Discovery of the neutron

The discovery of the neutron and its properties was central to the extraordinary developments in
The essential nature of the atomic nucleus was established with the discovery of the neutron by James Chadwick in 1932[6] and the determination that it was a new elementary particle, distinct from the proton.[7][8]: 55
The uncharged neutron was immediately exploited as a new means to probe nuclear structure, leading to such discoveries as the creation of new radioactive elements by neutron irradiation (1934) and the fission of uranium atoms by neutrons (1938).[9] The discovery of fission led to the creation of both nuclear power and nuclear weapons by the end of World War II. Both the proton and the neutron were presumed to be elementary particles until the 1960s, when they were determined to be composite particles built from quarks.[10]
Discovery of radioactivity
At the start of the 20th century, the vigorous debate as to the existence of atoms had not yet been resolved. Philosophers such as Ernst Mach and Wilhelm Ostwald denied that atoms were real, viewing them as a convenient mathematical construct, while scientists such as Arnold Sommerfeld and Ludwig Boltzmann saw that physical theories required the existence of atoms.[9]: 13–14
The gold foil experiment and the discovery of the atomic nucleus
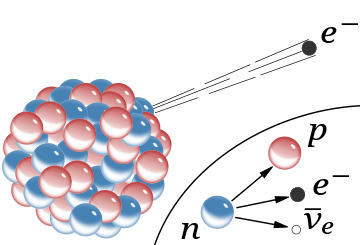
β−
radiation, the emission of a fast electron from the nucleus (the accompanying antineutrino is omitted). In the Rutherford model for the nucleus, a red sphere was a proton with positive charge, and a blue sphere was a proton tightly bound to an electron, with no net charge.
The inset shows beta decay of a free neutron as it is understood today; an electron and antineutrino are created in this process.
At the
The Rutherford model was very influential, motivating the Bohr model for electrons orbiting the nucleus in 1913[12] and eventually leading to quantum mechanics by the mid-1920s.
Discovery of isotopes
Concurrent with the work of Rutherford, Geiger, and Marsden, the
Building from work by
). Significantly, the one exception to this rule was hydrogen itself, which had a mass value of 1.008. The excess mass was small, but well outside the limits of experimental uncertainty.Since Einstein's
Atomic number and Moseley's law
Rutherford and others had noted the disparity between the mass of an atom, computed in atomic mass units, and the approximate charge required on the nucleus for the Rutherford model to work. The required charge of the atomic nucleus was usually about half its atomic mass.[21]: 82 Antonius van den Broek boldly hypothesized that the required charge, denoted by Z, was not half of the atomic weight for elements, but instead was exactly equal to the element's ordinal position in the periodic table.[1]: 228 At that time, the positions of the elements in the periodic table were not known to have any physical significance. If the elements were ordered based on increasing atomic mass, however, periodicity in chemical properties was exhibited. Exceptions to this periodicity were apparent, however, such as cobalt and nickel.[a][22]: 180
At the
Within a year it was noted that the equation for the relation, now called
Further research in atomic physics was interrupted by the outbreak of
Rutherford atom

In 1920 Rutherford gave a
In that lecture, Rutherford conjectured the existence of new particles. The alpha particle was known to be very stable, and it was assumed to retain its identity within the nucleus. The alpha particle was presumed to consist of four protons and two closely bound electrons to give it +2 charge and mass 4. In a 1919 paper,[32] Rutherford had reported the apparent discovery of a new doubly charged particle of mass 3, denoted the X++, interpreted to consist of three protons and a closely bound electron. This result suggested to Rutherford the likely existence of two new particles: one of two protons with a closely bound electron, and another of one proton and a closely bound electron. The X++ particle was later determined to have mass 4 and to be just a low-energy alpha particle.[8]: 25 Nevertheless, Rutherford had conjectured the existence of the deuteron, a +1 charge particle of mass 2, and the neutron, a neutral particle of mass 1.[31]: 396 The former is the nucleus of deuterium, discovered in 1931 by Harold Urey.[33] The mass of the hypothetical neutral particle would be little different from that of the proton. Rutherford determined that such a zero-charge particle would be difficult to detect by available techniques.[31]: 396
About the time of Rutherford's lecture, other publications appeared with similar suggestions of a proton–electron composite in the nucleus, and in 1921
Rutherford and Chadwick immediately began an experimental program at the Cavendish Laboratory in Cambridge to search for the neutron.[8]: 27 [1]: 398 The experiments continued throughout the 1920s without success.[6]
Rutherford's conjecture and the hypothetical "neutron" were not widely accepted. In his 1931 monograph on the Constitution of Atomic Nuclei and Radioactivity,
Problems of the nuclear electrons hypothesis
Throughout the 1920s, physicists assumed that the atomic nucleus was composed of protons and "nuclear electrons".[8]: 29–32 [42] Under this hypothesis, the nitrogen-14 (14N) nucleus would be composed of 14 protons and 7 electrons, so that it would have a net charge of +7 elementary charge units and a mass of 14 atomic mass units. This nucleus would also be orbited by another 7 electrons, termed "external electrons" by Rutherford,[31]: 375 to complete the 14N atom. However problems with the hypothesis soon became apparent.
