Lithium sulfide

| |

| |
 | |
| Names | |
|---|---|
| IUPAC name
Lithium hydrosulfide
| |
| Preferred IUPAC name
Lithium sulfide | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.032.013 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Li2S | |
| Molar mass | 45.95 g/mol |
| Appearance | white solid |
| Density | 1.67 g/cm3 |
| Melting point | 938 °C (1,720 °F; 1,211 K) |
| Boiling point | 1,372 °C (2,502 °F; 1,645 K) |
| very soluble, hydrolyses to LiOH and H2S | |
| Solubility | very soluble in ethanol |
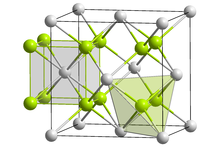
| Structure | |
Antifluorite (cubic), cF12
| |
| Fm3m, No. 225 | |
| Tetrahedral (Li+); cubic (S2−) | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
63 J/mol K |
Std enthalpy of (ΔfH⦵298)formation |
-9.401 kJ/g or -447 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
240 mg/kg (oral, rat)[1] |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Other anions
|
Lithium oxide Lithium selenide Lithium telluride Lithium polonide |
Other cations
|
Sodium sulfide Potassium sulfide Rubidium sulfide Caesium sulfide |
Related compounds
|
Lithium hydrosulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium sulfide is the
deliquescent powder. In air, it easily hydrolyses to release hydrogen sulfide (rotten egg odor).[2]
Preparation
Lithium sulfide is prepared by treating lithium with sulfur. This reaction is conveniently conducted in
anhydrous ammonia.[3]
- 2 Li + S → Li2S
The THF-soluble
superhydride.[4]
Reactions and applications
Lithium sulfide has been considered for use in lithium–sulfur batteries.[5]
References
- ^ https://chem.nlm.nih.gov/chemidplus/rn/12136-58-2 [dead link]
- ISBN 0-7506-3365-4.
- ISBN 978-0-470-13246-3.
- .
- ^ "Battery claims greater capacity than lithium ion". Electronics Weekly. 12 July 2005. Retrieved 2005-09-16.

