloids present in potatoes. Tubers damaged in harvesting and/or transport also produce increased levels of glycoalkaloids; this is believed to be a natural reaction of the plant in response to disease and damage.[citation needed]
Also, the tuber glycoalkaloids (such as solanine) can be affected by some chemical fertilization. For example, different studies have reported that glycoalkaloids content increases by increasing the concentration of nitrogen fertilizer.[32][33]
Green colouring under the skin strongly suggests solanine build-up in potatoes, although each process can occur without the other. A bitter taste in a potato is another – potentially more reliable – indicator of toxicity. Because of the bitter taste and appearance of such potatoes, solanine poisoning is rare outside conditions of food shortage. The symptoms are mainly vomiting and diarrhea, and the condition may be misdiagnosed as gastroenteritis. Most potato poisoning victims recover fully, although fatalities are known, especially when victims are undernourished or do not receive suitable treatment.[34]
The United States National Institutes of Health's information on solanine strongly advises against eating potatoes that are green below the skin.[3]
In other plants
Fatalities are also known from solanine poisoning from other plants in the nightshade family, such as the berries of Solanum dulcamara (woody nightshade).[35]
- In tomatoes
Some, such as the California Poison Control Center, have claimed that tomatoes and tomato leaves contain solanine. However, Mendel Friedman of the United States Department of Agriculture contradicts this claim, stating that tomatine, a relatively benign alkaloid, is the tomato alkaloid while solanine is found in potatoes. Food science writer Harold McGee has found scant evidence for tomato toxicity in the medical and veterinary literature.[36]
In popular culture
Dorothy L. Sayers's short story "The Leopard Lady", in the 1939 collection In the Teeth of the Evidence, features a child poisoned by potato berries injected with solanine to increase their toxicity.[citation needed]
See also
- Lenape (potato)
- Solanidine
References
- ^ Desfosses (1820). "Extrait d'une lettre de M. Desfosses, pharmacien, à Besançon, à M. Robiquet" [Extract of a letter from Mr. Desfosses, pharmacist in Besançon, to Mr. Robiquet]. Journal de Pharmacie. 2nd series (in French). 6: 374–376.
- ^ "Solanine poisoning – how does it happen?". 7 February 2014. Retrieved 24 September 2018.
- ^ a b MedlinePlus Encyclopedia: Potato plant poisoning – green tubers and sprouts
- ^ K. Annabelle Smith (21 October 2013). "Horrific Tales of Potatoes That Caused Mass Sickness and Even Death". Smithsonian Magazine.
- ^ Executive Summary of Chaconine & Solanine Archived 15 August 2006 at the Wayback Machine
- ^ "Solanine and Chaconine". Retrieved 31 May 2009.
- ^ PMID 6338654.
- PMID 10335373.
- PMID 16733852.
- ^ PMID 17090106.
- ^ .
- ^ PMID 22180624.
- PMID 23473422.
- ISSN 0370-663X.
- PMID 26938561.
- .
- .
- ^ ISBN 978-1-4612-7983-9.
- ^ PMID 7018841.
- ^ ISSN 0015-6647.
- PMID 30913609.
- ^ Haseena, R., Ganapathy, S., Pandiarajan, T., & Amirtham, D. (2019). Effect of storage methods on solanine content of potato tubers. Journal of Pharmacognosy and Phytochemistry, 8(6), 1677–1679.
- PMID 8655092.
- ^ Tice R (February 1998). Review of toxicological literature (PDF) (PhD Thesis).
- ^ PMID 6996922.
- ISBN 978-0-8493-6990-2.
- PMID 12720378.
- ISBN 9241660325.
- .
- PMID 504549.
- PMID 7737605.
- S2CID 98187389.
- .
- PMID 526812.
- PMID 18881287.
- ^ McGee H (29 July 2009). "Accused, Yes, but Probably Not a Killer". The New York Times. Retrieved 23 May 2010.
External links
- a-Chaconine and a-Solanine, Review of Toxicological Literature
- MedlinePlus Encyclopedia: 002875 – "Green tubers and sprouts"
