Hookworm infection
| Hookworm infection | |
|---|---|
| Other names | Hookworm disease |
iron supplements[3] | |
| Frequency | 428 million (2015)[4] |
Hookworm infection is an infection by a type of
Two common hookworm infections in humans are
The disease can be prevented on an individual level by not walking barefoot in areas where the disease is common. At a population level, decreasing
Hookworms infected about 428 million people in 2015.
Signs and symptoms
No symptoms or signs are specific for hookworm infection, but they give rise to a combination of intestinal
Incubation period
The incubation period can vary between a few weeks to many months, and is largely dependent on the number of hookworm parasites an individual is infected with.[9]
Cause

Hookworm infections in humans include
]Other animals such as
Morphology
A. duodenale worms are grayish white or pinkish with the head slightly bent in relation to the rest of the body. This bend forms a definitive hook shape at the anterior end for which hookworms are named. They possess well-developed mouths with two pairs of teeth. While males measure approximately one centimeter by 0.5 millimeter, the females are often longer and stouter. Additionally, males can be distinguished from females based on the presence of a prominent posterior copulatory bursa.[11]
N. americanus is very similar in morphology to A. duodenale. N. americanus is generally smaller than A. duodenale with males usually 5 to 9 mm long and females about 1 cm long. Whereas A. duodenale possesses two pairs of teeth, N. americanus possesses a pair of cutting plates in the buccal capsule. Additionally, the hook shape is much more defined in Necator than in Ancylostoma.[11]
Life cycle
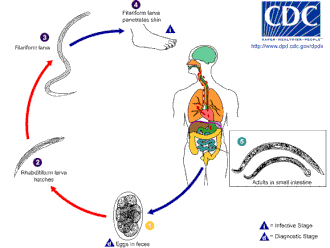
The hookworm thrives in warm soil where temperatures are over 18 °C (64 °F). They exist primarily in
Infection of the host is by the larvae, not the eggs. While A. duodenale can be ingested, the usual method of infection is through the skin; this is commonly caused by walking
Once in the host gut, Necator tends to cause a prolonged infection, generally 1 to 5 years (many worms die within a year or two of infecting), though some adult worms have been recorded to live for 15 years or more. Ancylostoma adults are short-lived, surviving on average for only about 6 months. However, the infection can be prolonged because dormant larvae can be "recruited" sequentially from tissue "stores" (see Pathology, above) over many years, to replace expired adult worms. This can give rise to seasonal fluctuations in infection prevalence and intensity (apart from normal seasonal variations in transmission).[citation needed]

They mate inside the host, females laying from 10,000 to 30,000 eggs per day and some 15 to 56 million eggs during their adult lifetimes, which pass out in feces. Because 5 to 7 weeks are needed for adult worms to mature, mate, and produce eggs, in the early stages of very heavy infection, acute symptoms might occur without any eggs being detected in the patient's feces. This can make diagnosis very difficult.[citation needed]
N. americanus and A. duodenale eggs can be found in warm, moist soil where they eventually hatch into first-stage larvae, or L1. L1, the feeding noninfective rhabditoform stage, will feed on soil microbes and eventually molt into second-stage larvae, L2, which is also in the rhabditoform stage. It will feed for about 7 days and then molt into the third-stage larvae, or L3. This is the filariform stage of the parasite, that is, the nonfeeding infective form of the larvae. The L3 larvae are extremely motile and seek higher ground to increase their chances of penetrating the skin of a human host. The L3 larvae can survive up to 2 weeks without finding a host. While N. americanus larvae only infect through penetration of skin, A. duodenale can infect both through penetration and orally. After the L3 larvae have successfully entered the host, they then travel through the subcutaneous venules and lymphatic vessels of the human host. Eventually, the L3 larvae enter the lungs through the pulmonary capillaries and break out into the alveoli. They then travel up the trachea to be coughed and swallowed by the host. After being swallowed, the L3 larvae are then found in the small intestine, where they molt into the L4, or adult worm stage. The entire process from skin penetration to adult development takes about 5–9 weeks. The female adult worms release eggs (N. americanus about 9,000–10,000 eggs/day and A. duodenale 25,000–30,000 eggs/day), which are passed in the feces of the human host. These eggs hatch in the environment within several days and the cycle starts anew.[12][14][15]
Pathophysiology
Hookworm infection is generally considered to be asymptomatic, but as Norman Stoll described in 1962, it is an extremely dangerous infection because its damage is "silent and insidious."
