Energy density
| Energy density | |
|---|---|
m3 | |
Other units | J/L, W⋅h/L |
| In SI base units | m−1⋅kg⋅s−2 |
Derivations from other quantities | U = E/V |
| Dimension | |
In physics, energy density is the amount of energy stored in a given system or region of space per unit volume. It is sometimes confused with energy per unit mass which is properly called specific energy or gravimetric energy density.
Often only the useful or extractable energy is measured, which is to say that inaccessible energy (such as
Energy per unit volume has the same physical units as pressure and in many situations is synonymous. For example, the energy density of a magnetic field may be expressed as and behaves like a physical pressure. Likewise, the energy required to compress a gas to a certain volume may be determined by multiplying the difference between the gas pressure and the external pressure by the change in volume. A pressure gradient describes the potential to perform work on the surroundings by converting internal energy to work until equilibrium is reached.
Overview
There are different types of energy stored in materials, and it takes a particular type of reaction to release each type of energy. In order of the typical magnitude of the energy released, these types of reactions are:
Types of energy content
There are several different types of energy content. One is the theoretical total amount of
There are two kinds of heat of combustion:
- The higher value (HHV), or gross heat of combustion, includes all the heat released as the products cool to room temperature and whatever water vapor is present condenses.
- The lower value (LHV), or net heat of combustion, does not include the heat which could be released by condensing water vapor, and may not include the heat released on cooling all the way down to room temperature.
A convenient table of HHV and LHV of some fuels can be found in the references.[2]
In energy storage and fuels
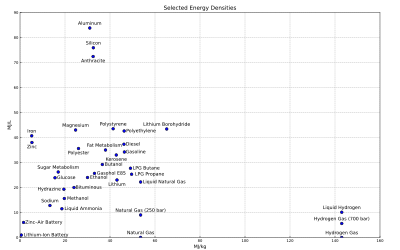
In energy storage applications the energy density relates the energy in an energy store to the volume of the storage facility, e.g. the fuel tank. The higher the energy density of the fuel, the more energy may be stored or transported for the same amount of volume. Given the high energy density of gasoline, the exploration of alternative media to store the energy of powering a car, such as hydrogen or battery, is strongly limited by the energy density of the alternative medium. The same mass of lithium-ion storage, for example, would result in a car with only 2% the range of its gasoline counterpart. If sacrificing the range is undesirable, it becomes necessary to carry that much more fuel.
The energy density of a fuel per unit mass is called the
Energy density differs from
No single energy storage method boasts the best in specific power, specific energy, and energy density. Peukert's law describes how the amount of useful energy that can be obtained (for a lead-acid cell) depends on how quickly it is pulled out.
Alternative options are discussed for energy storage to increase energy density and decrease charging time.[10][11][12][13]
The figure above shows the
Some values may not be precise because of
Generally the density values for chemical fuels do not include the weight of the oxygen required for combustion. The
List of material energy densities
