Cyclopentadienylchromium tricarbonyl dimer
| |
| Names | |
|---|---|
| IUPAC name
bis(tricarbonyl[η5-cyclopentadienyl]
| |
| Other names
cyclopentadienyl chromium
carbonyl dimer
Bis(tricarbonylcyclopentadienylchromium) | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C16H10Cr2O6 | |
| Molar mass | 402.242 g·mol−1 |
| Appearance | green solid |
| Density | 2.738 g/cm3 |
| insoluble | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
flammable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
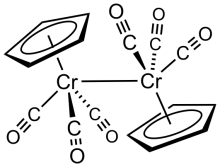
Cyclopentadienylchromium tricarbonyl dimer is the
organochromium compound with the formula Cp2Cr2(CO)6, where Cp is C5H5. A dark green crystalline
solid. It is the subject of research it exists in measureable equilibrium quantities with the monometallic radical CpCr(CO)3.
Structure and synthesis
The six CO ligands are terminal, and the Cr-Cr bond distance is 3.281 Å, 0.06 Å longer than the related dimolybdenum compound.[1] The compound is prepared by treatment of chromium hexacarbonyl with sodium cyclopentadienide followed by oxidation of the resulting NaCr(CO)3(C5H5).[2]
Related compounds
References
- .
- ISBN 9780470132593.