The disease was linked to nematode worms (Ankylostoma duodenalis) from one-third to half an inch long in the intestine chiefly through the labours of Theodor Bilharz and Griesinger in Egypt (1854).[18]
The symptoms can be linked to inflammation in the gut stimulated by feeding hookworms, such as nausea, abdominal pain and intermittent diarrhea, and to progressive anemia in prolonged disease:
Blood tests in early infection often show a rise in numbers of eosinophils, a type of white blood cell that is preferentially stimulated by worm infections in tissues (large numbers of eosinophils are also present in the local inflammatory response). Falling blood hemoglobin levels will be seen in cases of prolonged infection with anemia.[citation needed]
In contrast to most intestinal
Diagnosis

Diagnosis depends on finding characteristic worm eggs on microscopic examination of the stools, although this is not possible in early infection. Early signs of infection in most dogs include limbular limping and anal itching. The eggs are oval or elliptical, measuring 60 by 40 µm, colorless, not
Recent research has focused on the development of DNA-based tools for diagnosis of infection, specific identification of hookworm, and analysis of genetic variability within hookworm populations.[19] Because hookworm eggs are often indistinguishable from other parasitic eggs, PCR assays could serve as a molecular approach for accurate diagnosis of hookworm in the feces.[19][20]
Prevention
The infective larvae develop and survive in an environment of damp dirt, particularly sandy and loamy soil. They cannot survive in clay or muck. The main lines of precaution are those dictated by good hygiene behaviors:
- Do not defecate in the open, but rather in toilets.
- Do not use untreated human excreta or raw sewage as fertilizer in agriculture.
- Do not walk barefoot in known infected areas.
- Deworm pet dogs and cats. Canine and feline hookworms rarely develop to adulthood in humans. Ancylostoma caninum, the common dog hookworm, occasionally develops into an adult to cause eosinophilic enteritis in people, but their invasive larvae can cause an itchy rash called cutaneous larva migrans.

Children
Most of these public health concerns have focused on children who are infected with hookworm. This focus on children is largely due to the large body of evidence that has demonstrated strong associations between hookworm infection and impaired learning, increased absences from school, and decreased future economic productivity.[12] In 2001, the 54th World Health Assembly passed a resolution demanding member states to attain a minimum target of regular deworming of at least 75% of all at-risk school children by the year 2010.[21] A 2008 World Health Organization publication reported on these efforts to treat at-risk school children. Some of the interesting statistics were as follows: 1) only 9 out of 130 endemic countries were able to reach the 75% target goal; and 2) less than 77 million school-aged children (of the total 878 million at risk) were reached, which means that only 8.78% of at-risk children are being treated for hookworm infection.[22]
School-based mass deworming
School-based mass deworming programs have been the most popular strategy to address the issue of hookworm infection in children. School-based programs are extremely cost-effective as schools already have an available, extensive, and sustained infrastructure with a skilled workforce that has a close relationship with the community.[21] With little training from a local health system, teachers can easily administer the drugs which often cost less than US$0.50 per child per year.[23]
Recently, many people have begun to question if the school-based programs are necessarily the most effective approach. An important concern with school-based programs is that they often do not reach children who do not attend school, thus ignoring a large number of at-risk children. A 2008 study by Massa et al. continued the debate regarding school-based programs. They examined the effects of community-directed treatments versus school-based treatments in the Tanga Region of Tanzania. A major conclusion was that the mean infection intensity of hookworm was significantly lower in the villages employing the community-directed treatment approach than the school-based approach. The community-directed treatment model used in this specific study allowed villagers to take control of the child's treatment by having villagers select their own community drug distributors to administer the antihelminthic drugs. Additionally, villagers organized and implemented their own methods for distributing the drugs to all children.[24] The positive results associated with this new model highlight the need for large-scale community involvement in deworming campaigns.[citation needed]
Public health education