This article or section appears to contradict itself. (April 2019) |
The following unit conversions may be helpful when considering the data in the tables: 3.6
In chemical reactions (oxidation)
Unless otherwise stated, the values in the following table are
2O is liquid, it is generally less than the higher heat of combustion. But in the most relevant case of hydrogen, ΔG is 113 MJ/kg if water vapor is produced, and 118 MJ/kg if liquid water is produced, both being less than the lower heat of combustion (120 MJ/kg).[14]
| Material | Specific energy (MJ/kg) |
Energy density (MJ/L) |
Specific energy ( W⋅h/kg )
|
Energy density (W⋅h/L) |
Comment |
|---|---|---|---|---|---|
| Hydrogen, liquid | 141.86 (HHV) 119.93 (LHV) |
10.044 (HHV) 8.491 (LHV) |
39,405.6 (HHV) 33,313.9 (LHV) |
2,790.0 (HHV) 2,358.6 (LHV) |
Energy figures apply after reheating to 25 °C.[15]
See note above about use in fuel cells. |
| Hydrogen, gas (681 atm, 69 MPa, 25 °C) | 141.86 (HHV) 119.93 (LHV) |
5.323 (HHV) 4.500 (LHV) |
39,405.6 (HHV) 33,313.9 (LHV) |
1,478.6 (HHV) 1,250.0 (LHV) |
Date from same reference as for liquid hydrogen.[15]
High-pressure tanks weigh much more than the hydrogen they can hold. The hydrogen may be around 5.7% of the total mass,[16] giving just 6.8 MJ per kg total mass for the LHV. See note above about use in fuel cells. |
atm or 101.3 kPa, 25 °C)
|
141.86 (HHV) 119.93 (LHV) |
0.01188 (HHV) 0.01005 (LHV) |
39,405.6 (HHV) 33,313.9 (LHV) |
3.3 (HHV) 2.8 (LHV) |
[15] |
| Methane (101.3 kPa, 15 °C) | 55.6 | 0.0378 | 15,444.5 | 10.5 | |
| LNG (NG at −160 °C) | 53.6[17] | 22.2 | 14,888.9 | 6,166.7 | |
| CNG (NG compressed to 247 atm, 25 MPa ≈ 3,600 psi) | 53.6[17] | 9 | 14,888.9 | 2,500.0 | |
| Natural gas | 53.6[17] | 0.0364 | 14,888.9 | 10.1 | |
| LPG propane | 49.6 | 25.3 | 13,777.8 | 7,027.8 | [18] |
| LPG butane | 49.1 | 27.7 | 13,638.9 | 7,694.5 | [18] |
| Gasoline (petrol) | 46.4 | 34.2 | 12,888.9 | 9,500.0 | [18] |
| Polypropylene plastic | 46.4[19] | 41.7 | 12,888.9 | 11,583.3 | |
| Polyethylene plastic | 46.3[19] | 42.6 | 12,861.1 | 11,833.3 | |
| Residential heating oil | 46.2 | 37.3 | 12,833.3 | 10,361.1 | [18] |
| Diesel fuel | 45.6 | 38.6 | 12,666.7 | 10,722.2 | [18] |
100LL Avgas
|
44.0[20] | 31.59 | 12,222.2 | 8,775.0 | |
| Jet fuel (e.g. kerosene) | 43[21][22][23] | 35 | 11,944.4 | 9,722.2 | Aircraft engine |
Gasohol E10 (10% ethanol 90% gasoline by volume)
|
43.54 | 33.18 | 12,094.5 | 9,216.7 | |
| Lithium | 43.1 | 23.0 | 11,972.2 | 6,388.9 | |
| Biodiesel oil (vegetable oil) | 42.20 | 33 | 11,722.2 | 9,166.7 | |
| DMF (2,5-dimethylfuran) | 42[24] | 37.8 | 11,666.7 | 10,500.0 | [clarification needed] |
| Paraffin wax | 42[25] | 37.8 | 11,700 | 10,500 | |
Crude oil (tonne of oil equivalent )
|
41.868 | 37[17] | 11,630 | 10,278 | |
| Polystyrene plastic | 41.4[19] | 43.5 | 11,500.0 | 12,083.3 | |
| Body fat | 38 | 35 | 10,555.6 | 9,722.2 | Metabolism in human body (22% efficiency[26]) |
| Butanol | 36.6 | 29.2 | 10,166.7 | 8,111.1 | |
| Gasohol E85 (85% ethanol 15% gasoline by volume) | 33.1 | 25.65[citation needed] | 9,194.5 | 7,125.0 | |
| Graphite | 32.7 | 72.9 | 9,083.3 | 20,250.0 | |
| Coal, anthracite | 26–33 | 34–43 | 7,222.2–9,166.7 | 9,444.5–11,944.5 | Figures represent perfect combustion not counting oxidizer, but efficiency of conversion to electricity is ≈36%[6] |
| Silicon | 32.6 | 75.9 | 9,056 | 21,080 | See Table 1 [27] |
