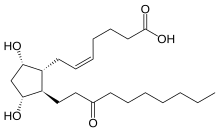
Unoprostone
 | |
| Clinical data | |
|---|---|
| Trade names | Rescula |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Topical (eye drops) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 14 min |
| Excretion | Renal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Unoprostone (
isopropyl ester, unoprostone isopropyl, was marketed under the trade name Rescula for the management of open-angle glaucoma and ocular hypertension.[1][2]
It was approved by the Food and Drug Administration in 2000.[3]
In 2009, Sucampo Pharmaceuticals acquired the rights to the drug in the U.S. and Canada.[4]
In 2015, the drug was discontinued in the U.S.[citation needed]
References
- ^ Micromedex Detailed Consumer Information
- PMID 24648719.
- ^ "Drug Approval Package". Food and Drug Administration.
- ^ "Sucampo Pharmaceuticals, Inc. Acquires Rights to Rescula for U.S. and Canada" (Press release). Business Wire. April 24, 2009.
