Dicarboxylic acid
In
Dicarboxylic acids are used in the preparation of
Linear and cyclic saturated dicarboxylic acids
The general formula for acyclic dicarboxylic acid is HO
2C(CH
2)
nCO
2H.[1] The PubChem links gives access to more information on the compounds, including other names, ids, toxicity and safety.
Acids from the two-carbon oxalic acid to the ten-member sebacic acid may be remembered using the mnemonic 'Oh My Son, Go And Pray Softly And Silently', and also 'Oh my! Such great Apple Pie, sweet as sugar!'.
n Common name Systematic IUPAC name Structure pKa1 pKa2 PubChem 0 Oxalic acid ethanedioic acid 
1.27 4.27 971 1 Malonic acid propanedioic acid 
2.85 5.05 867 2 Succinic acid butanedioic acid 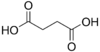
4.21 5.41 1110 3 Glutaric acid pentanedioic acid 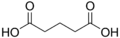
4.34 5.41 743 4 Adipic acid hexanedioic acid 
4.41 5.41 196 5 Pimelic acid heptanedioic acid 
4.50 5.43 385 6 Suberic acid octanedioic acid 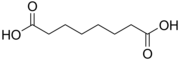
4.526 5.498 10457 6 1,4-Cyclohexanedicarboxylic acid 
14106 7 Azelaic acid nonanedioic acid 
4.550 5.498 2266 8 Sebacic acid decanedioic acid 
4.720 5.450 5192 9 undecanedioic acid 
15816 10 dodecanedioic acid 
12736 11 Brassylic acid tridecanedioic acid 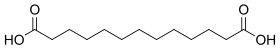
10458 14 Thapsic acid hexadecanedioic acid 
10459 19 Japanic acid heneicosanedioic acid 9543668 20 Phellogenic acid docosanedioic acid 
244872 28 Equisetolic acid triacontanedioic acid 5322010
Occurrence
- Adipic acid, despite its name (in Latin, adipis means fat), is not a normal constituent of natural lipids but is a product of leather tanning, urethane and also as an acidulantin foods.
- Pimelic acid (Greek pimelh, fat) was also first isolated from oxidized oil. Derivatives of pimelic acid are involved in the biosynthesis of lysine.
- Suberic acid was first produced by nitric acid oxidation of cork (Latin suber). This acid is also produced when castor oil is oxidised. Suberic acid is used in the manufacture of alkyd resins and in the synthesis of polyamides (nylonvariants).
- Azelaic acid's name stems from the action of nitric acid (azote, nitrogen, or azotic, nitric) oxidation of anaerobic micro-organisms present on acne-bearing skin. . Azelaic acid was identified as a molecule that accumulated at elevated levels in some parts of plants and was shown to be able to enhance the resistance of plants to infections.[2]
- Sebacic acid, named from sebum (tallow). Thenard isolated this compound from distillation products of beef tallow in 1802. It is produced industrially by alkali fission of castor oil.[3] Sebacic acid and its derivatives have a variety of industrial uses as plasticizers, lubricants, diffusion pump oils, cosmetics, candles, etc. It is also used in the synthesis of polyamide, as nylon, and of alkyd resins. An isomer, isosebacic acid, has several applications in the manufacture of vinyl resin plasticizers, extrusion plastics, adhesives, ester lubricants, polyesters, polyurethane resins and synthetic rubber.
- Brassylic acid can be produced from
- Dodecanedioic acid is used in the production of nylon (nylon-6,12), polyamides, coatings, adhesives, greases, polyesters, dyestuffs, detergents, flame retardants, and fragrances. It is now produced by fermentation of long-chain alkanes with a specific strain of Candida tropicalis.[5] Traumatic acid is its monounsaturated counterpart.
- Thapsic acid was isolated from the dried roots of the Mediterranean "deadly carrot", Thapsia garganica (Apiaceae).
Japan wax is a mixture containing triglycerides of C21, C22 and C23 dicarboxylic acids obtained from the sumac tree (Rhus sp.).
A large survey of the dicarboxylic acids present in Mediterranean nuts revealed unusual components.[6] A total of 26 minor acids (from 2 in pecan to 8% in peanut) were determined: 8 species derived from succinic acid, likely in relation with photosynthesis, and 18 species with a chain from 5 to 22 carbon atoms. Higher weight acids (>C20) are found in suberin present at vegetal surfaces (outer bark, root epidermis). C16 to C26 a, ω-dioic acids are considered as diagnostic for suberin. With C18:1 and C18:2, their content amount from 24 to 45% of whole suberin. They are present at low levels (< 5%) in plant cutin, except in Arabidopsis thaliana where their content can be higher than 50%.[7]
It was shown that
Dicarboxylic acids may be produced by ω-oxidation of fatty acids during their
Branched-chain dicarboxylic acids
Long-chain dicarboxylic acids containing
Some parent C29 to C32 diacids but with methyl groups on the carbons C-13 and C-16 have been isolated and characterized from the lipids of thermophilic anaerobic bacterium Thermoanaerobacter ethanolicus.[15] The most abundant diacid was the C30 a,ω-13,16-dimethyloctacosanedioic acid.