| Aluminium | 31.0 | 83.8 | 8,611.1 | 23,277.8 | |
| Ethanol | 30 | 24 | 8,333.3 | 6,666.7 | |
| DME | 31.7 (HHV) 28.4 (LHV) |
21.24 (HHV) 19.03 (LHV) |
8,805.6 (HHV) 7,888.9 (LHV) |
5,900.0 (HHV) 5,286.1 (LHV) |
[28][29] |
| Polyester plastic | 26.0[19] | 35.6 | 7,222.2 | 9,888.9 | |
| Magnesium | 24.7 | 43.0 | 6,861.1 | 11,944.5 | |
| Phosphorus (white) | 24.30 | 44.30 | 6,750 | 12,310 | [30] |
| Coal, bituminous | 24–35 | 26–49 | 6,666.7–9,722.2 | 7,222.2–13,611.1 | [6] |
| PET plastic (impure) | 23.5[31] | < ~32.4 | 6,527.8 | < ~9000 | |
| Methanol | 19.7 | 15.6 | 5,472.2 | 4,333.3 | |
| Titanium | 19.74 | 88.93 | 5,480 | 24,700 | burned to titanium dioxide |
| Hydrazine (combusted to N2+H2O) | 19.5 | 19.3 | 5,416.7 | 5,361.1 | |
| Liquid ammonia (combusted to N2+H2O) | 18.6 | 11.5 | 5,166.7 | 3,194.5 | |
| Potassium | 18.6 | 16.5 | 5,160 | 4,600 | burned to dry potassium oxide |
PVC plastic (improper combustion toxic )
|
18.0[19] | 25.2 | 5,000.0 | 7,000.0 | [clarification needed] |
| Wood | 18.0 | 5,000.0 | [32] | ||
| Peat briquette | 17.7 | 4,916.7 | [33] | ||
| Sugars, carbohydrates, and protein | 17 | 26.2 ( dextrose )
|
4,722.2 | 7,277.8 | Metabolism in human body (22% efficiency[34])[citation needed] |
| Calcium | 15.9 | 24.6 | 4,416.7 | 6,833.3 | [citation needed] |
| Glucose | 15.55 | 23.9 | 4,319.5 | 6,638.9 | |
| Dry cow dung and camel dung | 15.5[35] | 4,305.6 | |||
| Coal, lignite | 10–20 | 2,777.8–5,555.6 | [citation needed] | ||
| Sodium | 13.3 | 12.8 | 3,694.5 | 3,555.6 | burned to wet sodium hydroxide |
| Peat | 12.8 | 3,555.6 | |||
| Nitromethane | 11.3 | 12.85 | 3,138.9 | 3,570 | |
| Manganese | 9.46 | 68.2 | 2,630 | 18,900 | burned to manganese dioxide |
| Sulfur | 9.23 | 19.11 | 2,563.9 | 5,308.3 | burned to sulfur dioxide[36] |
| Sodium | 9.1 | 8.8 | 2,527.8 | 2,444.5 | burned to dry sodium oxide |
Battery, lithium-air rechargeable
|
9.0[37] | 2,500.0 | Controlled electric discharge | ||
Household waste
|
8.0[38] | 2,222.2 | |||
| Iron | 7.4 | 57.7 | 2052.9 | 16004.1 | burned to iron(III) oxide[39] |
| Iron | 6.7 | 52.2 | 1858.3 | 14487.2 | burned to Iron(II,III) oxide[39] |
| Zinc | 5.3 | 38.0 | 1,472.2 | 10,555.6 | |
Teflon plastic
|
5.1 | 11.2 | 1,416.7 | 3,111.1 | combustion toxic, but flame retardant |
| Iron | 4.9 | 38.2 | 1,361.1 | 10,611.1 | burned to iron(II) oxide[39] |
| Gunpowder | 4.7–11.3[40] | 5.9–12.9 | 1,600–3,580 | ||
TNT
|
4.184 | 6.92 | 1,162 | 1,920 | |
| Barium | 3.99 | 14.0 | 1,110 | 3,890 | burned to barium dioxide
|
| ANFO | 3.7 | 1,027.8 |
In nuclear reactions
| Material | Specific energy (MJ/kg) |
Energy density (MJ/L) |
Specific energy ( W⋅h/kg )
|
Energy density (W⋅h/L) |
Comment |
|---|---|---|---|---|---|
| Antimatter | 89,875,517,874 ≈ 90 PJ/kg | Depends on the density of the antimatter's form | 24,965,421,631,578 ≈ 25 TW⋅h/kg | Depends on the density of the antimatter's form | Annihilation, counting both the consumed antimatter mass and ordinary matter mass |
| Hydrogen (fusion) | 639,780,320[41] but at least 2% of this is lost to neutrinos. | Depends on conditions | 177,716,755,600 | Depends on conditions | Reaction 4H→4He |
| Deuterium (fusion) |
571,182,758[42] | Depends on conditions | 158,661,876,600 | Depends on conditions | Proposed fusion scheme for D+D→4He, by combining D+D→T+H, T+D→4He+n, n+H→D and D+D→3He+n, 3He+D→4He+H, n+H→D
|