Ralph Kronig pointed out in 1926 that the observed hyperfine structure of atomic spectra was inconsistent with the proton–electron hypothesis. This structure is caused by the influence of the nucleus on the dynamics of orbiting electrons. The magnetic moments of supposed "nuclear electrons" should produce hyperfine spectral line splittings similar to the Zeeman effect, but no such effects were observed.[43]: 199 It seemed that the magnetic moment of the electron vanished when it was within the nucleus.[1]: 299
While on a visit to Utrecht University in 1928, Kronig learned of a surprising aspect of the rotational spectrum of N2+. The precision measurement made by Leonard Ornstein, the director of Utrecht's Physical Laboratory, showed that the spin of nitrogen nucleus must be equal to one. However, if the nitrogen-14 (14N) nucleus was composed of 14 protons and 7 electrons, an odd number of spin-1/2 particles, then the resultant nuclear spin should be half-integer. Kronig therefore suggested that perhaps "protons and electrons do not retain their identity to the extent they do outside the nucleus".[1]: 299–301 [44]: 117
Observations of the rotational energy levels of diatomic molecules using Raman spectroscopy by Franco Rasetti in 1929 were inconsistent with the statistics expected from the proton–electron hypothesis. Rasetti obtained band spectra for H2 and N2 molecules. While the lines for both diatomic molecules showed alternation in intensity between light and dark, the pattern of alternation for H2 is opposite to that of the N2. After carefully analyzing these experimental results, German physicists Walter Heitler and Gerhard Herzberg showed that the hydrogen nuclei obey Fermi statistics and the nitrogen nuclei obey Bose statistics. However, a then unpublished result of Eugene Wigner showed that a composite system with an odd number of spin-1/2 particles must obey Fermi statistics; a system with an even number of spin-1/2 particle obeys Bose statistics. If the nitrogen nucleus had 21 particles, it should obey Fermi statistics, contrary to fact. Thus, Heitler and Herzberg concluded: "the electron in the nucleus ... loses its ability to determine the statistics of the nucleus."[44]: 117–118
The Klein paradox,[45] discovered by Oskar Klein in 1928, presented further quantum mechanical objections to the notion of an electron confined within a nucleus. Derived from the Dirac equation, this clear and precise paradox suggested that an electron approaching a high potential barrier has a high probability of passing through the barrier[40] by a pair creation process. Apparently, an electron could not be confined within a nucleus by any potential well. The meaning of this paradox was intensely debated at the time.[43]: 199–200
By about 1930 it was generally recognized that it was difficult to reconcile the proton–electron model for nuclei with the
In 1927, Charles Ellis and W. Wooster at the Cavendish Laboratory measured the energies of β-decay electrons. They found that the distribution of energies from any particular radioactive nuclei was broad and continuous, a result that contrasted notably with the distinct energy values observed in alpha and gamma decay. Further, the continuous energy distribution seemed to indicate that energy was not conserved by this "nuclear electrons" process. Indeed, in 1929 Bohr proposed to modify the law of energy conservation to account for the continuous energy distribution. The proposal earned the support of Werner Heisenberg. Such considerations were apparently reasonable, inasmuch as the laws of quantum mechanics had so recently overturned the laws of classical mechanics.
