Acetylcholine
This article needs additional citations for verification. (August 2019) |
tubocurarine, atropine | |
| Precursor | choline, acetyl-CoA |
|---|---|
| Biosynthesis | choline acetyltransferase |
| Metabolism | acetylcholinesterase |
| Identifiers | |
| |
ECHA InfoCard | 100.000.118 |
| Chemical and physical data | |
| Formula | C7H16NO2 |
| Molar mass | 146.210 g·mol−1 |
Acetylcholine (ACh) is an organic compound that functions in the brain and body of many types of animals (including humans) as a neurotransmitter.[1] Its name is derived from its chemical structure: it is an ester of acetic acid and choline.[2] Parts in the body that use or are affected by acetylcholine are referred to as cholinergic.
Acetylcholine is the neurotransmitter used at the neuromuscular junction—in other words, it is the chemical that motor neurons of the nervous system release in order to activate muscles. This property means that drugs that affect cholinergic systems can have very dangerous effects ranging from paralysis to convulsions. Acetylcholine is also a neurotransmitter in the autonomic nervous system, both as an internal transmitter for the sympathetic nervous system and as the final product released by the parasympathetic nervous system.[1] Acetylcholine is the primary neurotransmitter of the parasympathetic nervous system.[2][3]
In the brain, acetylcholine functions as a
Acetylcholine has also been found in cells of non-neural origins as well as microbes. Recently, enzymes related to its synthesis, degradation and cellular uptake have been traced back to early origins of unicellular eukaryotes.[5] The protist pathogen Acanthamoeba spp. have shown evidence of the presence of ACh, which provides growth and proliferative signals via a membrane located M1-muscarinic receptor homolog.[6]
Partly because of its muscle-activating function, but also because of its functions in the autonomic nervous system and brain, many important drugs exert their effects by altering cholinergic transmission. Numerous venoms and toxins produced by plants, animals, and bacteria, as well as chemical
Chemistry
Acetylcholine is a choline molecule that has been acetylated at the oxygen atom. Because of the charged ammonium group, acetylcholine does not penetrate lipid membranes. Because of this, when the molecule is introduced externally, it remains in the extracellular space and at present it is considered that the molecule does not pass through the blood–brain barrier.
Biochemistry
Acetylcholine is synthesized in certain neurons by the enzyme choline acetyltransferase from the compounds choline and acetyl-CoA. Cholinergic neurons are capable of producing ACh. An example of a central cholinergic area is the nucleus basalis of Meynert in the basal forebrain.[8][9] The enzyme
Functions

Acetylcholine functions in both the central nervous system (CNS) and the peripheral nervous system (PNS). In the CNS, cholinergic projections from the basal forebrain to the cerebral cortex and hippocampus support the cognitive functions of those target areas. In the PNS, acetylcholine activates muscles and is a major neurotransmitter in the autonomic nervous system.[10][2]
Cellular effects

Like many other biologically active substances, acetylcholine exerts its effects by binding to and activating receptors located on the surface of cells. There are two main classes of acetylcholine receptor, nicotinic and muscarinic. They are named for chemicals that can selectively activate each type of receptor without activating the other: muscarine is a compound found in the mushroom Amanita muscaria; nicotine is found in tobacco.
Nicotinic acetylcholine receptors are ligand-gated ion channels permeable to sodium, potassium, and calcium ions. In other words, they are ion channels embedded in cell membranes, capable of switching from a closed to an open state when acetylcholine binds to them; in the open state they allow ions to pass through. Nicotinic receptors come in two main types, known as muscle-type and neuronal-type. The muscle-type can be selectively blocked by curare, the neuronal-type by hexamethonium. The main location of muscle-type receptors is on muscle cells, as described in more detail below. Neuronal-type receptors are located in autonomic ganglia (both sympathetic and parasympathetic), and in the central nervous system.
Neuromuscular junction
Acetylcholine is the substance the nervous system uses to activate
When a motor neuron generates an action potential, it travels rapidly along the nerve until it reaches the neuromuscular junction, where it initiates an electrochemical process that causes acetylcholine to be released into the space between the presynaptic terminal and the muscle fiber. The acetylcholine molecules then bind to nicotinic ion-channel receptors on the muscle cell membrane, causing the ion channels to open. Sodium ions then flow into the muscle cell, initiating a sequence of steps that finally produce muscle contraction.
Factors that decrease release of acetylcholine (and thereby affecting P-type calcium channels):[11]
- Antibiotics (clindamycin, polymyxin)
- Magnesium: antagonizes P-type calcium channels
- Hypocalcemia
- Anticonvulsants
- Diuretics (furosemide)
- Eaton-Lambert syndrome: inhibits P-type calcium channels
- Myasthenia gravis
- Botulinum toxin: inhibits SNARE proteins
Calcium channel blockers (nifedipine, diltiazem) do not affect P-channels. These drugs affect L-type calcium channels.