| Deuterium+tritium (fusion) | 337,387,388[41] | Depends on conditions | 93,718,718,800 | Depends on conditions | D + T → 4He + n Being developed. |
| Lithium-6 deuteride (fusion) | 268,848,415[41] | Depends on conditions | 74,680,115,100 | Depends on conditions | 6LiD → 24He Used in weapons. |
| Plutonium-239 | 83,610,000 | 1,300,000,000–1,700,000,000 (Depends on crystallographic phase) | 23,222,915,000 | 370,000,000,000–460,000,000,000 (Depends on crystallographic phase) | Heat produced in Fission reactor
|
| Plutonium-239 | 31,000,000 | 490,000,000–620,000,000 (Depends on crystallographic phase) | 8,700,000,000 | 140,000,000,000–170,000,000,000 (Depends on crystallographic phase) | Electricity produced in Fission reactor
|
| Uranium | 80,620,000[43] | 1,539,842,000 | 22,394,000,000 | Heat produced in breeder reactor | |
| Thorium | 79,420,000[43] | 929,214,000 | 22,061,000,000 | Heat produced in breeder reactor (Experimental) | |
| Plutonium-238 | 2,239,000 | 43,277,631 | 621,900,000 | Radioisotope thermoelectric generator. The heat is only produced at a rate of 0.57 W/g. |
Other release mechanisms
| Material | Specific energy (MJ/kg) |
Energy density (MJ/L) |
Specific energy ( W⋅h/kg )
|
Energy density (W⋅h/L) |
Comment |
|---|---|---|---|---|---|
Battery, zinc-air
|
1.59 | 6.02 | 441.7 | 1,672.2 | Controlled electric discharge[44] |
| Silicon (phase change) | 1.790 | 4.5 | 500 | 1,285 | Energy stored through solid to liquid phase change of silicon[45] |
| Strontium bromide hydrate | 0.814 [46] | 1.93 | 628 | Thermal energy of phase change at 88.6 °C (361.8 K) | |
Liquid nitrogen
|
0.77[47] | 0.62 | 213.9 | 172.2 | Maximum reversible work at 77.4 K with 300 K reservoir |
| Sodium sulfur battery | 0.54–0.86 | 150–240 | |||
| Compressed air at 30 MPa | 0.5 | 0.2 | 138.9 | 55.6 | Potential energy |
| Latent heat of fusion of ice (thermal) | 0.334 | 0.334 | 93.1 | 93.1 | |
| Lithium metal battery | 1.8 | 4.32 | 500 | 1,200 | Controlled electric discharge |
| Lithium-ion battery | 0.36–0.875[50] | 0.9–2.63 | 100.00–243.06 | 250.00–730.56 | Controlled electric discharge |
| Lithium-ion battery with silicon nanowire anodes | 1.566 | 4.32 | 435[51] | 1,200[51] | Controlled electric discharge |
| Flywheel | 0.36–0.5 | 5.3 | Kinetic energy | ||
| Alkaline battery | 0.48[52] | 1.3[53] | Controlled electric discharge | ||
Nickel-metal hydride battery
|
0.41[54] | 0.504–1.46[54] | Controlled electric discharge | ||
| Lead-acid battery | 0.17 | 0.56 | 47.2 | 156 | Controlled electric discharge |
EDLC )
|
0.01–0.030[55][56][57][58][59][60][61] | 0.006–0.06[55][56][57][58][59][60] | up to 8.57[61] | Controlled electric discharge | |
| Water at 100 m dam height | 0.000981 | 0.000978 | 0.272 | 0.272 | Figures represent potential energy, but efficiency of conversion to electricity is 85–90%[62][63] |
| Electrolytic capacitor | 0.00001–0.0002[64] | 0.00001–0.001[64][65][66] | Controlled electric discharge |
In material deformation
The mechanical energy storage capacity, or
| Material | Energy density by mass
(J/kg) |
Resilience: Energy density by volume
(J/L) |
Density
(kg/L) |
Young's modulus
(GPa) |
Tensile yield strength
(MPa) |
|---|---|---|---|---|---|
| Rubber band | 1,651–6,605[67] | 2,200–8,900[67] | 1.35[67] | ||
| Steel, ASTM A228 (yield, 1 mm diameter)
|
1,440–1,770 | 11,200–13,800 | 7.80[68] | 210[68] | 2,170–2,410[68] |
| Acetals | 908 | 754 | 0.831[69] | 2.8[70] | 65 (ultimate)[70] |