Many mass deworming programs also combine their efforts with a public health education. These health education programs often stress important preventative techniques such as: washing your hands before eating, and staying away from water/areas contaminated by human feces. These programs may also stress that shoes must be worn, however, these come with their own health risks and may not be effective.[25] Shoe wearing patterns in towns and villages across the globe are determined by cultural beliefs, and the levels of education within that society. The wearing of shoes will prevent the entry of hookworm infections from the surrounding soils into tender skin regions; such as areas between the toes.[26]
Sanitation
Historical examples, such as the hookworm campaigns in Mississippi and Florida from 1943 to 1947 have shown that the primary cause of hookworm infection is poor sanitation, which can be solved by building and maintaining toilets. But while these may seem like simple tasks, they raise important public health challenges. Most infected populations are from poverty-stricken areas with very poor sanitation. Thus, it is most likely that at-risk children do not have access to clean water to wash their hands and live in environments with no proper sanitation infrastructure. Health education, therefore, must address preventive measures in ways that are both feasible and sustainable in the context of resource-limited settings.[citation needed]
Integrated approaches
Evaluation of numerous public health interventions has generally shown that improvement in each individual component ordinarily attributed to poverty (for example, sanitation, health education and underlying nutrition status) often have minimal impact on transmission. For example, one study found that the introduction of latrines into a resource-limited community only reduced the prevalence of hookworm infection by four percent.[27] However, another study in Salvador, Brazil found that improved drainage and sewerage had a significant impact on the prevalence of hookworm infection but no impact at all on the intensity of hookworm infection.[28] This seems to suggest that environmental control alone has a limited but incomplete effect on the transmission of hookworms. It is imperative, therefore, that more research is performed to understand the efficacy and sustainability of integrated programs that combine numerous preventive methods including education, sanitation, and treatment.
Treatment
Anthelmintic drugs
The most common treatment for hookworm are
Hookworm infection can be treated with local cryotherapy when the hookworm is still in the skin.[30]
Albendazole is effective both in the intestinal stage and during the stage the parasite is still migrating under the skin.[30]
In case of anemia,
During the 1910s, common treatments for hookworm included
Reinfection and drug resistance
Other important issues related to the treatment of hookworm are reinfection and drug resistance. It has been shown that reinfection after treatment can be extremely high. Some studies even show that 80% of pretreatment hookworm infection rates can be seen in treated communities within 30–36 months.[14] While reinfection may occur, it is still recommended that regular treatments be conducted as it will minimize the occurrence of chronic outcomes. There are also increasing concerns about the issue of drug resistance. Drug resistance has appeared in front-line anthelmintics used for livestock nematodes. Generally human nematodes are less likely to develop resistance due to longer reproducing times, less frequent treatment, and more targeted treatment. Nonetheless, the global community must be careful to maintain the effectiveness of current anthelmintic as no new anthelmintic drugs are in the late-stage development.[14]
Epidemiology
It is estimated that between 576 and 740 million individuals are infected with hookworm.[33][14] Of these infected individuals, about 80 million are severely affected.[19] The major cause of hookworm infection is N. americanus which is found in the Americas, sub-Saharan Africa, and Asia.[12] A. duodenale is found in more scattered focal environments, namely Europe and the Mediterranean. Most infected individuals are concentrated in sub-Saharan Africa and East Asia/the Pacific Islands with each region having estimates of 198 million and 149 million infected individuals, respectively. Other affected regions include: South Asia (50 million), Latin America and the Caribbean (50 million), South Asia (59 million), Middle East/North Africa (10 million).[14] A majority of these infected individuals live in poverty-stricken areas with poor sanitation. Hookworm infection is most concentrated among the world's poorest who live on less than $2 a day.[12]
While hookworm infection may not directly lead to mortality, its effects on morbidity demand immediate attention. When considering
It has been estimated that as many as 22.1 million
Many of the numbers regarding the prevalence of hookworm infection are estimates as there is no international surveillance mechanism currently in place to determine prevalence and global distribution.[12] Some prevalence rates have been measured through survey data in endemic regions around the world. The following are some of the most recent findings on prevalence rates in regions endemic with hookworm.