Autonomic nervous system
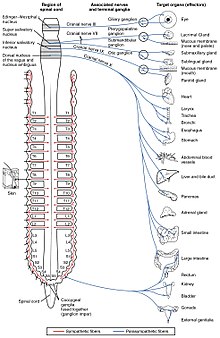
The autonomic nervous system controls a wide range of involuntary and unconscious body functions. Its main branches are the sympathetic nervous system and parasympathetic nervous system. Broadly speaking, the function of the sympathetic nervous system is to mobilize the body for action; the phrase often invoked to describe it is fight-or-flight. The function of the parasympathetic nervous system is to put the body in a state conducive to rest, regeneration, digestion, and reproduction; the phrase often invoked to describe it is "rest and digest" or "feed and breed". Both of these aforementioned systems use acetylcholine, but in different ways.
At a schematic level, the sympathetic and parasympathetic nervous systems are both organized in essentially the same way: preganglionic neurons in the central nervous system send projections to neurons located in autonomic ganglia, which send output projections to virtually every tissue of the body. In both branches the internal connections, the projections from the central nervous system to the autonomic ganglia, use acetylcholine as a neurotransmitter to innervate (or excite) ganglia neurons. In the parasympathetic nervous system the output connections, the projections from ganglion neurons to tissues that do not belong to the nervous system, also release acetylcholine but act on muscarinic receptors. In the sympathetic nervous system the output connections mainly release
Direct vascular effects
Acetylcholine in the
Central nervous system
In the central nervous system, ACh has a variety of effects on plasticity, arousal and reward. ACh has an important role in the enhancement of alertness when we wake up,[13] in sustaining attention [14] and in learning and memory.[15]
Damage to the cholinergic (acetylcholine-producing) system in the brain has been shown to be associated with the memory deficits associated with Alzheimer's disease.[16] ACh has also been shown to promote REM sleep.[17]
In the brainstem acetylcholine originates from the
- The pontomesencephalotegmental complex acts mainly on
- M1 receptors in the neocortex.
- Medial M1 receptors in the hippocampus and parts of the cerebral cortex.
In addition, ACh acts as an important internal transmitter in the
Memory
Acetylcholine has been implicated in learning and memory in several ways. The anticholinergic drug scopolamine impairs acquisition of new information in humans[22] and animals.[15] In animals, disruption of the supply of acetylcholine to the neocortex impairs the learning of simple discrimination tasks, comparable to the acquisition of factual information[23] and disruption of the supply of acetylcholine to the hippocampus and adjacent cortical areas produces forgetfulness, comparable to anterograde amnesia in humans.[24]
Diseases and disorders
Myasthenia gravis
The disease myasthenia gravis, characterized by muscle weakness and fatigue, occurs when the body inappropriately produces antibodies against acetylcholine nicotinic receptors, and thus inhibits proper acetylcholine signal transmission. Over time, the motor end plate is destroyed. Drugs that competitively inhibit acetylcholinesterase (e.g., neostigmine, physostigmine, or primarily pyridostigmine) are effective in treating the symptoms of this disorder. They allow endogenously released acetylcholine more time to interact with its respective receptor before being inactivated by acetylcholinesterase in the synaptic cleft (the space between nerve and muscle).
Pharmacology
Blocking, hindering or mimicking the action of acetylcholine has many uses in medicine. Drugs acting on the acetylcholine system are either agonists to the receptors, stimulating the system, or antagonists, inhibiting it. Acetylcholine receptor agonists and antagonists can either have an effect directly on the receptors or exert their effects indirectly, e.g., by affecting the enzyme acetylcholinesterase, which degrades the receptor ligand. Agonists increase the level of receptor activation; antagonists reduce it.
Acetylcholine itself does not have therapeutic value as a drug for intravenous administration because of its multi-faceted action (non-selective) and rapid inactivation by cholinesterase. However, it is used in the form of eye drops to cause constriction of the pupil during cataract surgery, which facilitates quick post-operational recovery.
Nicotinic receptors
Nicotine binds to and activates nicotinic acetylcholine receptors, mimicking the effect of acetylcholine at these receptors. ACh opens a Na+ channel upon binding so that Na+ flows into the cell. This causes a depolarization, and results in an excitatory post-synaptic potential. Thus, ACh is excitatory on skeletal muscle; the electrical response is fast and short-lived. Curares are arrow poisons, which act at nicotinic receptors and have been used to develop clinically useful therapies.
Muscarinic receptors
Muscarinic receptors form G protein-coupled receptor complexes in the cell membranes of neurons and other cells. Atropine is a non-selective competitive antagonist with Acetylcholine at muscarinic receptors.