| Nylon-6 | 233–1,870 | 253–2,030 | 1.084 | 2–4[70] | 45–90 (ultimate)[70] |
| Copper Beryllium 25-1/2 HT (yield) | 684 | 5,720[71] | 8.36[72] | 131[71] | 1,224[71] |
| Polycarbonates | 433–615 | 520–740 | 1.2[73] | 2.6[70] | 52–62 (ultimate)[70] |
| ABS plastics | 241–534 | 258–571 | 1.07 | 1.4–3.1[70] | 40 (ultimate)[70] |
| Acrylic | 1,530 | 3.2[70] | 70 (ultimate)[70] | ||
| Aluminium 7077-T8 (yield) | 399 | 1,120[71] | 2.81[74] | 71.0[71] | 400[71] |
| Steel, stainless, 301-H (yield) | 301 | 2,410[71] | 8.0[75] | 193[71] | 965[71] |
| Aluminium 6061-T6 (yield @ 24 °C) | 205 | 553 | 2.70[76] | 68.9[76] | 276[76] |
| Epoxy resins | 113–1,810 | 2–3[70] | 26–85 (ultimate)[70] | ||
| Douglas fir Wood | 158–200 | 96 | .481–.609[77] | 13[70] | 50 (compression)[70] |
| Steel, Mild AISI 1018
|
42.4 | 334 | 7.87[78] | 205[78] | 370 (440 Ultimate)[78] |
| Aluminium (not alloyed) | 32.5 | 87.7 | 2.70[79] | 69[70] | 110 (ultimate)[70] |
| Pine (American Eastern White, flexural) | 31.8–32.8 | 11.1–11.5 | .350[80] | 8.30–8.56 (flexural)[80] | 41.4 (flexural)[80] |
| Brass | 28.6–36.5 | 250–306 | 8.4–8.73[81] | 102–125[70] | 250 (ultimate)[70] |
| Copper | 23.1 | 207 | 8.93[81] | 117[70] | 220 (ultimate)[70] |
| Glass | 5.56–10.0 | 13.9–25.0 | 2.5[82] | 50–90[70] | 50 (compression)[70] |
In batteries
| Storage device | Energy content (Joule) |
Energy content (W⋅h) |
Energy type | Typical mass (g) |
Typical dimensions (diameter × height in mm) |
Typical volume (mL) | Energy density by volume (MJ/L) |
Energy density by mass (MJ/kg) |
|---|---|---|---|---|---|---|---|---|
| Alkaline AA battery[83] | 9,360 | 2.6 | Electrochemical | 24 | 14.2 × 50 | 7.92 | 1.18 | 0.39 |
| Alkaline C battery[83] | 34,416 | 9.5 | Electrochemical | 65 | 26 × 46 | 24.42 | 1.41 | 0.53 |
| NiMH AA battery | 9,072 | 2.5 | Electrochemical | 26 | 14.2 × 50 | 7.92 | 1.15 | 0.35 |
| NiMH C battery | 19,440 | 5.4 | Electrochemical | 82 | 26 × 46 | 24.42 | 0.80 | 0.24 |
| Lithium-ion 18650 battery | 28,800–46,800 | 8–13 | Electrochemical | 44–49[84] | 18 × 65 | 16.54 | 1.74–2.83 | 0.59–1.06 |
Nuclear energy sources
The greatest energy source by far is matter itself. This energy, E = mc2, where m = ρV, ρ is the mass per unit volume, V is the volume of the mass itself and c is the speed of light. This energy, however, can be released only by the processes of nuclear fission (0.1%), nuclear fusion (1%), or the annihilation of some or all of the matter in the volume V by matter-antimatter collisions (100%).[citation needed] Nuclear reactions cannot be realized by chemical reactions such as combustion. Although greater matter densities can be achieved, the density of a neutron star would approximate the most dense system capable of matter-antimatter annihilation possible. A black hole, although denser than a neutron star, does not have an equivalent anti-particle form, but would offer the same 100% conversion rate of mass to energy in the form of Hawking radiation. In the case of relatively small black holes (smaller than astronomical objects) the power output would be tremendous.
The highest density sources of energy aside from antimatter are fusion and fission. Fusion includes energy from the sun which will be available for billions of years (in the form of sunlight) but so far (2021), sustained fusion power production continues to be elusive.