Biphytanic diacids are present in geological sediments and are considered as tracers of past anaerobic oxidation of methane.[16] Several forms without or with one or two pentacyclic rings have been detected in Cenozoic seep limestones. These lipids may be unrecognized metabolites from Archaea.

Crocetin is the core compound of crocins (crocetin glycosides) which are the main red pigments of the stigmas of saffron (Crocus sativus) and the fruits of gardenia (Gardenia jasminoides). Crocetin is a 20-carbon chain dicarboxylic acid which is a diterpenenoid and can be considered as a carotenoid. It was the first plant carotenoid to be recognized as early as 1818 while the history of saffron cultivation reaches back more than 3,000 years. The major active ingredient of saffron is the yellow pigment crocin 2 (three other derivatives with different glycosylations are known) containing a gentiobiose (disaccharide) group at each end of the molecule. A simple and specific HPLC-UV method has been developed to quantify the five major biologically active ingredients of saffron, namely the four crocins and crocetin.[17]
Unsaturated dicarboxylic acids
Type Common name IUPAC name Isomer Structural formula PubChem Monounsaturated Maleic acid (Z)-Butenedioic acid cis 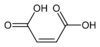
444266 Fumaric acid (E)-Butenedioic acid trans 
444972 Acetylenedicarboxylic acid But-2-ynedioic acid not applicable 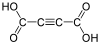
371 Glutaconic acid (Z)-Pent-2-enedioic acid cis 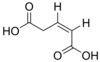
5370328 (E)-Pent-2-enedioic acid trans 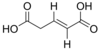
5280498 2-Decenedioic acid trans 
6442613 Traumatic acid Dodec-2-enedioic acid trans 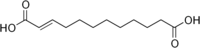
5283028 Diunsaturated Muconic acid (2E,4E)-Hexa-2,4-dienedioic acid trans,trans 
5356793 (2Z,4E)-Hexa-2,4-dienedioic acid cis,trans 
280518 (2Z,4Z)-Hexa-2,4-dienedioic acid cis,cis 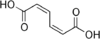
5280518 Glutinic acid
(Allene-1,3-dicarboxylic acid)(RS)-Penta-2,3-dienedioic acid HO2CCH=C=CHCO2H 5242834 Branched Citraconic acid (2Z)-2-Methylbut-2-enedioic acid cis 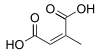
643798 Mesaconic acid (2E)-2-Methyl-2-butenedioic acid trans 
638129 Itaconic acid 2-Methylidenebutanedioic acid – 
811
Traumatic acid, was among the first biologically active molecules isolated from plant tissues. This dicarboxylic acid was shown to be a potent wound healing agent in plant that stimulates cell division near a wound site,[18] it derives from 18:2 or 18:3 fatty acid hydroperoxides after conversion into oxo- fatty acids.
trans,trans-Muconic acid is a metabolite of benzene in humans. The determination of its concentration in urine is therefore used as a biomarker of occupational or environmental exposure to benzene.[19][20]
Glutinic acid, a substituted
While polyunsaturated fatty acids are unusual in plant cuticles, a diunsaturated dicarboxylic acid has been reported as a component of the surface waxes or polyesters of some plant species. Thus, octadeca-c6,c9-diene-1,18-dioate, a derivative of
Alkylitaconates

PubChem 811
Several dicarboxylic acids having an alkyl side chain and an itaconate core have been isolated from
A series of alkyl- and alkenyl-itaconates, known as ceriporic acids (Pub Chem 52921868), were found in cultures of a selective lignin-degrading fungus (white rot fungus), Ceriporiopsis subvermispora.[24][25] The absolute configuration of ceriporic acids, their stereoselective biosynthetic pathway and the diversity of their metabolites have been discussed in detail.[26]
Substituted dicarboxylic acids
Common name IUPAC name Structural formula PubChem Tartronic acid 2-Hydroxypropanedioic acid 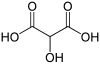
45 Mesoxalic acid Oxopropanedioic acid 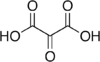
10132 Malic acid Hydroxybutanedioic acid 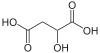
525 Tartaric acid 2,3-Dihydroxybutanedioic acid 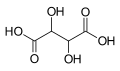
875 Oxaloacetic acid Oxobutanedioic acid 
970 Aspartic acid 2-Aminobutanedioic acid 
