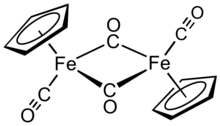
Cyclopentadienyliron dicarbonyl dimer
| |
| |
| |
| Names | |
|---|---|
| IUPAC name
Di-μ-carbonyldicarbonylbis(η5-cyclopenta-2,4-dien-1-yl)diiron
| |
| Other names
Bis(cyclopentadienyl)tetracarbonyl-diiron,
Di(cyclopentadienyl)tetracarbonyl-diiron, Bis(dicarbonylcyclopentadienyliron) | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.032.057 |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C14H10Fe2O4 | |
| Molar mass | 353.925 g/mol |
| Appearance | Dark purple crystals |
| Density | 1.77 g/cm3, solid |
| Melting point | 194 °C (381 °F; 467 K) |
| Boiling point | decomposition |
| insoluble | |
| Solubility in other solvents | benzene, THF, chlorocarbons |
| Structure | |
| distorted octahedral | |
| 3.1 D (benzene solution) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
CO source |
| GHS labelling: | |
  
| |
| Danger | |
| H228, H302, H330, H331 | |
| Related compounds | |
Related compounds
|
Fe(C5H5)2 Fe(CO)5 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclopentadienyliron dicarbonyl dimer is an
Structure
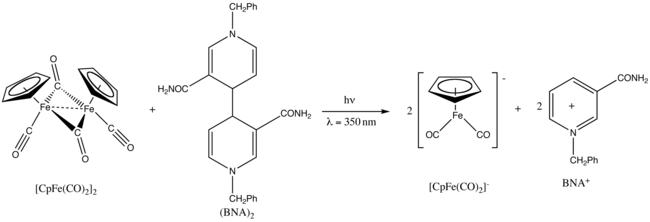
In solution, Cp2Fe2(CO)4 can be considered a dimeric half-sandwich complex. It exists in three isomeric forms: cis, trans, and an unbridged, open form. These isomeric forms are distinguished by the position of the ligands. The cis and trans isomers differ in the relative position of C5H5 (Cp) ligands. The cis and trans isomers have the formulation [(η5-C5H5)Fe(CO)(μ-CO)]2, that is, two CO ligands are terminal whereas the other two CO ligands bridge between the iron atoms. The cis and trans isomers interconvert via the open isomer, which has no bridging ligands between iron atoms. Instead, it is formulated as (η5-C5H5)(OC)2Fe−Fe(CO)2(η5-C5H5) — the metals are held together by an iron–iron bond. At equilibrium, the cis and trans isomers are predominant.
In addition, the terminal and bridging carbonyls are known to undergo exchange: the trans isomer can undergo bridging–terminal CO ligand exchange through the open isomer, or through a twisting motion without going through the open form. In contrast, the bridging and terminal CO ligands of the cis isomer can only exchange via the open isomer.[2]
In solution, the cis, trans, and open isomers interconvert rapidly at room temperature, making the molecular structure
The solid-state molecular structure of both cis and trans isomers have been analyzed by
Synthesis
Cp2Fe2(CO)4 was first prepared in 1955 at Harvard by Geoffrey Wilkinson using the same method employed today: the reaction of iron pentacarbonyl and dicyclopentadiene.[7][8]
- 2 Fe(CO)5 + C10H12 → (η5-C5H5)2Fe2(CO)4 + 6 CO + H2
In this preparation, dicyclopentadiene cracks to give cyclopentadiene, which reacts with Fe(CO)5 with loss of CO. Thereafter, the pathways for the photochemical and thermal routes differ subtly but both entail formation of a hydride intermediate.[5] The method is used in the teaching laboratory.[3]
Reactions
Although of no major commercial value, Fp2 is a workhorse in organometallic chemistry because it is inexpensive and FpX derivatives are rugged (X = halide, organyl).
"Fp−" (FpNa and FpK)
Reductive cleavage of [CpFe(CO)2]2 (formally an iron(I) complex) produces alkali metal derivatives formally derived from the cyclopentadienyliron dicarbonyl anion, [CpFe(CO)2]− or called Fp− (formally iron(0)), which are assumed to exist as a
- [CpFe(CO)2]2 + 2 Na → 2 CpFe(CO)2Na
- [CpFe(CO)2]2 + 2 KBH(C2H5)3 → 2 CpFe(CO)2K + H2 + 2 B(C2H5)3
Treatment of NaFp with an alkyl halide (RX, X = Br, I) produces FeR(η5-C5H5)(CO)2
- CpFe(CO)2K + CH3I → CpFe(CO)2CH3 + KI
Fp2 can also be cleaved with alkali metals
FpX (X = Cl, Br, I)
Halogens oxidatively cleave [CpFe(CO)2]2 to give the Fe(II) species FpX (X = Cl, Br, I):
- [CpFe(CO)2]2 + X2 → 2 CpFe(CO)2X
One example is cyclopentadienyliron dicarbonyl iodide.
