Graphene
Graphene (
Each atom in a graphene sheet is connected to its three nearest neighbors by

Scientists theorized the potential existence and production of graphene for decades. It has likely been unknowingly produced in small quantities for centuries, through the use of pencils and other similar applications of graphite. It was possibly observed in electron microscopes in 1962, but studied only while supported on metal surfaces.[12]
In 2004, the material was rediscovered, isolated and investigated at the University of Manchester,[13][14] by Andre Geim and Konstantin Novoselov. In 2010, Geim and Novoselov were awarded the Nobel Prize in Physics for their "groundbreaking experiments regarding the two-dimensional material graphene".[15] High-quality graphene proved to be surprisingly easy to isolate.
Graphene has become a valuable and useful
The IUPAC (International Union for Pure and Applied Chemistry) recommends use of the name "graphite" for the three-dimensional material, and "graphene" only when the reactions, structural relations, or other properties of individual layers are discussed.[18] A narrower definition, of "isolated or free-standing graphene" requires that the layer be sufficiently isolated from its environment,[19] but would include layers suspended or transferred to silicon dioxide or silicon carbide.[20]
History
Structure of graphite and its intercalation compounds
In 1859, Benjamin Brodie noted the highly lamellar structure of thermally reduced graphite oxide.[21][22] In 1916, Peter Debye and Paul Scherrer determined the structure of graphite by powder X-ray diffraction.[23][24][25] The structure was studied in more detail by V. Kohlschütter and P. Haenni in 1918, who also described the properties of graphite oxide paper.[26] Its structure was determined from single-crystal diffraction in 1924.[27][28]
The theory of graphene was first explored by
Transmission electron microscopy (TEM) images of thin graphite samples consisting of a few graphene layers were published by G. Ruess and F. Vogt in 1948.[34] Eventually, single layers were also observed directly.[35] Single layers of graphite were also observed by transmission electron microscopy within bulk materials, in particular inside soot obtained by chemical exfoliation.[6]
In 1961–1962, Hanns-Peter Boehm published a study of extremely thin flakes of graphite, and coined the term "graphene" for the hypothetical single-layer structure.[36] This paper reports graphitic flakes that give an additional contrast equivalent of down to ~0.4 nm or 3 atomic layers of amorphous carbon. This was the best possible resolution for 1960 TEMs. However, neither then nor today is it possible to argue how many layers were in those flakes. Now we know that the TEM contrast of graphene most strongly depends on focusing conditions.[35] For example, it is impossible to distinguish between suspended monolayer and multilayer graphene by their TEM contrasts, and the only known way is to analyze the relative intensities of various diffraction spots. The first reliable TEM observations of monolayers are probably given in refs. 24 and 26 of Geim and Novoselov's 2007 review.[2]
Starting in the 1970s, C. Oshima and others described single layers of carbon atoms that were grown epitaxially on top of other materials.
The term "graphene" was used again in 1987 to describe single sheets of graphite as a constituent of graphite intercalation compounds,[39] which can be seen as crystalline salts of the intercalant and graphene. It was also used in the descriptions of carbon nanotubes by R. Saito and Mildred and Gene Dresselhaus in 1992,[40] and of polycyclic aromatic hydrocarbons in 2000 by S. Wang and others.[41]
Efforts to make thin films of graphite by mechanical exfoliation started in 1990.[42] Initial attempts employed exfoliation techniques similar to the drawing method. Multilayer samples down to 10 nm in thickness were obtained.[2]
In 2002, Robert B. Rutherford and Richard L. Dudman filed for a patent in the US on a method to produce graphene by repeatedly peeling off layers from a graphite flake adhered to a substrate, achieving a graphite thickness of 0.00001 inches (2.5×10−7 metres). The key to success was high-throughput visual recognition of graphene on a properly chosen substrate, which provides a small but noticeable optical contrast.[43]
Another U.S. patent was filed in the same year by Bor Z. Jang and Wen C. Huang for a method to produce graphene based on exfoliation followed by attrition.[44]
In 2014, inventor Larry Fullerton patents a process for producing single layer graphene sheets.[45]
Full isolation and characterization