Power from fission of uranium and thorium in
Thermal power of nuclear fission reactors
The density of thermal energy contained in the core of a
Energy density of electric and magnetic fields
Electric and magnetic fields store energy. The (volumetric) energy density is given by
where E is the
In ideal (linear and nondispersive) substances, the energy density (in SI units) is
where D is the
In the case of absence of magnetic fields, by exploiting
When a pulsed laser impacts a surface, the radiant exposure, i.e. the energy deposited per unit of surface, may be called energy density or fluence.[89]
See also
Footnotes
- ^ "The Two Classes of SI Units and the SI Prefixes". NIST Guide to the SI. 2009-07-02. Retrieved 2012-01-25.
- ^ "Fossil and Alternative Fuels - Energy Content (2008)". Engineering ToolBox. Retrieved 2018-10-08.
- .
- ^ "Panasonic Develops New Higher-Capacity 18650 Li-Ion Cells." Green Car Congress. N.p., 25 Dec. 2009. Web.
- PMID 17761246.
- ^ a b c Fisher, Julia (2003). Elert, Glenn (ed.). "Energy density of coal". The Physics Factbook. Retrieved 2019-07-28.
- ^ "Heat Values of Various Fuels - World Nuclear Association." World Nuclear Association. N.p., Sept. 2016. Web.
- ^ "Overview of Storage Development DOE Hydrogen Program." Office of Energy Efficiency & Renewable Energy. N.p., May 2000. Web.
- .
- S2CID 120910476.
- PMID 22433167.
- S2CID 6994736.
- S2CID 709782.
- ^ CRC Handbook of Chemistry and Physics, 49th Edition, page D-42.
- ^ a b c College of the Desert, “Module 1, Hydrogen Properties”, Revision 0, December 2001 Hydrogen Properties. Retrieved 2014-06-08.
- ^ Mike Millikin (2014-11-18). "Toyota FCV Mirai launches in LA; initial TFCS specs; $57,500 or $499 lease; leaning on Prius analogy". Green Car Congress. Retrieved 2014-11-23.
- ^ a b c d Envestra Limited. Natural Gas Archived 2008-10-10 at the Wayback Machine. Retrieved 2008-10-05.
- ^ a b c d e IOR Energy. List of common conversion factors (Engineering conversion factors). Retrieved 2008-10-05.
- ^ a b c d e Paul A. Kittle, Ph.D. "Alternate daily cover materials and subtitle D – The selection technique" (PDF). Archived from the original (PDF) on 2008-05-27. Retrieved 2012-01-25.
- ^ "537.pdf" (PDF). June 1993. Archived from the original (PDF) on 2011-09-29. Retrieved 2012-01-25.
- ^ Gofman, Evelyn (2003). Elert, Glenn (ed.). "Energy density of aviation fuel". The Physics Factbook. Retrieved 2019-07-28.
- ^ "Handbook of Products" (PDF). Air BP. pp. 11–13. Archived from the original (PDF) on 2011-06-08.
- ^ Characteristics of Petroleum Products Stored and Dispensed (PDF), Petroleum Products Division - GN, p. 132, archived from the original (PDF) on 16 January 2017, retrieved 15 January 2017
- S2CID 4366510.
- PMID 20291038.
- ^ Justin Lemire-Elmore (2004-04-13). "The Energy Cost of Electric and Human-Powered Bicycles" (PDF). p. 5. Retrieved 2009-02-26.
properly trained athlete will have efficiencies of 22 to 26%
- ^ "Silicon as an intermediary between renewable energy and hydrogen" (PDF). Deutsche Bank Research. p. 5. Archived from the original (PDF) on 2008-11-16. Retrieved 16 November 2016.
- ^ Bossel, Ulf (July 2003). "The Physics of the Hydrogen Economy" (PDF). European Fuel Cell News. Archived from the original (PDF) on 2006-03-19. Retrieved 2019-04-06.
The Higher Heating Values are 22.7, 29.7 or 31.7 MJ/kg for methanol, ethanol and DME, respectively, while gasoline contains about 45 MJ per kg.
- ^ "Dimethyl Ether (DME)" (PDF). European Biofuels Technology Platform. 2013-11-18. Retrieved 2019-04-06. DME density and lower heating value were obtained from the table on the first page.