5960 dioxosuccinic acid dioxobutanedioic acid 
82062 α-hydroxyGlutaric acid2-hydroxypentanedioic acid 
43 Arabinaric acid 2,3,4-Trihydroxypentanedioic acid 109475 Acetonedicarboxylic acid 3-Oxopentanedioic acid 
68328 α-Ketoglutaric acid 2-Oxopentanedioic acid 
51 Glutamic acid 2-Aminopentanedioic acid 
611 Diaminopimelic acid (2R,6S)-2,6-Diaminoheptanedioic acid 
865 Saccharic acid (2S,3S,4S,5R)-2,3,4,5-Tetrahydroxyhexanedioic acid 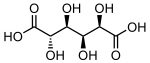
33037
Aromatic dicarboxylic acids
Common names IUPAC name Structure PubChem Phthalic acid
o-phthalic acidBenzene-1,2-dicarboxylic acid 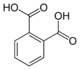
1017 Isophthalic acid
m-phthalic acidBenzene-1,3-dicarboxylic acid 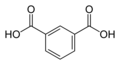
8496 Terephthalic acid
p-phthalic acidBenzene-1,4-dicarboxylic acid 
7489 Diphenic acid
Biphenyl-2,2′-dicarboxylic acid2-(2-Carboxyphenyl)benzoic acid 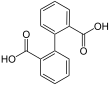
10210 2,6-Naphthalenedicarboxylic acid 2,6-Naphthalenedicarboxylic acid 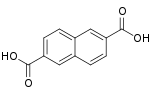
14357
Terephthalic acid is a
Properties
Dicarboxylic acids are crystalline solids. Solubility in water and melting point of the α,ω- compounds progress in a series as the carbon chains become longer with alternating between odd and even numbers of carbon atoms, so that for even numbers of carbon atoms the melting point is higher than for the next in the series with an odd number.[27] These compounds are weak dibasic acids with pKa tending towards values of ca. 4.5 and 5.5 as the separation between the two carboxylate groups increases. Thus, in an aqueous solution at pH about 7, typical of biological systems, the Henderson–Hasselbalch equation indicates they exist predominantly as dicarboxylate anions.
The dicarboxylic acids, especially the small and linear ones, can be used as crosslinking reagents.[28] Dicarboxylic acids where the carboxylic groups are separated by none or one carbon atom decompose when they are heated to give off carbon dioxide and leave behind a monocarboxylic acid.[27]
Blanc's Rule says that heating a barium salt of a dicarboxylic acid, or dehydrating it with acetic anhydride will yield a cyclic acid anhydride if the carbon atoms bearing acid groups are in position 1 and (4 or 5). So succinic acid will yield succinic anhydride. For acids with carboxylic groups at position 1 and 6 this dehydration causes loss of carbon dioxide and water to form a cyclic ketone, for example, adipic acid will form cyclopentanone.[27]
Derivatives
As for monofunctional carboxylic acids, derivatives of the same types exist. However, there is the added complication that either one or two of the carboxylic groups could be altered. If only one is changed then the derivative is termed "acid", and if both ends are altered it is called "normal". These derivatives include salts, chlorides, esters, amides, and anhydrides. In the case of anhydrides or amides, two of the carboxyl groups can come together to form a cyclic compound, for example succinimide.[29]
See also
References
- S2CID 206518245.
- S2CID 189786702.
- ISBN 978-3-642-13440-1. Retrieved 18 May 2021.
- ^ a b Kroha, Kyle (September 2004). "Industrial biotechnology provides opportunities for commercial production of new long-chain dibasic acids". Inform. 15: 568–571.
- .
- PMID 18440267.
- PMID 9098079.
- S2CID 34894138.
- .
- PMID 2831753.
- PMID 540040.
- S2CID 12709437.
- PMID 9098079.
- PMID 8077844.
- .
- PMID 10457433.
- S2CID 3712976.
- S2CID 6392962.
- PMID 8919771.
- PMID 22279375.
- PMID 15584957.
- PMID 10732974. – via ScienceDirect(Subscription may be required or content may be available in libraries.)
- .
- PMID 14623447.
- PMID 19477313.
- ^ a b c Schmidt, Julius (1955). Organic Chemistry. London: Oliver and Boyd. pp. 283–284.
- S2CID 221283821.
- ^ Bernthsen, A. (1922). Organic Chemistry. London: Blackie & Son. p. 242.
External links
- Lipidomics gateway Structure Database Dicarboxylic acids
- Dijkstra, Albert J. "Trivial names of fatty acids-Part 1". lipidlibrary.aocs.org. Retrieved 24 June 2019.