Graphene was properly isolated and characterized in 2004 by
2 could be used as a "back gate" electrode to vary the charge density in the graphene over a wide range.
This work resulted in the two winning the Nobel Prize in Physics in 2010 "for groundbreaking experiments regarding the two-dimensional material graphene."[47][48][46] Their publication, and the surprisingly easy preparation method that they described, sparked a "graphene gold rush". Research expanded and split off into many different subfields, exploring different exceptional properties of the material—quantum mechanical, electrical, chemical, mechanical, optical, magnetic, etc.
Exploring commercial applications
Since the early 2000s, a number of companies and research laboratories have been working to develop commercial applications of graphene. In 2014 a
Structure
Graphene is a single layer (monolayer) of carbon atoms, tightly bound in a hexagonal honeycomb lattice. It is an allotrope of carbon in the form of a plane of sp2-bonded atoms with a molecular bond length of 0.142 nanometres.
Bonding
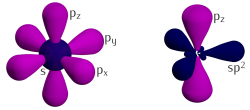
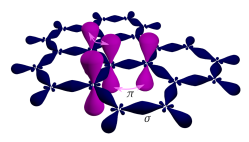
Three of the four outer-
The remaining outer-shell electron occupies a pz orbital that is oriented perpendicularly to the plane. These orbitals hybridize together to form two half-filled
Graphene sheets stack to form graphite with an interplanar spacing of 0.335
Graphene sheets in solid form usually show evidence in diffraction for graphite's (002) layering. This is true of some single-walled nanostructures.[57] However, unlayered graphene with only (hk0) rings has been found in the core of presolar graphite onions.[58] TEM studies show faceting at defects in flat graphene sheets[59] and suggest a role for two-dimensional crystallization from a melt.
Geometry

The hexagonal lattice
The hexagonal structure is also seen in scanning tunneling microscope (STM) images of graphene supported on silicon dioxide substrates[62] The rippling seen in these images is caused by conformation of graphene to the subtrate's lattice, and is not intrinsic.[62]
Stability
Ab initio calculations show that a graphene sheet is thermodynamically unstable if its size is less than about 20 nm and becomes the most stable fullerene (as within graphite) only for molecules larger than 24,000 atoms.[63]
Properties
This section may be too long to read and navigate comfortably. (October 2023) |
Electronic

Graphene is a zero-gap
However, if the in-plane direction is no longer infinite, but confined, its electronic structure would change. They are referred to as graphene nanoribbons. If it is "zig-zag", the bandgap would still be zero. If it is "armchair", the bandgap would be non-zero.
Graphene's hexagonal lattice can be regarded as two interleaving triangular lattices. This perspective was successfully used to calculate the band structure for a single graphite layer using a tight-binding approximation.[54]
Electronic spectrum
Electrons propagating through graphene's honeycomb lattice effectively lose their mass, producing
Dispersion relation
The cleavage technique led directly to the first observation of the anomalous quantum Hall effect in graphene in 2005, by Geim's group and by
When the atoms are placed onto the graphene hexagonal lattice, the overlap between the pz(π) orbitals and the s or the px and py orbitals is zero by symmetry. The pz electrons forming the π bands in graphene can therefore be treated independently. Within this π-band approximation, using a conventional
with the nearest-neighbor (π orbitals) hopping energy γ0 ≈ 2.8 eV and the
As a consequence, at low energies, even neglecting the true spin, the electrons can be described by an equation that is formally equivalent to the massless
Here vF ~ 106 m/s (.003 c) is the
The equation describing the electrons' linear dispersion relation is
where the
Single-atom wave propagation
Electron waves in graphene propagate within a single-atom layer, making them sensitive to the proximity of other materials such as
Ambipolar electron and hole transport

