Methyllithium

| |
| Names | |
|---|---|
| IUPAC name
Methyllithium
| |
| Other names
Lithium methanide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 3587162 | |
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.011.843 |
| EC Number |
|
| 288 | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CH3Li | |
| Molar mass | 21.98 g·mol−1 |
| Reacts | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
pyrophoric |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Methyllithium is the simplest
Synthesis
In the direct synthesis,
- 2 Li + MeBr → LiMe + LiBr
The
Reactivity
Methyllithium is both strongly
Methyllithium is mainly used as the synthetic equivalent of the methyl anion synthon. For example, ketones react to give tertiary alcohols in a two-step process:
- Ph2CO + MeLi → Ph2C(Me)OLi
- Ph2C(Me)OLi + H+ → Ph2C(Me)OH + Li+
Nonmetal halides are converted to methyl compounds with methyllithium:
- PCl3 + 3 MeLi → PMe3 + 3 LiCl
Such reactions more commonly employ the Grignard reagents methylmagnesium halides, which are often equally effective, and less expensive or more easily prepared in situ.
It also reacts with carbon dioxide to give Lithium acetate:
- CH3Li + CO2 → CH3CO2−Li+
- ZrCl4 + 6 MeLi → Li2ZrMe6 + 4 LiCl
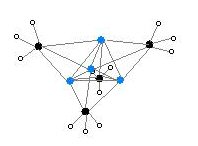
Structure
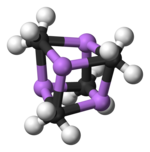
Two structures have been verified by single crystal
Colour code: Li- purple C- black H- white
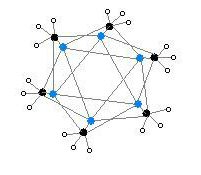
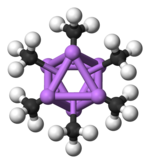
The hexameric form features hexagonal prisms with Li and C atoms again at alternate corners.
Colour code: Li- purple C- black H- white
The degree of aggregation, "n" for (MeLi)n, depends upon the solvent and the presence of additives (such as lithium bromide). Hydrocarbon solvents such as benzene[7] favour formation of the hexamer, whereas ethereal solvents favour the tetramer.
Bonding
These clusters are considered "electron-deficient," that is, they do not follow the
The strength of the C-Li bond has been estimated at around 57
References
- ^ Lusch, M. J.; Phillips, W. V.; Sieloff, R. F.; Nomura, G. S.; House, H. O. (1984). "Preparation of Low-Halide Methyllithium". Organic Syntheses. 62: 101; Collected Volumes, vol. 7, p. 346.
- .
- S2CID 224930545.
- ISBN 9780471264187.
- .
- ISBN 978-3-527-29390-2.
- ^ .