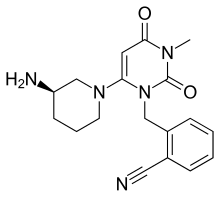
Alogliptin
 | |
| Clinical data | |
|---|---|
| Trade names | Nesina, Vipidia Kazano, Vipidomet (with metformin) Oseni, Incresync (with pioglitazone) |
| Other names | SYR-322 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a613026 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% |
| Protein binding | 20% |
| Metabolism | Limited, liver (CYP2D6- and 3A4-mediated) |
| Elimination half-life | 12–21 hours |
| Excretion | Kidney (major)[1] and fecal (minor) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Alogliptin, sold under the brand names Nesina and Vipidia,
In April 2016, the U.S. Food and Drug Administration (FDA) added a warning about increased risk of heart failure.[5] It was developed by Syrrx, a company which was acquired by Takeda Pharmaceutical Company in 2005.[6] In 2020, it was the 295th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[7][8]
Medical uses
Alogliptin is a dipeptidyl peptidase-4 inhibitor (DDP-4) that decreases blood sugar levels similar to other DPP-4 inhibitors.[9]
Side effects
Adverse events include
A 2014 letter to the editor claimed alogliptin is not associated with increased risk of cardiovascular events.
Market access

In December 2007, Takeda submitted a
In 2013, the FDA approved the drug in three formulations: as a stand-alone with the brand-name Nesina,[13] combined with metformin using the name Kazano,[18] and when combined with pioglitazone as Oseni.[19]
References
- ^ a b c d "www.aace.com" (PDF). Archived from the original (PDF) on 2018-11-01.
- ^ a b "Takeda Submits New Drug Application for Alogliptin (SYR-322) in the U.S." (Press release). Takeda Pharmaceutical Company. January 3, 2008. Retrieved March 11, 2021.
- ^ a b "Vipidia" (PDF). European Medicines Agency. Archived from the original (PDF) on 1 November 2018. Retrieved 31 March 2024.
- PMID 17441705.
- ^ a b "FDA Drug Safety Communication: FDA adds warnings about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin". U.S. Food and Drug Administration (FDA). Retrieved 16 March 2018.
- ^ "The San Diego Union-Tribune - San Diego, California & National News".
- ^ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ^ "Alogliptin - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- PMID 25914541.
- S2CID 24082863.
- S2CID 45948727.
- S2CID 1092260.
- ^ a b "Highlights of Prescribing Information: Nesina" (PDF). US Food and Drug Administration. Retrieved 31 March 2024.
- ^ "DPP-4 Inhibitors for Type 2 Diabetes: Drug Safety Communication - May Cause Severe Joint Pain". U.S. Food and Drug Administration (FDA). 2015-08-28. Retrieved 1 September 2015.
- PMID 24482824.
- ^ a b c d Grogan K (April 26, 2012), "FDA wants yet more data on Takeda diabetes drug alogliptin", PharmaTimes, PharmaTimes, PharmaTimes online, retrieved April 26, 2012
- ^ Genetic Engineering & Biotechnology News. June 4, 2009.
- ^ "Highlights of Prescribing Information: Kazano" (PDF). US Food and Drug Administration. Retrieved 31 March 2024.
- ^ "Highlights of Prescribing Information: Oseni" (PDF). US Food and Drug Administration. Retrieved 31 March 2024.
External links
 Media related to Alogliptin at Wikimedia Commons
Media related to Alogliptin at Wikimedia Commons- "Alogliptin". Drug Information Portal. U.S. National Library of Medicine.
