Chromium(II) acetate
| |

| |
| Names | |
|---|---|
| IUPAC name
Chromium(II) acetate hydrate
| |
| Other names
chromous acetate,
chromium diacetate, chromium(II) ethanoate | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.224.848 |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H16Cr2O10 | |
| Molar mass | 376.198 g·mol−1 |
| Appearance | brick-red solid |
| Density | 1.79 g/cm3 |
| Melting point | dehydrates |
| soluble in hot water, MeOH | |
| -5104.0·10−6 cm3/mol | |
| Structure | |
monoclinic
| |
| octahedral counting the Cr–Cr bond | |
| 0 D | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
could react exothermically in air |
| Related compounds | |
Related compounds
|
Rh2(OAc)4(H2O)2 Cu2(OAc)4(H2O)2, molybdenum(II) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chromium(II) acetate hydrate, also known as chromous acetate, is the
Cr2(OAc)4(H2O)2 is a reddish

Structure
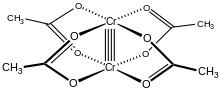
The Cr2(OAc)4(H2O)2 molecule contains two atoms of chromium, two ligated molecules of water, and four acetate bridging ligands. The coordination environment around each chromium atom consists of four oxygen atoms (one from each acetate ligand) in a square, one water molecule (in an axial position), and the other chromium atom (opposite the water molecule), giving each chromium centre an octahedral geometry. The chromium atoms are joined by a quadruple bond, and the molecule has D4h symmetry (ignoring the position of the hydrogen atoms). The same basic structure is adopted by Rh2(OAc)4(H2O)2 and Cu2(OAc)4(H2O)2, although these species do not have such short M–M contacts.[1]
The quadruple bond between the two chromium atoms arises from the overlap of four
History
Preparation
The preparation usually begins with reduction of an aqueous solution of a Cr(III) compound using
- 2 Cr3+ + Zn → 2 Cr2+ + Zn2+
- 2 Cr2+ + 4 OAc− + 2 H2O → Cr2(OAc)4(H2O)2
The synthesis of Cr2(OAc)4(H2O)2 has been traditionally used to test the synthetic skills and patience of inorganic laboratory students in universities because the accidental introduction of a small amount of air into the apparatus is readily indicated by the discoloration of the otherwise bright red product.[7] The anhydrous form of chromium(II) acetate, and also related chromium(II) carboxylates, can be prepared from chromocene:
- 4 RCO2H + 2 Cr(C5H5)2 → Cr2(O2CR)4 + 4 C5H6
This method provides anhydrous derivatives in a straightforward manner.[8]
Because it is so easily prepared, Cr2(OAc)4(H2O)2 is a starting material for other chromium(II) compounds. Also, many analogues have been prepared using other carboxylic acids in place of acetate and using different bases in place of the water.
Applications
Chromium(II) acetate has few practical applications. It has been used to dehalogenate organic compounds such as α-bromoketones and
Because the compound is a good reducing agent, it will reduce the O2 found in air and can be used as an oxygen scrubber.
See also
References
- ISBN 0-19-855649-7.
- .
- C. R. Acad. Sci.(in French). 19: 609–618.
- Ann. Chim. Phys.(in French). 12: 527–548.
- S2CID 4292992.
- )
- ISBN 9780138799328.
- .
- ISBN 9780471936237.
Further reading
- Rice, Steven F.; Wilson, Randall B.; .