While all these considerations did not "prove" an electron could not exist in the nucleus, they were confusing and challenging for physicists to interpret. Many theories were invented to explain how the above arguments could be wrong.[48]: 4–5 In his 1931 monograph, Gamow summarized all these contradictions, marking the statements regarding electrons in the nucleus with warning symbols.[42]: 23
Discovery of the neutron
In 1930,

Two years later
On hearing of the Paris results, Rutherford and James Chadwick at the Cavendish Laboratory also did not believe the gamma ray hypothesis since it failed to conserve energy.[54] Assisted by Norman Feather,[55] Chadwick quickly performed a series of experiments showing that the gamma ray hypothesis was untenable. The previous year, Chadwick, J.E.R. Constable, and E.C. Pollard had already conducted experiments on disintegrating light elements using alpha radiation from polonium.[56] They had also developed more accurate and efficient methods for detecting, counting, and recording the ejected protons. Chadwick repeated the creation of the radiation using beryllium to absorb the alpha particles: 9Be + 4He (α) → 12C + 1n. Following the Paris experiment, he aimed the radiation at paraffin wax, a hydrocarbon high in hydrogen content, hence offering a target dense with protons. As in the Paris experiment, the radiation energetically scattered some of the protons. Chadwick measured the range of these protons, and also measured how the new radiation impacted the atoms of various gases.[57] Measurements of the recoil energy showed that the mass of the radiation particles must be similar to the mass of the proton: the new radiation could not consist of gamma rays. Uncharged particles with about the same mass as the proton matched the properties Rutherford described in 1920 and which had later been called neutrons.[58][6][59][60] Chadwick won the Nobel Prize in Physics in 1935 for this discovery.[61]
The year 1932 was later referred to as the "annus mirabilis" for nuclear physics in the Cavendish Laboratory,[57] with discoveries of the neutron, artificial nuclear disintegration by the Cockcroft–Walton particle accelerator, and the positron.
Proton–neutron model of the nucleus
Given the problems of the proton–electron model,[42][62] it was quickly accepted that the atomic nucleus is composed of protons and neutrons, although the precise nature of the neutron was initially unclear. Within months after the discovery of the neutron, Werner Heisenberg[63][64][65][60] and Dmitri Ivanenko[66] had proposed proton–neutron models for the nucleus.[67] Heisenberg's landmark papers approached the description of protons and neutrons in the nucleus through quantum mechanics. While Heisenberg's theory for protons and neutrons in the nucleus was a "major step toward understanding the nucleus as a quantum mechanical system,"[68] he still assumed the presence of nuclear electrons. In particular, Heisenberg assumed the neutron was a proton–electron composite, for which there is no quantum mechanical explanation. Heisenberg had no explanation for how lightweight electrons could be bound within the nucleus. Heisenberg introduced the first theory of nuclear exchange forces that bind the nucleons. He considered protons and neutrons to be different quantum states of the same particle, i.e., nucleons distinguished by the value of their nuclear isospin quantum numbers.
The proton–neutron model explained the puzzle of dinitrogen. When 14N was proposed to consist of 3 pairs each of protons and neutrons, with an additional unpaired neutron and proton each contributing a spin of 1⁄2 ħ in the same direction for a total spin of 1 ħ, the model became viable.[69][70][71] Soon, neutrons were used to naturally explain spin differences in many different nuclides in the same way.
If the proton–neutron model for the nucleus resolved many issues, it highlighted the problem of explaining the origins of beta radiation. No existing theory could account for how electrons, or positrons,[72] could emanate from the nucleus.[73] In 1934, Enrico Fermi published his classic paper describing the process of beta decay, in which the neutron decays to a proton by creating an electron and a (as yet undiscovered) neutrino.[74] The paper employed the analogy that photons, or electromagnetic radiation, were similarly created and destroyed in atomic processes. Ivanenko had suggested a similar analogy in 1932.[69][75] Fermi's theory requires the neutron to be a spin-1⁄2 particle. The theory preserved the principle of conservation of energy, which had been thrown into question by the continuous energy distribution of beta particles. The basic theory for beta decay proposed by Fermi was the first to show how particles could be created and destroyed. It established a general, basic theory for the interaction of particles by weak or strong forces.[74] While this influential paper has stood the test of time, the ideas within it were so new that when it was first submitted to the journal Nature in 1933 it was rejected as being too speculative.[68]
The nature of the neutron

The question of whether the neutron was a composite particle of a proton and an electron persisted for a few years after its discovery.[76][77] In 1932 Harrie Massey explored a model for a composite neutron to account for its great penetrating power through matter and its electrical neutrality,[78] for example. The issue was a legacy of the prevailing view from the 1920s that the only elementary particles were the proton and electron.