Cholinesterase inhibitors
Many ACh receptor agonists work indirectly by inhibiting the enzyme acetylcholinesterase. The resulting accumulation of acetylcholine causes continuous stimulation of the muscles, glands, and central nervous system, which can result in fatal convulsions if the dose is high.
They are examples of
Synthesis inhibitors
Organic mercurial compounds, such as methylmercury, have a high affinity for sulfhydryl groups, which causes dysfunction of the enzyme choline acetyltransferase. This inhibition may lead to acetylcholine deficiency, and can have consequences on motor function.
Release inhibitors
Comparative biology and evolution
Acetylcholine is used by organisms in all domains of life for a variety of purposes. It is believed that choline, a precursor to acetylcholine, was used by single celled organisms billions of years ago[citation needed] for synthesizing cell membrane phospholipids.[25] Following the evolution of choline transporters, the abundance of intracellular choline paved the way for choline to become incorporated into other synthetic pathways, including acetylcholine production. Acetylcholine is used by bacteria, fungi, and a variety of other animals. Many of the uses of acetylcholine rely on its action on ion channels via GPCRs like membrane proteins.
The two major types of acetylcholine receptors, muscarinic and nicotinic receptors, have convergently evolved to be responsive to acetylcholine. This means that rather than having evolved from a common homolog, these receptors evolved from separate receptor families. It is estimated that the
History
In 1867, Adolf von Baeyer resolved the structures of choline and acetylcholine and synthesized them both, referring to the latter as "acetylneurin" in the study.[27][28] Choline is a precursor for acetylcholine. This is why Frederick Walker Mott and William Dobinson Halliburton noted in 1899 that choline injections decreased the blood pressure of animals.[29][28] Acetylcholine was first noted to be biologically active in 1906, when Reid Hunt (1870–1948) and René de M. Taveau found that it decreased blood pressure in exceptionally tiny doses.[30][28][31]
In 1914, Arthur J. Ewins was the first to extract acetylcholine from nature. He identified it as the blood pressure-decreasing contaminant from some
The concept of
In 1936, H. H. Dale and O. Loewi shared the Nobel Prize in Physiology or Medicine for their studies of acetylcholine and nerve impulses.[28]
See also
- Ann Silver
- Acetylcholinesterase
- Neuromuscular junction
- Nicotinic acetylcholine receptor
- Muscarinic acetylcholine receptor
Specific references
- ^ PMC 4027320.
- ^ PMID 32491757. Retrieved 2023-04-06.
- PMID 30969605.
- ISBN 978-0-12-374927-7.
- PMID 29058403.
- S2CID 5234123.
- PMID 26589572.
Delirium is only associated with the antagonism of post‐synaptic M1 receptors and to date other receptor subtypes have not been implicated
- S2CID 45579740.
- PMID 27199688.
- PMID 30969667. Retrieved 2023-04-06.
- ISBN 978-0-443-06959-8.
- S2CID 293055.
- PMID 16183137.
- PMID 10808142.
- ^ S2CID 7110504.
- PMID 10071091.
- S2CID 25323695.
- ^ S2CID 39665815.
- ^ S2CID 4721282.
- S2CID 21908514.
- PMID 15233923.
- PMID 4793334.
- S2CID 29182517.
- PMID 12050084.
- ^ S2CID 31059344.
- S2CID 18062185.
- .
- ^ PMID 25907239.
- PMID 20758460.
- ^ Hunt R, Taveau M (1906). "On the physiological action of certain choline derivatives and new methods for detecting choline". BMJ. 2: 1788–1791.
- PMID 7047939.
- ^ Dale HH (1914). "The action of certain esters and ethers of choline, and their relation to muscarine". J Pharmacol Exp Ther. 6 (2): 147–190.
- S2CID 34861770.
- ^ PMID 23183298.
- S2CID 43748121.
- PMID 16596840.
General bibliography
- Brenner GM, Stevens CW (2006). Pharmacology (2nd ed.). Philadelphia PA: W. B. Saunders. ISBN 1-4160-2984-2.
- Canadian Pharmacists Association (2000). Compendium of Pharmaceuticals and Specialties (25th ed.). Toronto ON: Webcom. ISBN 0-919115-76-4.
- Carlson NR (2001). Physiology of Behavior (7th ed.). Needham Heights MA: Allyn and Bacon. ISBN 0-205-30840-6.
- Gershon MD (1998). The Second Brain. New York NY: HarperCollins. ISBN 0-06-018252-0.
- Siegal A, Sapru HN (2006). "Ch. 15". Essential Neuroscience (Revised 1st ed.). Philadelphia: Lippincott, Williams & Wilkins. pp. 255–267.
- Yu AJ,