- 43% infection rate of predominantly N. americanus although with some A. duodenale infection
- Both hookworm infection load and degree of anemia in the mild range
- 60% infection rate of predominantly N. americanus
- Important trends noted were that prevalence increased with age (plateau of about 41 years) and women had higher prevalence rates than men
- 52% of a total of 526 tested households infected
- Could not identify species, but previous studies in North Vietnam reported N. americanus in more than 95% of hookworm larvae
Minas Gerais, Brazil (Fleming et al. 2006)[39]
- 63% infection rate of predominantly N. americanus
KwaZulu-Natal, South Africa (Mabaso et al. 2004)[40]
- Inland areas had a prevalence rate of 9% of N. americanus
- Coastal plain areas had a prevalence rate of 63% of N. americanus
Lowndes County, Alabama, United States [41][42]
- 35% infection rate of predominantly N. americanus
There have also been technological developments that may facilitate more accurate mapping of hookworm prevalence. Some researchers have begun to use geographical information systems (GIS) and remote sensing (RS) to examine helminth ecology and epidemiology. Brooker et al. utilized this technology to create helminth distribution maps of sub-Saharan Africa. By relating satellite derived environmental data with prevalence data from school-based surveys, they were able to create detailed prevalence maps. The study focused on a wide range of helminths, but interesting conclusions about hookworm specifically were found. As compared to other helminths, hookworm is able to survive in much hotter conditions and was highly prevalent throughout the upper end of the thermal range.[43]
Improved molecular diagnostic tools are another technological advancement that could help improve existing prevalence statistics. Recent research has focused on the development of a DNA-based tool that can be used for diagnosis of infection, specific identification of hookworm, and analysis of genetic variability in hookworm populations. Again this can serve as a major tool for different public health measures against hookworm infection. Most research regarding diagnostic tools is now focused on the creation of a rapid and cost-effective assay for the specific diagnosis of hookworm infection. Many are hopeful that its development can be achieved within the next five years.[when?][19]
History
Discovery
The symptoms now attributed to hookworm appear in papyrus papers of ancient Egypt (c. 1500 BC), described as a derangement characterized by anemia. Avicenna, a Persian physician of the eleventh century, discovered the worm in several of his patients and related it to their disease. In later times, the condition was noticeably prevalent in the mining industry in England, France, Germany, Belgium, North Queensland, and elsewhere.[18]
Italian physician
A breakthrough came 25 years later following a
Eradication programmes
In 1899, American zoologist Charles Wardell Stiles identified progressive pernicious anemia seen in the southern United States as being caused by the hookworm A. duodenale. Testing in the 1900s revealed very heavy infestations in school-age children. In Puerto Rico, Dr. Bailey K. Ashford, a US Army physician, organized and conducted a parasite treatment campaign, which cured approximately 300,000 people (one-third of the Puerto Rican population) and reduced the death rate from this anemia by 90 percent during the years 1903–04.

On October 26, 1909, the Rockefeller Sanitary Commission for the Eradication of Hookworm Disease was organized as a result of a gift of US$1 million from John D. Rockefeller, Sr. The five-year program was a remarkable success and a great contribution to the United States' public health, instilling public education, medication, field work and modern government health departments in eleven southern states.[46] The hookworm exhibit was a prominent part of the 1910 Mississippi state fair.
The commission found that an average of 40% of school-aged children were infected with hookworm. Areas with higher levels of hookworm infection prior to the eradication program experienced greater increases in school enrollment, attendance, and literacy after the intervention. Econometric studies have shown that this effect cannot be explained by a variety of alternative factors, including differential trends across areas, changing crop prices, shifts in certain educational and health policies and the effect of malaria eradication.