Graphene displays remarkable
The corresponding
2 substrates, scattering of electrons by optical phonons of the substrate is a larger effect than scattering by graphene's own phonons. This limits mobility to 40000 cm2⋅V−1⋅s−1.[72]
Charge transport has major concerns due to adsorption of contaminants such as water and oxygen molecules. This leads to non-repetitive and large hysteresis I-V characteristics. Researchers must carry out electrical measurements in vacuum. The protection of graphene surface by a coating with materials such as SiN, PMMA, h-BN, etc., have been discussed by researchers. In January 2015, the first stable graphene device operation in air over several weeks was reported, for graphene whose surface was protected by aluminum oxide.[75][76] In 2015, lithium-coated graphene exhibited superconductivity, a first for graphene.[77]
Electrical resistance in 40-nanometer-wide
Transport is dominated by two modes. One is ballistic and temperature-independent, while the other is thermally activated. Ballistic electrons resemble those in cylindrical carbon nanotubes. At room temperature, resistance increases abruptly at a particular length—the ballistic mode at 16 micrometres and the other at 160 nanometres (1% of the former length).[78]
Graphene electrons can cover micrometer distances without scattering, even at room temperature.[64]
Despite zero carrier density near the Dirac points, graphene exhibits a minimum
2 substrate may lead to local puddles of carriers that allow conduction.[65] Several theories suggest that the minimum conductivity should be ; however, most measurements are of order or greater[2] and depend on impurity concentration.[80]
Near zero carrier density graphene exhibits positive photoconductivity and negative photoconductivity at high carrier density. This is governed by the interplay between photoinduced changes of both the Drude weight and the carrier scattering rate.[81]
Graphene doped with various gaseous species (both acceptors and donors) can be returned to an undoped state by gentle heating in vacuum.[80][82] Even for dopant concentrations in excess of 1012 cm−2 carrier mobility exhibits no observable change.[82] Graphene doped with potassium in ultra-high vacuum at low temperature can reduce mobility 20-fold.[80][83] The mobility reduction is reversible on heating the graphene to remove the potassium.
Due to graphene's two dimensions, charge fractionalization (where the apparent charge of individual pseudoparticles in low-dimensional systems is less than a single quantum
Chiral half-integer quantum Hall effect

The quantum Hall effect is a quantum mechanical version of the Hall effect, which is the production of transverse (perpendicular to the main current) conductivity in the presence of a magnetic field. The quantization of the Hall effect at integer multiples (the "
Graphene shows the quantum Hall effect with respect to conductivity quantization: the effect is unordinary in that the sequence of steps is shifted by 1/2 with respect to the standard sequence and with an additional factor of 4. Graphene's Hall conductivity is , where N is the Landau level and the double valley and double spin degeneracies give the factor of 4.[2] These anomalies are present not only at extremely low temperatures but also at room temperature, i.e. at roughly 20 °C (293 K).[31]
This behavior is a direct result of graphene's chiral, massless Dirac electrons.[2][87] In a magnetic field, their spectrum has a Landau level with energy precisely at the Dirac point. This level is a consequence of the Atiyah–Singer index theorem and is half-filled in neutral graphene,[29] leading to the "+1/2" in the Hall conductivity.[32] Bilayer graphene also shows the quantum Hall effect, but with only one of the two anomalies (i.e. ). In the second anomaly, the first plateau at N = 0 is absent, indicating that bilayer graphene stays metallic at the neutrality point.[2]