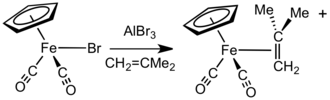
Fp(η2-alkene)+, Fp(η2-alkyne)+ and other "Fp+"
In the presence of halide anion acceptors such as
BF−
4, with the alkene or alkyne.[18] [FpL]+
BF−
4 complexes can also be prepared by treatment of FpMe with HBF4·Et2O in CH2Cl2 at −78 °C, followed by addition of L.[19]
Alkene–Fp complexes can also be prepared from Fp anion indirectly. Thus, hydride abstraction from Fp–alkyl compounds using triphenylmethyl hexafluorophosphate affords [Fp(α-alkene)]+ complexes.
- FpNa + RCH2CH2I → FpCH2CH2R + NaI
- FpCH2CH2R + Ph3CPF6 → [Fp(CH
2=CHR)+
]PF−
6 + Ph3CH
Reaction of NaFp with an
The alkene ligand in these cations is activated toward attack by
Fp(alkene)+ and Fp(alkyne)+ π-complexes are also quite acidic at the allylic and propargylic positions, respectively, and can be quantitatively deprotonated with amine bases like Et3N to give neutral Fp–allyl and Fp–allenyl σ-complexes (eqn 1).[16]
- (1) Fp(H
2C=CHCH
2CH
3)+
BF−
4 + Et3N → FpCH2CH=CHCH3 + Et
3NH+
BF−
4 - (2) FpCH2CH=CHCH3 + E+
BF−
4 → Fp(H
2C=CHCH(E)CH
3)+
BF−
4
Fp–allyl and Fp–allenyl react with cationic electrophiles E (such as Me3O+, carbocations, oxocarbenium ions) to generate allylic and propargylic functionalization products, respectively (eqn 2).[16] The related complex [Cp*Fe(CO)2(thf)]+[BF4]− has been shown to catalyze propargylic and allylic C−H functionalization by combining the deprotonation and electrophilic functionalization processes described above.[22]
η2-Allenyl complexes of Fp+ and substituted cyclopentadienyliron dicarbonyl cations have also been characterized, with X-ray crystallographic analysis showing substantial bending at the central allenic carbon (bond angle < 150°).[23][24]
Fp-based cyclopropanation reagents
Fp-based reagents have been developed for
- FpNa + ClCH2SCH3 → FpCH2SCH3 + NaCl
- FpCH2SCH3 + CH3I + NaBF4 → FpCH2S(CH3)2]BF4 + NaI
Use of [FpCH2S(CH3)2]BF4 does not require specialized conditions.
- Fp(CH
2S+
(CH
3)
2)BF−
4 + (Ph)2C=CH2 → 1,1-diphenylcyclopropane + …
Iron(III) chloride is added to destroy any byproduct.
Precursors to Fp=CH+
2, like FpCH2OMe which is converted to the iron carbene upon protonation, have also been used as cyclopropanation reagents.[26]
Photochemical reaction
Fp2 exhibits photochemistry.[27] For example, upon UV irradiation at 350 nm, it is reduced by benzylnicotinamide|1-benzyl-1,4-dihydronicotinamide dimer, also known as (BNA)2.[28]
References
- ISBN 0471936235.
- ^ ISSN 0300-9246.
- ^ ISBN 978-0-935702-48-4.
- ISSN 0020-1669.
- ^ ISBN 978-0-08-025269-8.
- PMID 23047247.
- ^ ISSN 0020-1693.
- .
- ^ Chang, T. C. T.; Rosenblum, M.; Simms, N. (1988). "Vinylation of Enolates with a Vinyl Cation Equivalent: trans-3-Methyl-2-Vinylcyclohexanone". Organic Syntheses. 66: 95; Collected Volumes, vol. 8, p. 479.
- .
- ISSN 0002-7863.
- .
- .
- .
- OCLC 840309324.
- ^ ISSN 0002-7863.
- ISBN 0471936235.
- OCLC 953660855.
- ^ Redlich, Mark D.; Mayer, Michael F.; Hossain, M. Mahmun (2003). "Iron Lewis Acid [(η5-C5H5)Fe(CO)2(THF)]+ Catalyzed Organic Reactions". Aldrichimica Acta. 36: 3–13.
- ISBN 978-0-12-548270-7.
- ISSN 0276-7333.
- S2CID 208611984.
- ISSN 0022-4936.
- PMID 34491051.
- ^ Mattson, M. N.; O'Connor, E. J.; Helquist, P. (1992). "Cyclopropanation Using an Iron-Containing Methylene Transfer Reagent: 1,1-Diphenylcyclopropane". Organic Syntheses. 70: 177; Collected Volumes, vol. 9, p. 372.
- ISBN 978-0-08-046518-0, retrieved 2019-12-11
- .
- PMID 11300821.



![Addition of carbanion to [Fp(alkene)]+.](http://upload.wikimedia.org/wikipedia/commons/thumb/7/7e/FpMalonateRxn.png/330px-FpMalonateRxn.png)