- ISBN 9780071422949.
- ^ "Elite_bloc.indd" (PDF). Archived from the original (PDF) on 2011-07-15. Retrieved 2010-05-07.
- ^ "Biomass Energy Foundation: Fuel Densities". Woodgas.com. Archived from the original on 2010-01-10. Retrieved 2010-05-07.
- ^ "Bord na Mona, Peat for Energy" (PDF). Bnm.ie. Archived from the original (PDF) on 2007-11-19. Retrieved 2012-01-25.
- ^ Justin Lemire-elmore (April 13, 2004). "The Energy Cost of Electric and Human-Powered Bicycle" (PDF). Retrieved 2012-01-25.
- ^ "energy buffers". Home.hccnet.nl. Retrieved 2010-05-07.
- ISBN 978-0-582-54974-6
- S2CID 96799565.
- ^ David E. Dirkse. energy buffers. "household waste 8..11 MJ/kg"
- ^ a b c Thomas C. Allison. (2013). NIST-JANAF Thermochemical Tables - SRD 13 (1.0.2) [dataset]. National Institute of Standards and Technology. https://doi.org/10.18434/T42S31
- S2CID 36130191.
- ^ a b c Calculated from fractional mass loss times c squared.
- S2CID 199405246.
- ^ a b "Computing the energy density of nuclear fuel". whatisnuclear.com. Retrieved 2014-04-17.
- ^ "Technical bulletin on Zinc-air batteries". Duracell. Archived from the original on 2009-01-27. Retrieved 2009-04-21.
- S2CID 197448761.
- ^ A. Fopah-Lele, J. G. Tamba "A review on the use of SrBr2·6H2O as a potential material for low temperature energy storage systems and building applications", Solar Energy Materials and Solar Cells 164 175-84 (2017).
- ^ C. Knowlen, A.T. Mattick, A.P. Bruckner and A. Hertzberg, "High Efficiency Conversion Systems for Liquid Nitrogen Automobiles", Society of Automotive Engineers Inc, 1988.
- ^ "Overview of lithium ion batteries" (PDF). Panasonic. Jan 2007. Archived (PDF) from the original on November 7, 2011.
- ^ "Panasonic NCR18650B" (PDF). Archived from the original (PDF) on 2015-07-22.
- ^ [48][49]
- ^ a b "Amprius' silicon nanowire Li-ion batteries power Airbus Zephyr S HAPS solar aircraft". Green Car Congress. Retrieved 2022-12-31.
- ^ "Test of Duracell Ultra Power AA". lygte-info.dk. Retrieved 2019-02-16.
- ^ "Energizer EN91 AA alkaline battery datasheet" (PDF). Retrieved 2016-01-10.
- ^ a b "Test of GP ReCyko+ AA 2700mAh (Green)". lygte-info.dk. Retrieved 2019-02-16.
- ^ a b "Maxwell supercapacitor comparison" (PDF). Archived from the original (PDF) on 2016-03-04. Retrieved 2016-01-10.
- ^ a b "Nesscap ESHSP series supercapacitor datasheet" (PDF). Archived from the original (PDF) on 2016-03-29. Retrieved 2016-01-10.
- ^ a b "Cooper PowerStor XL60 series supercapacitor datasheet" (PDF). Archived from the original (PDF) on 2016-04-02. Retrieved 2016-01-10.
- ^ a b "Kemet S301 series supercapacitor datasheet" (PDF). Archived from the original (PDF) on 2016-03-04. Retrieved 2016-01-10.
- ^ a b "Nichicon JJD series supercapatcitor datasheet" (PDF). Retrieved 2016-01-10.
- ^ a b "skelcap High Energy Ultracapacitor" (PDF). Skeleton Technologies. Archived from the original (PDF) on 2 April 2016. Retrieved 13 October 2015.
- ^ a b "3.0V 3400F Ultracapacitor cell datasheet BCAP3400 P300 K04/05" (PDF). Archived from the original (PDF) on 2020-11-01. Retrieved 2020-01-12.
- ^ "Hydroelectric Power Generation". www.mpoweruk.com. Woodbank Communications Ltd. Retrieved 13 April 2018.
- ^ "2.1 Power, discharge, head relationship | River Engineering & Restoration at OSU | Oregon State University". rivers.bee.oregonstate.edu. Archived from the original on 14 April 2018. Retrieved 13 April 2018.