The RF's hookworm campaign in Mexico showed how science and politics play a role in developing health policies. It brought together government officials, health officials, public health workers, Rockefeller officials and the community. This campaign was launched to eradicate hookworms in Mexico. Although the campaign did not focus on long-term treatments, it did set the terms of the relationship between Mexico and the Rockefeller Foundation. The scientific knowledge behind this campaign helped shape public health policies, improved public health and built a strong relationship between US and Mexico.[49]
In the 1920s, hookworm eradication reached the Caribbean and Latin America, where great mortality was reported among people in the West Indies towards the end of the 18th century, as well as through descriptions sent from Brazil and various other tropical and sub-tropical regions.[18]
Treatments
Treatment in the early 20th century relied on the use of
Research
Anemia in pregnancy
It is estimated that a third of all pregnant women in developing countries are infected with hookworm, 56% of all pregnant women in developing countries experience anemia, 20% of all maternal deaths are either directly or indirectly related to anemia. Numbers like this have led to an increased interest in the topic of hookworm-related anemia during pregnancy.[52] With the understanding that chronic hookworm infection can often lead to anemia, many people are now questioning if the treatment of hookworm could effect change in severe anemia rates and thus also on maternal and child health as well. Most evidence suggests that the contribution of hookworm to maternal anemia merits that all women of child-bearing age living in endemic areas be subject to periodic anthelmintic treatment. The World Health Organization even recommends that infected pregnant women be treated after their first trimester.[14] Regardless of these suggestions, only Madagascar, Nepal and Sri Lanka have added deworming to their antenatal care programs.[53]
This lack of deworming of pregnant women is explained by the fact that most individuals still fear that anthelmintic treatment will result in adverse birth outcomes. But a 2006 study by Gyorkos et al. found that when comparing a group of pregnant women treated with mebendazole with a control placebo group, both illustrated rather similar rates in adverse birth outcomes. The treated group demonstrated 5.6% adverse birth outcomes, while the control group had 6.25% adverse birth outcomes.[52] Furthermore, Larocque et al. illustrated that treatment for hookworm infection actually led to positive health results in the infant. This study concluded that treatment with mebendazole plus iron supplements during antenatal care significantly reduced the proportion of very low birth weight infants when compared to a placebo control group.[54] Studies so far have validated recommendations to treat infected pregnant women for hookworm infection during pregnancy.
A review found that a single dose of antihelminthics (anti-worm drugs) given in the second trimester of pregnancy "may reduce maternal anaemia and worm prevalence when used in settings with high prevalence of maternal helminthiasis".[55]
The intensity of hookworm infection as well as the species of hookworm have yet to be studied as they relate to hookworm-related anemia during pregnancy. Additionally, more research must be done in different regions of the world to see if trends noted in completed studies persist.[citation needed]
Malaria co-infection
Co-infection with hookworm and Plasmodium falciparum is common in Africa.[56] Although exact numbers are unknown, preliminary analyses estimate that as many as a quarter of African schoolchildren (17.8–32.1 million children aged 5–14 years) may be coincidentally at-risk of both P. falciparum and hookworm.[57] While original hypotheses stated that co-infection with multiple parasites would impair the host's immune response to a single parasite and increase susceptibility to clinical disease, studies have yielded contrasting results. For example, one study in Senegal showed that the risk of clinical malaria infection was increased in helminth-infected children in comparison to helminth-free children while other studies have failed to reproduce such results,[58] and even among laboratory mouse experiments the effect of helminths on malaria is variable.[59]
Some hypotheses and studies suggest that helminth infections may protect against cerebral malaria due to the possible modulation of pro-inflammatory and anti-inflammatory cytokines responses.
Hygiene hypothesis and hookworm as therapy
The
Vaccines
While annual or semi-annual mass antihelminthic administration is a critical aspect of any public health intervention, many have begun to realize how unsustainable it is due to aspects such as poverty, high rates of re-infection, and diminished efficacy of drugs with repeated use. Current research, therefore, has focused on the development of a vaccine that could be integrated into existing control programs. The goal of vaccine development is not necessarily to create a vaccine with sterilizing immunity or complete protection against immunity. A vaccine that reduces the likelihood of vaccinated individuals developing severe infections and thus reduced blood and nutrient levels could still have a significant impact on the high burden of disease throughout the world.
