Polyamide
A polyamide is a
Polyamides occur both naturally and artificially. Examples of naturally occurring polyamides are
Classification
Polymers of
According to the composition of their main chain, synthetic polyamides are classified as follows:
| Family | Main chain | Examples | Commercial products |
|---|---|---|---|
| Aliphatic polyamides | Aliphatic | PA 66 |
Solvay, Rilsan and Rilsamid from Arkema, Radipol from Radici Group
|
| Polyphthalamides | Semi-aromatic | PA 6T = hexamethylenediamine + terephthalic acid | Trogamid T from Solvay
|
| Aromatic polyamides, or aramids | Aromatic | Paraphenylenediamine + terephthalic acid |
Kevlar and Nomex from DuPont, Teijinconex, Twaron and Technora from Teijin, Kermel from Kermel. |
All polyamides are made by the formation of an amide function to link two molecules of monomer together. The monomers can be amides themselves (usually in the form of a cyclic lactam such as caprolactam), α,ω-amino acids or a stoichiometric mixture of a diamine and a diacid. Both these kinds of precursors give a homopolymer. Polyamides are easily copolymerized, and thus many mixtures of monomers are possible which can in turn lead to many copolymers. Additionally many nylon polymers are miscible with one another allowing the creation of blends.
Polymerization chemistry
Production of polymers requires the repeated joining of two groups to form an amide linkage. In this case this specifically involves
The condensation reaction is used to synthetically produce nylon polymers in industry. Nylons must specifically include a straight chain (aliphatic) monomer. The amide link is produced from an amine group (alternatively known as an amino group), and a carboxylic acid group. The hydroxyl from the carboxylic acid combines with a hydrogen from the amine, and gives rise to water, the elimination byproduct that is the namesake of the reaction.
As an example of condensation reactions, consider that in living organisms,

For fully aromatic polyamides or aramids e.g. Kevlar, the more reactive acyl chloride is used as a monomer. The polymerization reaction with the amine group eliminates hydrogen chloride. The acid chloride route can be used as a laboratory synthesis to avoid heating and obtain an almost instantaneous reaction.[3] The aromatic moiety itself does not participate in elimination reaction, but it does increase the rigidity and strength of the resulting material which leads to Kevlar's renowned strength.
In the diagram below, an aramid is made from two different monomers which continuously alternate to form the polymer chain. Aramids are aromatic polyamides:

Polyamides can also be synthesized from dinitriles using acid catalysis via an application of the Ritter reaction. This method is applicable for preparation of nylon 1,6 from adiponitrile, formaldehyde and water.[4] Additionally, polyamides can be synthesized from glycols and dinitriles using this method as well.[5]
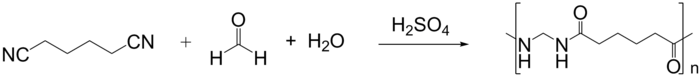
See also
References
- ^ Market Study Engineering Plastics, Ceresana, Sep 2013
- ^ "Making nylon: The "nylon rope trick"". Royal Society of Chemistry. Retrieved 19 April 2015.
- ISSN 0002-7863.
- S2CID 98232570.
Further reading
- Kohan, Melvin I. (1995). Nylon Plastics Handbook. Hanser/Gardner Publications. ISBN 9781569901892


