Lithium ruthenate
Appearance
| |
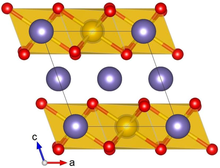
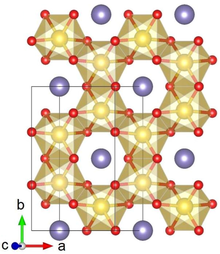 Crystal structure with Ru shown in yellow, Li in purple and O in red
| |
 Scale bar 0.1 mm[1]
| |
| Names | |
|---|---|
| Preferred IUPAC name
Lithium ruthenate | |
| Identifiers | |
3D model (
JSmol ) |
|
| |
| |
| Properties | |
| Li2RuO3 | |
| Appearance | Dark blue crystals |
| Structure | |
| Monoclinic, P21/m[2] | |
Formula units (Z)
|
4 |
| Related compounds | |
Other anions
|
Lithium iridate, lithium platinate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium ruthenate, Li2RuO3, is a chemical compound of lithium, ruthenium and oxygen. It has a layered honeycomb crystal structure, and can be prepared by direct calcination of Ru metal and lithium carbonate at ca. 700 °C.[2] It is a potential lithium-ion battery electrode material,[2] though this application is hindered by the high costs of Ru, as compared to the cheaper Li2MnO3 alternative.[3]
References
- PMID 27748402.
- ^ .
- ISBN 978-0-387-34445-4.