The nature of the neutron was a primary topic of discussion at the 7th
The difficulty of making the measurement is illustrated by the wide-ranging values for the mass of the neutron obtained from 1932 to 1934. The accepted value today is 1.00866
In 1935 Chadwick and his doctoral student
In this reaction, the resulting proton and neutron have about equal kinetic energy, since their masses are about equal. The kinetic energy of the resulting proton could be measured (0.24 MeV), and therefore the deuteron's binding energy could be determined (2.6 MeV − 2(0.24 MeV) = 2.1 MeV, or 0.0023
md + b.e. = mp + mn
where md,p,n refer to the deuteron, proton, or neutron mass, and "b.e." is the binding energy. The masses of the deuteron and proton were known; Chadwick and Goldhaber used values 2.0142 u and 1.0081 u, respectively. They found that the neutron's mass was slightly greater than the mass of the proton 1.0084
Neutron physics in the 1930s
Soon after the discovery of the neutron, indirect evidence suggested the neutron had an unexpected non-zero value for its magnetic moment. Attempts to measure the neutron's magnetic moment originated with the discovery by

The discovery of the neutron immediately gave scientists a new tool for probing the properties of atomic nuclei. Alpha particles had been used over the previous decades in scattering experiments, but such particles, which are helium nuclei, have +2 charge. This charge makes it difficult for alpha particles to overcome the Coulomb repulsive force and interact directly with the nuclei of atoms. Since neutrons have no electric charge, they do not have to overcome this force to interact with nuclei. Almost coincident with their discovery, neutrons were used by Norman Feather, Chadwick's colleague and protege, in scattering experiments with nitrogen.[90] Feather was able to show that neutrons interacting with nitrogen nuclei scattered to protons or induced nitrogen to disintegrate to form boron with the emission of an alpha particle. Feather was therefore the first to show that neutrons produce nuclear disintegrations.
In Rome, Enrico Fermi and his team bombarded heavier elements with neutrons and found the products to be radioactive. By 1934 they had used neutrons to induce radioactivity in 22 different elements, many of these elements of high atomic number. Noticing that other experiments with neutrons at his laboratory seemed to work better on a wooden table than a marble table, Fermi suspected that the protons of the wood were slowing the neutrons and so increasing the chance for the neutron to interact with nuclei. Fermi therefore passed neutrons through paraffin wax to slow them and found that the radioactivity of some bombarded elements increased by a factor of tens to hundreds.[91] The cross section for interaction with nuclei is much larger for slow neutrons than for fast neutrons. In 1938 Fermi received the Nobel Prize in Physics "for his demonstrations of the existence of new radioactive elements produced by neutron irradiation, and for his related discovery of nuclear reactions brought about by slow neutrons".[92][93]


In
Hahn and his collaborators had detected the splitting of uranium nuclei, made unstable by neutron absorption, into lighter elements. Meitner and Frisch also showed that the fission of each uranium atom would release about 200 MeV of energy. The discovery of fission electrified the global community of atomic physicists and the public.
After 1939
The discovery of nuclear fission at the end of 1938 marked a shift in the centers of nuclear research from
The discoveries of the neutron and positron in 1932 were the start of the discoveries of many new particles. Muons were discovered in 1936. Pions and kaons were discovered in 1947, while lambda particles were discovered in 1950. Throughout the 1950s and 1960s, a large number of particles called hadrons were discovered. A classification scheme for organizing all these particles, proposed independently by Murray Gell-Mann[103] and George Zweig[104][105] in 1964, became known as the quark model. By this model, particles such as the proton and neutron were not elementary, but composed of various configurations of a small number of other truly elementary particles called partons or quarks. The quark model received experimental verification beginning in the late 1960s and finally provided an explanation for the neutron's anomalous magnetic moment.[106][10]
Videos
- Ernest Rutherford summarizes the state of nuclear physics in 1935. (7 min., Nobelprize.org)
- Hans Bethe discusses Chadwick and Goldhaber's work on deuteron disintegration. (2 min., Web of Stories)
Explanatory notes
- ^ The atomic number and atomic mass for cobalt are respectively 27 and 58.97, for nickel they are respectively 28 and 58.68.
