Pyrazine
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Pyrazine[1] | |||
| Other names
1,4-Diazabenzene, p-Diazine, 1,4-Diazine, Paradiazine, Piazine, UN 1325
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.480 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H4N2 | |||
| Molar mass | 80.09 g/mol | ||
| Appearance | White crystals | ||
| Density | 1.031 g/cm3 | ||
| Melting point | 52 °C (126 °F; 325 K) | ||
| Boiling point | 115 °C (239 °F; 388 K) | ||
| Soluble | |||
| Acidity (pKa) | 0.37[2] (protonated pyrazine) | ||
| -37.6·10−6 cm3/mol | |||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H228, H315, H319, H335 | |||
| P210, P261, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 55 °C (131 °F; 328 K) c.c. | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Pyrazine is a
aromatic organic compound with the chemical formula C4H4N2. It is a symmetrical molecule with point group D2h. Pyrazine is less basic than pyridine, pyridazine and pyrimidine. It is a "deliquescent crystal or wax-like solid with a pungent, sweet, corn-like, nutty odour".[3]
Pyrazine and a variety of
Granulocytes.[4]
Synthesis
Many methods exist for the organic synthesis of pyrazine and its derivatives. Some of these are among the oldest synthesis reactions still in use.
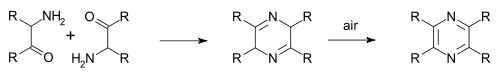
In the Staedel–Rugheimer pyrazine synthesis (1876),
The Gastaldi synthesis (1921) is another variation:[8][9]
See also
- Alkylpyrazines
- Benzene, an analog without the nitrogen atoms
- Methoxypyrazines
- Pyridazine, an analog with the second nitrogen atom in position 2
- Pyridine, an analog with only one nitrogen atom
- Pyrimidine, an analog with the second nitrogen atom in position 3
- Simple aromatic rings
References
- ISBN 978-0-85404-182-4.
- ^ Brown, H.C., et al., in Baude, E.A. and Nachod, F.C., Determination of Organic Structures by Physical Methods, Academic Press, New York, 1955.
- ^ "Pyrazine | C4H4N2 | ChemSpider". www.chemspider.com. Retrieved 4 January 2022.
- PMID 12650854.
- Berichte der deutschen chemischen GesellschaftVolume 9, Issue 1, pp. 563–564, 1876
- ISBN 0-582-01421-2
- ^ G. Gastaldi, Gazz. Chim. Ital. 51, (1921) 233
- ISBN 0-521-78284-8