Lithium cobalt oxide
 | |
| |
| Names | |
|---|---|
| IUPAC name
lithium cobalt(III) oxide
| |
| Other names
lithium cobaltite
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.032.135 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| LiCoO 2 | |
| Molar mass | 97.87 g mol−1 |
| Appearance | dark blue or bluish-gray crystalline solid |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
harmful |
| GHS labelling: | |
 
| |
| Danger | |
| H317, H350, H360 | |
| P201, P202, P261, P272, P280, P281, P302+P352, P308+P313, P321, P333+P313, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium cobalt oxide, sometimes called lithium cobaltate
Lithium cobalt oxide is a dark blue or bluish-gray crystalline solid,
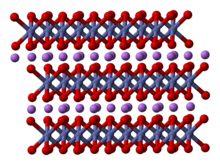
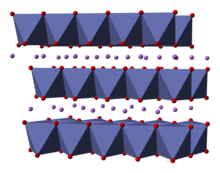
Structure
The structure of LiCoO
2 has been studied with numerous techniques including
The solid consists of layers of monovalent lithium cations (Li+
) that lie between extended anionic sheets of cobalt and oxygen atoms, arranged as edge-sharing octahedra, with two faces parallel to the sheet plane.[6] The cobalt atoms are formally in the trivalent oxidation state (Co3+
) and are sandwiched between two layers of oxygen atoms (O2−
).
In each layer (cobalt, oxygen, or lithium), the atoms are arranged in a regular triangular lattice. The lattices are offset so that the lithium atoms are farthest from the cobalt atoms, and the structure repeats in the direction perpendicular to the planes every three cobalt (or lithium) layers. The point group symmetry is in Hermann-Mauguin notation, signifying a unit cell with threefold improper rotational symmetry and a mirror plane. The threefold rotational axis (which is normal to the layers) is termed improper because the triangles of oxygen (being on opposite sides of each octahedron) are anti-aligned.[7]
Preparation
Fully reduced lithium cobalt oxide can be prepared by heating a stoichiometric mixture of

Nanometer-size particles more suitable for cathode use can also be obtained by calcination of
2O
4·2H
2O, in the form of rod-like crystals about 8 μm long and 0.4 μm wide, with lithium hydroxide LiOH, up to 750–900 °C.[9]
A third method uses
Use in rechargeable batteries
The usefulness of lithium cobalt oxide as an intercalation electrode was discovered in 1980 by an
The compound is now used as the cathode in some rechargeable
xCoO
2 with 0 < x < 1.[3]
Batteries produced with LiCoO
2 cathodes have very stable capacities, but have lower capacities and power than those with cathodes based on (especially nickel-rich)
2 decomposition generates oxygen, which then reacts with the organic electrolyte of the cell, this reaction is often seen in Lithium-Ion batteries where the battery becomes highly volatile and must be recycled in a safe manner. The decomposition of LiCoO2 is a safety concern due to the magnitude of this highly exothermic reaction, which can spread to adjacent cells or ignite nearby combustible material.[13]
The delithiation process is usually by chemical means,[14] although a novel physical process has been developed based on ion sputtering and annealing cycles,[15] leaving the material properties intact.
See also
References
- ^ 442704 - Lithium cobalt(III) oxide (2012-09-14). "Sigma-Aldrich product page". Sigmaaldrich.com. Retrieved 2013-01-21.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ^ a b c Ondřej Jankovský, Jan Kovařík, Jindřich Leitner, Květoslav Růžička, David Sedmidubský (2016) "Thermodynamic properties of stoichiometric lithium cobaltite LiCoO2". Thermochimica Acta, volume 634, pages 26-30.
- ^ LinYi Gelon New Battery Materials Co., Ltd, "Lithium Cobalt Oxide (LiCoO2) for lithium ion battery ". Catalog entry, accessed on 2018-04-10,
- .
- ^ S2CID 34357573.
- ^ .
- ISSN 2365-6549.
- ^ .
- ^ .
- S2CID 258406065.
- ^ Doughty, Daniel; Pesaran, Ahmad. "Vehicle Battery Safety Roadmap Guidance" (PDF). National Renewable Energy Laboratory. Retrieved 19 January 2013.
- .
- ISSN 1944-8244.

