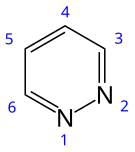
Pyridazine
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Pyridazine[1] | |||
| Systematic IUPAC name
1,2-Diazabenzene | |||
| Other names
1,2-Diazine
Orthodiazine Oizine | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 103906 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.478 | ||
| EC Number |
| ||
| 49310 | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H4N2 | |||
| Molar mass | 80.090 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 1.107 g/cm3 | ||
| Melting point | −8 °C (18 °F; 265 K) | ||
| Boiling point | 208 °C (406 °F; 481 K) | ||
| miscible | |||
| Solubility | miscible in | ||
Refractive index (nD)
|
1.52311 (23.5 °C) | ||
| Thermochemistry | |||
Std enthalpy of (ΔfH⦵298)formation |
224.9 kJ/mol | ||
| Hazards | |||
| GHS labelling:[2] | |||

| |||
| Warning | |||
| H302, H315, H319, H335 | |||
| P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |||
| Flash point | 85 °C (185 °F; 358 K) | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Pyridazine is an
heterocyclic, organic compound with the molecular formula C4H4N2. It contains a six-membered ring with two adjacent nitrogen atoms.[3] It is a colorless liquid with a boiling point of 208 °C. It is isomeric with two other diazine (C4H4N2) rings, pyrimidine and pyrazine
.
Occurrence
Pyridazines are rare in nature, possibly reflecting the scarcity of naturally occurring hydrazines, common building blocks for the synthesis of these heterocycles. The pyridazine structure is a popular pharmacophore which is found within a number of herbicides such as credazine, pyridafol and pyridate. It is also found within the structure of several drugs such as cefozopran, cadralazine, minaprine, pipofezine, and hydralazine.
Synthesis
In the course of his classic investigation on the
diketones or 4-ketoacids with hydrazines.[5]
References
- ISBN 978-0-85404-182-4.
- ^ "Pyridazine". pubchem.ncbi.nlm.nih.gov.
- ^ Gumus, S. (2011). "A computational study on substituted diazabenzenes" (PDF). Turk J Chem. 35: 803–808. Archived from the original (PDF) on 2016-03-03. Retrieved 2014-04-10.
- .
- ISBN 9780120206094.