Silicon disulfide
Appearance
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
silicon(IV) sulfide
| |
| Other names
silicon disulfide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.033.935 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| SiS2 | |
| Molar mass | 92.218 g/mol |
| Appearance | White (samples are sometimes grey or brown) needles. Rotten egg smell in moist air. |
| Density | 1.853 g/cm3 |
| Melting point | 1,090 °C (1,990 °F; 1,360 K) sublimes |
| Decomposes | |
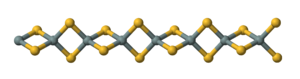
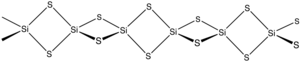
| Structure | |
Orthorhombic, oI12
| |
| Ibam, No.72[1] | |
Tetrahedral
| |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
silicon dioxide |
Other cations
|
carbon disulfide germanium disulfide tin(IV) sulfide lead(IV) sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Silicon disulfide is the
forms
of SiO2.
Synthesis, structure, and properties
The material is formed by heating silicon and sulfur or by the exchange reaction between
tetrahedra, -Si(μ-S)2Si(μ-S)2-.[2]
Like other silicon sulfur-compounds (e.g., bis(trimethylsilyl)sulfide) SiS2 hydrolyzes readily to release H2S. In liquid ammonia it is reported to form the imide Si(NH)2 and NH4SH,[3] but a recent report has identified crystalline (NH4)2[SiS3(NH3)]·2NH3 as a product which contains the tetrahedral thiosilicate anion, SiS3(NH3)2-.[4]
Reaction with ethanol gives the alkoxide tetraethyl orthosilicate and H2S.[3] With bulky tert-butanol, alcoholysis gives tris(tert-butoxy)silanethiol:[5]
- 3 (CH3)3COH + SiS2 → [(CH3)3CO]3SiSH + H2S
Reaction with
aluminum sulfide give thiosilicates.[3]
SiS2 is claimed to occur in certain interstellar objects.[6]
References
- .
- picometers, when in fact that distance describes rSiS.
- ^ ISBN 978-0-08-022057-4.
- PMID 19421590.
- .
- Bibcode:1993A&A...278..226G.

