Peptide nucleic acid
Peptide nucleic acid (PNA) is an artificially synthesized polymer similar to DNA or RNA.[1]
Synthetic peptide nucleic acid
PNA is not known to occur naturally but N-(2-aminoethyl)-glycine (AEG), the backbone of PNA, has been hypothesized to be an early form of genetic molecule for life on Earth and is produced by cyanobacteria and is a neurotoxin.[4]
PNA was invented by Peter E. Nielsen (Univ. Copenhagen), Michael Egholm (Univ. Copenhagen), Rolf H. Berg (Risø National Lab), and Ole Buchardt (Univ. Copenhagen) in 1991.[1]
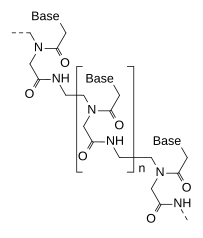
Structure
DNA and RNA have a
Binding
Since the backbone of PNA contains no charged phosphate groups, the binding between PNA/DNA strands is stronger than between DNA/DNA strands due to the lack of electrostatic repulsion. Unfortunately, this also causes it to be rather hydrophobic, which makes it difficult to deliver to body cells in solution without being flushed out of the body first. Early experiments with homopyrimidine strands (strands consisting of only one repeated pyrimidine base) have shown that the Tm ("melting" temperature) of a 6-base thymine PNA/adenine DNA double helix was 31 °C in comparison to an equivalent 6-base DNA/DNA duplex that denatures at a temperature less than 10 °C. Mixed base PNA molecules are true mimics of DNA molecules in terms of base-pair recognition. PNA/PNA binding is stronger than PNA/DNA binding.
PNA translation from other nucleic acids
Several labs have reported sequence-specific polymerization of peptide nucleic acids from DNA or RNA templates.[6][7][8] Liu and coworkers used these polymerization methods to evolve functional PNAs with the ability to fold into three-dimensional structures, similar to proteins, aptamers and ribozymes.[6]
Delivery
In 2015, Jain et al. described a trans-acting DNA-based amphiphatic delivery system for convenient delivery of poly A tailed uncharged nucleic acids (UNA) such as PNAs and morpholinos, so that several UNA's can be easily screened ex vivo.[9][non-primary source needed]
PNA world hypothesis
It has been hypothesized that the earliest life on Earth may have used PNA as a genetic material due to its extreme robustness, simpler formation, and possible spontaneous polymerization at 100 °C[10] (while water at standard pressure boils at this temperature, water at high pressure—as in deep ocean—boils at higher temperatures). If this is so, life evolved to a DNA/RNA-based system only at a later stage.[11][12] Evidence for this PNA world hypothesis is, however, far from conclusive.[13] If it existed though, it must have preceded the widely accepted RNA world.
Applications
Applications include alteration of gene expression - both as inhibitor and promoter in different cases, antigene and antisense therapeutic agent, anticancer agent, antiviral, antibacterial and antiparasitic agent, molecular tools and probes of biosensor, detection of DNA sequences, and nanotechnology.[14][15]
PNAs can be used to improve high-throughput 16S ribosomal RNA gene sequencing of plant and soil samples by blocking amplification of contaminant plastid and mitochondrial sequences.[16]
Cellular – Functional Antagonism/Inhibition. In 2001, Strauss and colleagues reported the design of an application for PNA oligomers in living mammalian cells. The Xist chromatin binding region was first elucidated in female mouse fibroblastic cells, and embryonic stem cells though the use of a PNA molecular antagonist. The novel PNA approach directly demonstrated function of a lncRNA. The long non-coding (lncRNA) RNA, Xist directly binds to the inactive X-chromosome. Functional PNA inhibition experiments revealed that specific repeat regions of the Xist RNA were responsible for chromatin binding, and hence could be considered domain regions of the RNA transcript. The PNA molecular antagonist was administered to living cells and functionally inhibited the association of Xist with inactive X-chromosome using the approach for studying noncoding RNA function in living cells called peptide nucleic acid (PNA) interference mapping. In the reported experiments, a single 19-bp antisense cell-permeating PNA targeted against a particular region of Xist RNA caused the disruption of the Xi. The association of the Xi with macro-histone H2A is also disturbed by PNA interference mapping.[17]
See also
- Clicked peptide polymer
- Glycol nucleic acid
- Oligonucleotide synthesis
- Peptide synthesis
- Threose nucleic acid
References
Further reading
- Nielsen PE (December 2008). "Triple Helix: Designing a New Molecule of Life". Scientific American. 299 (6): 64–71. PMID 19143446.
- Krämer R, Mokhir A (2012). "Chapter 12. Metal Complex Derivatives of Peptide Nucleic Acids (PNA)". In Sigel A, Helmut Sigel H, Sigel RK (eds.). Interplay between Metal Ions and Nucleic Acids. Metal Ions in Life Sciences. Vol. 10. Springer. pp. 319–40. PMID 22210345.
- Shakeel S, Karim S, Ali A (June 2006). "Peptide nucleic acid (PNA)—a review". Journal of Chemical Technology & Biotechnology. 81 (6): 892–9. .
- Bhadauria, Vijai (June 2017). Next-generation sequencing and bioinformatics for plant science. Caister Academic Press. ISBN 978-1-910190-66-1.
- Kaihatsu K, Janowski BA, Corey DR (June 2004). "Recognition of chromosomal DNA by PNAs". Chemistry & Biology. 11 (6): 749–58. PMID 15217608.
- Ng PS, Bergstrom DE (January 2005). "Alternative nucleic acid analogues for programmable assembly: hybridization of LNA to PNA". Nano Letters. 5 (1): 107–11. PMID 15792422.
- Paulasova P, Pellestor F (2004). "The peptide nucleic acids (PNAs): a new generation of probes for genetic and cytogenetic analyses". Annales de Génétique. 47 (4): 349–58. PMID 15581832.
- Castelvecchi D (June 2004). "A New Game of Life". Blog at WordPress.com.
- Nielsen PE, Egholm M (1999). "An Introduction to Peptide Nucleic Acid" (PDF). Curr. Issues Mol. Biol. 1 (2): 89–104. PMID 11475704.