Current research focuses on targeting two stages in the development of the worm: the larval stage and the adult stage. Research on larval antigens has focused on proteins that are members of the
Adult hookworm antigens have also been identified as potential candidates for vaccines. When adult worms attach to the intestinal mucosa of the human host, erythrocytes are ruptured in the worm's digestive tract which causes the release of free hemoglobin which is subsequently degraded by a proteolytic cascade. Several of these proteins that are responsible for this proteolytic cascade are also essential for the worm's nutrition and survival.[66] Therefore, a vaccine that could induce antibodies for these antigens could interfere with the hookworm's digestive pathway and impair the worm's survival. Three proteins have been identified: the aspartic protease-hemoglobinase APR-1, the cysteine protease-hemoglobinase CP-2, and a glutathione S-transferase.[67][68][69] Vaccination with APR-1 and CP-2 led to reduced host blood loss and fecal egg counts in dogs.[67][68] With APR-1, vaccination even led to reduced worm burden.[67] Research is currently stymied at the development of at least one of these antigens as a recombinant protein for testing in clinical trials.
Terminology
The term "hookworm" is sometimes used to refer to hookworm infection.
See also
- List of parasites (human)
References
- ^ a b c d e f g h i "CDC - Hookworm - General Information - Frequently Asked Questions (FAQs)". www.cdc.gov. 16 December 2014. Archived from the original on 22 April 2017. Retrieved 22 April 2017.
- ^ a b "CDC - Hookworm - Disease". www.cdc.gov. 10 January 2013. Archived from the original on 23 April 2017. Retrieved 22 April 2017.
- ^ a b "CDC - Hookworm - Treatment". www.cdc.gov. 10 January 2013. Archived from the original on 23 April 2017. Retrieved 22 April 2017.
- ^ PMID 27733282.)
{{cite journal}}:|first1=has generic name (help)CS1 maint: numeric names: authors list (link - ^ Prevention, CDC - Centers for Disease Control and. "CDC - Hookworm - Biology". www.cdc.gov. Archived from the original on 21 June 2017. Retrieved 21 June 2017.
- PMID 27733281.)
{{cite journal}}:|first1=has generic name (help)CS1 maint: numeric names: authors list (link - ^ "Neglected Tropical Diseases". cdc.gov. June 6, 2011. Archived from the original on 4 December 2014. Retrieved 28 November 2014.
- ISBN 978-0-7216-2921-6.
- ^ "Hookworms." The Center for Food Security and Public Health. May 2005. Iowa State University
- ^ "CDC - Zoonotic Hookworm - General Information". www.cdc.gov. 25 April 2019. Retrieved 27 December 2019.
- ^ ISBN 978-0-7216-4793-7.
- ^ PMID 15783256.
- ^ "CDC Factsheet: Hookworm" Archived 2010-09-04 at the Wayback Machine, accessed September 29, 2008
- ^ S2CID 8425278.
- PMID 8939719.
- PMID 13917420.
- PMID 7761817.
- ^ a b c d e One or more of the preceding sentences incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Ankylostomiasis". Encyclopædia Britannica. Vol. 2 (11th ed.). Cambridge University Press. p. 58.
- ^ S2CID 32970805.
- PMID 17374982.
- ^ a b "School Deworming". Public Health at a Glance. World Bank. 2003.
- ^ "Soil-transmitted helminthiasis". Wkly. Epidemiol. Rec. 83 (27/28): 237–252. 4 July 2008. Archived from the original on August 5, 2012.
- ^ "How does deworming work?" Deworm the World. <dewormtheworld.org Archived 2009-02-08 at the Wayback Machine>
- PMID 18771789.
- ISBN 978-0897935548.
- ^ Birn & Solórzano 1999, pp. 1200, 1205
- PMID 2146815.
- PMID 15049458.
- PMID 18430913.
- ^ S2CID 40314184.