- ^ In a nucleus of radius r in the order of 5×10−13cm, the uncertainty principle would require an electron to have a momentum p of the order of h/r. Such a momentum implies that the electron has a (relativistic) kinetic energy of about 40MeV.[46]: 89
References
- ^ ISBN 978-0198519973.
- ^ . Retrieved 15 November 2017.
- ISBN 978-0-521-52878-8.
- ^ doi:10.1039/a804629h.
- ^ ISBN 0486482383
- ^ .
- ^ .
- ^ ISBN 978-90-277-1584-5.
- ^ ISBN 978-0-8176-3732-3.
- ^ ISBN 978-0-201-05757-7
- ISBN 9780199766413
- .
- ^ "The Nobel Prize in Chemistry 1921 – Frederick Soddy Biographical". Nobelprize.org. Retrieved 5 September 2019.
- ISBN 978-0124058972
- ^ Others had also suggested the possibility of isotopes; for example:
- Strömholm, D. and Svedberg, T. (1909) "Untersuchungen über die Chemie der radioactiven Grundstoffe II." (Investigations into the chemistry of the radioactive elements, part 2), Zeitschrift für anorganischen Chemie, 63: 197–206; see especially page 206.
- Cameron, Alexander Thomas (1910). Radiochemistry. London, England: J. M. Dent & Sons, p. 141. (Cameron also anticipated the displacement law.)
- ^ "The Nobel Prize in Chemistry 1921: Frederick Soddy – Biographical". Nobelprize.org. Retrieved 16 March 2014.
- ^ Aston, Francis William. Mass spectra and isotopes. London: Edward Arnold, 1942.
- ^ "Atomic Weights and the International Committee — A Historical Review". 26 January 2004.
- ^ "The Nobel Prize in Chemistry 1922: Francis W. Aston – Biographical". Nobelprize.org. Retrieved 18 November 2017.
- S2CID 36422819.
- ^ a b
Heilbron, J. L. (1974). H.G.J. Moseley: The Life and Letters of an English Physicist, 1887–1915. University of California Press. ISBN 0520023757.
- ^ ISBN 0-19-852049-2.
- S2CID 144765815
- .
- ASIN B009I5KZGM
- ISBN 9780486318585
- ISBN 978-0691095523.
- ^ "Oral History Interview: Niels Bohr, Session I". American Institute of Physics, Niels Bohr Library and Archives. 31 October 1962. Retrieved 25 October 2017.
- ^ Reynosa, Peter (7 January 2016). "An Ode to Henry Moseley". Huffington Post. Retrieved 16 November 2017.
- APS News. 16 (5). 2007. Retrieved 16 November 2017.
- ^ .
- ^ Rutherford, E. (1919). "Collision of α particles with light atoms". Philosophical Magazine. 37: 571.
- .
- ^ .
- .
- .
- .
- ISBN 978-3-540-13609-5.
- ISBN 978-0852747612
- ^ a b c d George Gamow "Constitution of Atomic Nuclei and Radioactivity". (The International Series of Monographs on Physics.) Pp.viii + 114.(Oxford: Clarendon Press; London: Oxford University Press, 1931.)
- New Scientist and Science Journal. 51 (3): 464–466. Retrieved 27 September 2017.
- ^ S2CID 121080564.
During the 1920s physicists came to accept the view that matter is built of only two kinds of elementary particles, electrons and protons.
- ^ ISBN 978-0674624160.
- ^ ISBN 9780192562906
- S2CID 121771000.
- ^
- .
- ISBN 978-0-471-80553-3.
- ^ "The Nobel Prize in Physics 1954". nobelprize.org. Retrieved 23 March 2023.
In 1930 Bothe, in collaboration with H. Becker, bombarded beryllium of mass 9 (and also boron and lithium) with alpha rays derived from polonium, and obtained a new form of radiation ...