Let ε = 0.85, signifying an 85% efficiency rating, typical of an older powerplant.
- ^ a b "Vishay STE series tantalum capacitors datasheet" (PDF). Retrieved 2016-01-10.
- ^ "nichicon TVX aluminum electrolytic capacitors datasheet" (PDF). Retrieved 2016-01-10.
- ^ "nichicon LGU aluminum electrolytic capacitors datasheet" (PDF). Retrieved 2016-01-10.
- ^ ISSN 1059-1028. Retrieved 2020-01-21.
- ^ a b c "MatWeb - The Online Materials Information Resource". www.matweb.com. Retrieved 2019-12-15.
- ^ PubChem. "Acetal". pubchem.ncbi.nlm.nih.gov. Retrieved 2019-12-12.
- ^ a b c d e f g h i j k l m n o p q r s t u v "Young's Modulus - Tensile and Yield Strength for common Materials". www.engineeringtoolbox.com. Retrieved 2019-12-12.
- ^ a b c d e f g h i Brush Wellman Alloy Products. "Elastic Resilience" (PDF). Technical Tidbits. Retrieved December 15, 2019.
- ^ "C17200 Alloy Specifications | E. Jordan Brookes Company". www.ejbmetals.com. Retrieved 2019-12-15.
- ^ "polycarbonate information and properties". www.polymerprocessing.com. Retrieved 2019-12-12.
- ^ "ASM Material Data Sheet". asm.matweb.com. Retrieved 2019-12-15.
- ^ Sutherland, Karen; Martin, Monica (2004). Elert, Glenn (ed.). "Density of steel". The Physics Factbook. Retrieved 2020-06-18.
- ^ a b c "Aluminum 6061-T6; 6061-T651". www.matweb.com. Retrieved 2021-06-13.
- ^ "Wood Species - Moisture Content and Weight". www.engineeringtoolbox.com. Retrieved 2019-12-12.
- ^ a b c "AISI 1018 Mild/Low Carbon Steel". AZoM.com. 2012-06-28. Retrieved 2020-01-22.
- ^ "ASM Material Data Sheet". asm.matweb.com. Retrieved 2019-12-12.
- ^ a b c "American Eastern White Pine Wood". www.matweb.com. Retrieved 2019-12-15.
- ^ a b "Mass, Weight, Density or Specific Gravity of Different Metals". www.simetric.co.uk. Retrieved 2019-12-12.
- ^ "Physical properties of glass | Saint Gobain Building Glass UK". uk.saint-gobain-building-glass.com. Retrieved 2019-12-12.
- ^ a b "Battery Energy Tables". Archived from the original on 2011-12-04.
- ^ "18650 Battery capacities".
- ^ "Supply of Uranium". world-nuclear.org. 2014-10-08. Archived from the original on 2015-10-17. Retrieved 2015-06-13.
- ^ "Facts from Cohen". Formal.stanford.edu. 2007-01-26. Archived from the original on 2007-04-10. Retrieved 2010-05-07.
- ^ "U.S. Energy Information Administration (EIA) - Annual Energy Review". Eia.doe.gov. 2009-06-26. Archived from the original on 2010-05-06. Retrieved 2010-05-07.
- S2CID 125817506.
- ^ "Terminology". Regenerative Laser Therapy.
Further reading
- The Inflationary Universe: The Quest for a New Theory of Cosmic Origins by Alan H. Guth (1998) ISBN 0-201-32840-2
- Cosmological Inflation and Large-Scale Structure by Andrew R. Liddle, David H. Lyth (2000) ISBN 0-521-57598-2
- Richard Becker, "Electromagnetic Fields and Interactions", Dover Publications Inc., 1964
External links
- ^ "Aircraft Fuels." Energy, Technology and the Environment Ed. Attilio Bisio. Vol. 1. New York: John Wiley and Sons, Inc., 1995. 257–259
- "Fuels of the Future for Cars and Trucks" – Dr. James J. Eberhardt – Energy Efficiency and Renewable Energy, U.S. Department of Energy – 2002 Diesel Engine Emissions Reduction (DEER) Workshop San Diego, California - August 25–29, 2002
- "Heat values of various fuels – World Nuclear Association". www.world-nuclear.org. Retrieved 4 November 2018.
- "Energy and Energy Types – Springer" (PDF). Retrieved 4 November 2018.