- ^ a b Milton, Joseph Rosenau (1913). Preventive Medicine and Hygiene. D. Appleton. p. 119.
- ^ a b "Clinical Aspects and Treatment of the More Common Intestinal Parasites of Man (TB-33)". Veterans Administration Technical Bulletin 1946 & 1947. 10: 1–14. 1948.
- PMID 22325616.
- ^ "Global network for neglected tropical diseases receives $34 million from Gates Foundation: IDB leads campaign to greatly reduce the burden of most neglected diseases by 2020 in Latin America and the Caribbean." Press Release. Global Network for Neglected Tropical Diseases. 30 January 2009.
- ^ "Deworm the World at Clinton Global Initiative 2008 Annual Meeting: up to 10 million children to benefit from deworming!" Press Release. Deworm the World, 2008.
- PMID 17883107.
- S2CID 28630527.
- PMID 14516309.
- S2CID 20407618.
- PMID 15078265.
- PMID 29016326.
- ^ Pilkington, Ed (5 September 2017). "Hookworm, a disease of extreme poverty, is thriving in the US south. Why?". The Guardian. Retrieved 4 December 2017 – via www.TheGuardian.com.
- )
- ^ Dubini, Angelo (1843). "Nuovo verme intestinale umano (Agchylostoma duodenale), costituente un sesto genere dei Nematoidei proprii dell'uomo" [New human intestinal worm (Ancylostoma duodenale), constituting a sixth genus of the human nematoids]. Annali universali di medicina (in Italian). 106: 5–13.
- PMID 6418279.
- ^ Page, Walter H. (September 1912). "The Hookworm And Civilization: The Work Of The Rockefeller Sanitary Commission In The Souther States". The World's Work: A History of Our Time. Vol. XXIV. pp. 504–18. Retrieved 2009-07-10.
- PMID 24146438.
- ^ Wallace, Barbara; Kirkley, James; McGuire, Thomas; Austin, Diane; Goldfield, David (April 2001). Assessment of Historical, Social, and Economic Impacts of OCS Development on Gulf Coast Communities (PDF) (Report). New Orleans: U .S. Department of the Interior. pp. 35–36. Retrieved December 11, 2017.
Inadequate public health services and a general lack of basic citizen knowledge of health and hygiene reflected the weak public education system. Health problems, especially in the Gulf Coast States where frost came late, if at all, abounded in an era when active public health departments in other parts of the country were eradicating nutritional and bacterial diseases. The hookworm, an intestinal parasite, infected and chronically debilitated a great many southerners, perhaps as many as 2 million. In the 1930s, a cooperative study by the Florida State Board of Health, the Rockefeller Foundation, and Vanderbilt University found the State's adolescents aged 15 to 18 the most affected group (44.7 percent), and the Panhandle the most severely affected area with nearly half of its teenagers (49 .2 percent) infested with hookworm (Eberson, 1980; and Link, 1988). John D. Rockefeller found the situation so appalling in the early twentieth century that he established and funded the Rockefeller Sanitary Commission for the Eradication of Hookworm Disease.
{{cite report}}: Unknown parameter|agency=ignored (help) - PMID 10501641.
- ^ Ferrell, John A. (1914). The Rural School and Hookworm Disease. U.S. Government Printing Office.
- ^ )
- ^ S2CID 8637824.
- PMID 18820740.
- S2CID 46261382.
- PMID 33998661.
- PMID 18165479.
- PMID 17083720.
- PMID 14584377.
- PMID 21777589.
- PMID 16989681.
- S2CID 20956686.
- PMID 2513902.
- S2CID 23348293.
- ^ PMID 18171264.
- PMID 18396361.
- PMID 15199048.
- ^ PMID 16231975.
- ^ PMID 15122534.
- PMID 16177370.
External links
- CDC Department of Parasitic Diseases images of the hookworm life cycle
- Centers for Disease Control and Prevention
- Dog hookworm (Ancylostoma caninum) at MetaPathogen: facts, life cycle, references
- Human hookworms (Ancylostoma duodenale and Necator americanus) at MetaPathogen: facts, life cycle, references