- S2CID 122888356.
- S2CID 121188471.
- Comptes Rendus des Séances de l'Académie des Sciences(in French). 194: 273.
- ^ Zichichi, A., Ettore Majorana: genius and mystery, CERN Courier, 25 July 2006, Access date: 16 November 2017.
- ^ ISBN 9780198539926.
- ^ "Oral History Interview: Norman Feather, Session I". American Institute of Physics, Niels Bohr Library and Archives. 25 February 1971. Retrieved 16 November 2017.
- .
- ^ a b "Atop the Physics Wave: Rutherford Back in Cambridge, 1919–1937". Rutherford's Nuclear World. American Institute of Physics. 2011–2014. Archived from the original on 21 October 2014. Retrieved 19 August 2014.
- S2CID 4076465.
- ^ .
- ^ a b Ley, Willy (October 1966). "The Delayed Discovery". For Your Information. Galaxy Science Fiction. pp. 116–127.
- ^ "The Nobel Prize in Physics 1935: James Chadwick – Biographical". Nobel Foundation. Retrieved 18 November 2017.
- ^ Friedlander, G.; Kennedy, J.W.; Miller, J.M. (1964) Nuclear and Radiochemistry (2nd edition), Wiley, pp. 22–23 and 38–39
- S2CID 186218053.
- S2CID 186221789.
- S2CID 126422047.
- ^ Iwanenko, D.D., The neutron hypothesis, Nature. 129 (1932) 798.
- ISBN 0521568919
- ^ ISBN 978-0750303736.
heisenberg proton neutron model.
- ^ Comptes Rendus de l'Académie des Sciences de Paris. 195: 439–441.
- .
- Bibcode:2009BMNAS2009....1W. Archived from the original(PDF) on 2014-05-31. Retrieved 2015-03-21.
- S2CID 4001646.
- .
- ^ .
- ^ Iwanenko, D. (1932). "Neutronen und kernelektronen". Physikalische Zeitschrift der Sowjetunion. 1: 820–822.
- .
- ^ .
- .
- ISBN 978-0520089068.
neutron.
- ISBN 9780520064263.
- S2CID 4137231.
- S2CID 120793548.
- S2CID 186232193.
- .
- .
- .
- ^ ISBN 9780674004351.
- .
- Doklady Akademii Nauk SSSR. 8: 455. Retrieved 30 January 2015.
- .
- ^ E. Fermi; E. Amaldi; B. Pontecorvo; F. Rasetti; E. Segrè (October 1934). "Azione di sostanze idrogenate sulla radioattività provocata da neutroni" [Influence of hydrogenous substances on the radioactivity produced by neutrons]. La Ricerca Scientifica (in Italian). II (7–8). Retrieved 16 August 2021.
- ^ "The Nobel Prize in Physics 1938: Enrico Fermi – Biographical". Nobelprize.org. Retrieved 18 November 2017.
- OCLC 39508200.
- .
- S2CID 4113262.
- S2CID 33512939.
- ^ "The Nobel Prize in Chemistry 1944: Otto Hahn – Biographical". Nobelprize.org. Retrieved 18 November 2017.
- ISBN 978-0-387-95089-1.
- ISBN 978-0743264747.
- ^ "About Enrico Fermi". The University of Chicago Library: Digital Activities and Collections. Guide to the Enrico Fermi Collection, Special Collections Research Center, University of Chicago Library. Archived from the original on 26 November 2012. Retrieved 24 November 2017.
- ^ "Fermi at Columbia: The Manhattan Project and the First Nuclear Pile". Columbia University Department of Physics. Archived from the original on 29 October 2017. Retrieved 24 November 2017.
- ISBN 978-0-671-44133-3.
- ^ Gell-Mann, M. (1964). "A Schematic Model of Baryons and Mesons". .
- ^ Zweig, G. (1964). "An SU(3) Model for Strong Interaction Symmetry and its Breaking" (PDF). CERN Report No.8182/TH.401.
- ^ Zweig, G. (1964). "An SU(3) Model for Strong Interaction Symmetry and its Breaking: II" (PDF). CERN Report No.8419/TH.412.
- S2CID 123822660.
