L-amino-acid oxidase
| L-amino acid oxidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
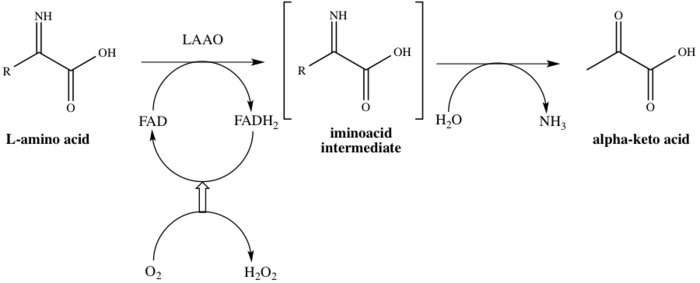
In
- an L-amino acid + H2O + O2 a 2-oxo acid + NH3 + H2O2
The enzyme was first described in 1944 by A. Zeller and A. Maritz.
As suggested by the name of the family, LAAOs are

Abundance
Snake Venom
Although LAAOs are present in a variety of eukaryotic and prokaryotic organisms,[5] snake venom is a particularly rich source of the enzyme and the LAAOs are proposed to supply toxic effects upon envenomation.[3][6][7] LAAOs that have been purified from the venoms of various snake species have proven to be the best suitors for examining this novel family of enzymes.[8] It has been determined in most cases concerning the snake families, such as Viperidae, Crotalidae, and Elapidae, that snake venom-LAAO (sv-LAAO) constitutes about 1-9% of the total protein quantity.[9]
Structure
Most sv-LAAOs are reported as being
One unusual characteristic reported for sv-LAAOs regards the cold inactivation and heat reactivation properties of the protein.[11] Thereby, most sv-LAAOs are considered to be thermolabile enzymes.[9]
This enzyme belongs to the family of
Biological Function
Specificity
The specific activities of sv-LAAOs with various L-amino acids have been explored. Many studies show that a number of sv-LAAOs exhibit a preference for hydrophobic L-amino acids as substrates.[12] For example, results have indicated that most sv-LAAOs demonstrate relatively high specificities toward hydrophobic amino acids such as L-Met, L-Leu, and L-Ile in addition to aromatic amino acids such as L-Phe and L-Trp.[8][13]
Activity
This enzyme participates in 8
A variety of biological activities have been discovered from isolated sv-LAAOs including
In recent studies, it has been shown that LAAOs have been isolated from the skin and/or gill mucous secretions of rockfish, great sculpin, and flounder.[17] The presence of these enzymes were identified to be a unique type of antibacterial protein in the external defense employed by certain fish species.
Hydrogen Peroxide Formation
Notably, because of its potential in relevant
In one case study, it was reported that the sv-LAAO (isolated from C. durissus cascavella venom) caused the rupture of bacteria membranes while promoting extravasation, or leakage, of plasmatic contents out of the cellular structure. They argued that the amount of hydrogen peroxide generated was sufficient to inhibit bacterial growth, and that the ability of the enzyme to bind to bacterial membranes is not important in its antibacterial activity.[18]
Disease Relevance
Cardiovascular Disease
Venom-Induced Pathology
As a consequence of the numerous activities the enzyme participates in, it could be deduced that there is likely some enzymatic function regarding the complex network of venom toxin activities.[4] However, the role of sv-LAAOs in venom-induced pathology has not reported to be sufficiently assessed.
Evolution
By virtue of its antibacterial properties, it has been speculated that sv-LAAOs are in part responsible for the maintenance and stabilization of both the venom and venom gland in snake species.[4]
References
- .
- PMID 19101583.
- ^ S2CID 25067549.
- ^ PMID 23010165.
- PMID 23772385.
- ^ PMID 12175601.
- ^ PMID 21059402.
- ^ PMID 10944103.
- ^ PMID 22579637.
- ^ Baby J, Sheeja RS, Jeevitha MV, Ajisha SU (2011). "Pharmacological effects of snake venom L-amino acid oxidase". International Journal of Research in Ayurveda and Pharmacy. 2 (1): 114–120.
- PMID 4967582.
- PMID 8080286.
- PMID 17854853.
- PMID 19689417.
- PMID 18294891.
- PMID 7886693.
- PMID 20728563.
- PMID 16307769.
Further reading
- Meister A, Wellner D (1963). "Flavoprotein amino acid oxidase". In Boyer PD, Lardy H, Myrbäck K (eds.). The Enzymes. Vol. 7 (2nd ed.). New York: Academic Press. pp. 609–648.
- Wellner D, Meister A (July 1960). "Crystalline L-amino acid oxidase of Crotalus adamanteus". The Journal of Biological Chemistry. 235: 2013–8. PMID 13843884.