Unlike normal metals, graphene's longitudinal resistance shows maxima rather than minima for integral values of the Landau filling factor in measurements of the Shubnikov–de Haas oscillations, whereby the term integral quantum Hall effect. These oscillations show a phase shift of π, known as Berry's phase.[31][65] Berry's phase arises due to chirality or dependence (locking) of the pseudospin quantum number on momentum of low-energy electrons near the Dirac points.[33] The temperature dependence of the oscillations reveals that the carriers have a non-zero cyclotron mass, despite their zero effective mass in the Dirac-fermion formalism.[31]
Graphene samples prepared on nickel films, and on both the silicon face and carbon face of
Strong magnetic fields
In magnetic fields above 10 tesla or so additional plateaus of the Hall conductivity at σxy = νe2/h with ν = 0, ±1, ±4 are observed.[95] A plateau at ν = 3[96] and the fractional quantum Hall effect at ν = 1/3 were also reported.[96][97]
These observations with ν = 0, ±1, ±3, ±4 indicate that the four-fold degeneracy (two valley and two spin degrees of freedom) of the Landau energy levels is partially or completely lifted.
Casimir effect
The Casimir effect is an interaction between disjoint neutral bodies provoked by the fluctuations of the electrodynamical vacuum. Mathematically it can be explained by considering the normal modes of electromagnetic fields, which explicitly depend on the boundary (or matching) conditions on the interacting bodies' surfaces. Since graphene/electromagnetic field interaction is strong for a one-atom-thick material, the Casimir effect is of growing interest.[98][99]
Van der Waals force
The Van der Waals force (or dispersion force) is also unusual, obeying an inverse cubic, asymptotic power law in contrast to the usual inverse quartic.[100]
'Massive' electrons
Graphene's unit cell has two identical carbon atoms and two zero-energy states: one in which the electron resides on atom A, the other in which the electron resides on atom B. However, if the two atoms in the unit cell are not identical, the situation changes. Hunt et al. show that placing hexagonal boron nitride (h-BN) in contact with graphene can alter the potential felt at atom A versus atom B enough that the electrons develop a mass and accompanying band gap of about 30 meV [0.03 Electron Volt(eV)].[101]
The mass can be positive or negative. An arrangement that slightly raises the energy of an electron on atom A relative to atom B gives it a positive mass, while an arrangement that raises the energy of atom B produces a negative electron mass. The two versions behave alike and are indistinguishable via
If the mass in graphene can be controlled, electrons can be confined to massless regions by surrounding them with massive regions, allowing the patterning of quantum dots, wires, and other mesoscopic structures. It also produces one-dimensional conductors along the boundary. These wires would be protected against backscattering and could carry currents without dissipation.[101]
Permittivity
Graphene's permittivity varies with frequency. Over a range from microwave to millimeter wave frequencies it is roughly 3.3.[102] This permittivity, combined with the ability to form both conductors and insulators, means that theoretically, compact capacitors made of graphene could store large amounts of electrical energy.
Optical
Graphene's unique optical properties produce an unexpectedly high opacity for an atomic monolayer in vacuum, absorbing πα ≈ 2.3% of light, from visible to infrared.[8][9][103] Here, α is the fine-structure constant. This is a consequence of the "unusual low-energy electronic structure of monolayer graphene that features electron and hole conical bands meeting each other at the Dirac point... [which] is qualitatively different from more common quadratic massive bands."[8] Based on the Slonczewski–Weiss–McClure (SWMcC) band model of graphite, the interatomic distance, hopping value and frequency cancel when optical conductance is calculated using Fresnel equations in the thin-film limit.
Although confirmed experimentally, the measurement is not precise enough to improve on other techniques for determining the fine-structure constant.[104]
Graphene's
A graphene-based
Saturable absorption
Such unique absorption could become saturated when the input optical intensity is above a threshold value. This nonlinear optical behavior is termed
Saturable absorption in graphene could occur at the Microwave and Terahertz band, owing to its wideband optical absorption property. The microwave saturable absorption in graphene demonstrates the possibility of graphene microwave and terahertz photonics devices, such as a microwave saturable absorber, modulator, polarizer, microwave signal processing and broad-band wireless access networks.[116]
Nonlinear Kerr effect
Under more intensive laser illumination, graphene could also possess a nonlinear phase shift due to the optical nonlinear Kerr effect. Based on a typical open and close aperture z-scan measurement, graphene possesses a giant nonlinear Kerr coefficient of 10−7 cm2⋅W−1, almost nine orders of magnitude larger than that of bulk dielectrics.[117] This suggests that graphene may be a powerful nonlinear Kerr medium, with the possibility of observing a variety of nonlinear effects, the most important of which is the soliton.[118]
Excitonic
First-principle calculations with quasiparticle corrections and many-body effects are performed to study the electronic and optical properties of graphene-based materials. The approach is described as three stages.
Spin transport
Graphene is claimed to be an ideal material for
Magnetic properties
Strong magnetic fields
Graphene's quantum Hall effect in magnetic fields above approximately 10 Teslas reveals additional interesting features. Additional plateaus of the Hall conductivity at with are observed.[95] Also, the observation of a plateau at [96] and the fractional quantum Hall effect at were reported.[96][97]
These observations with indicate that the four-fold degeneracy (two valley and two spin degrees of freedom) of the Landau energy levels is partially or completely lifted. One hypothesis is that the magnetic catalysis of symmetry breaking is responsible for lifting the degeneracy.[citation needed]
Spintronic and magnetic properties can be present in graphene simultaneously.[129] Low-defect graphene nanomeshes manufactured by using a non-lithographic method exhibit large-amplitude ferromagnetism even at room temperature. Additionally a spin pumping effect is found for fields applied in parallel with the planes of few-layer ferromagnetic nanomeshes, while a magnetoresistance hysteresis loop is observed under perpendicular fields. Charge-neutral graphene has been shown to exhibit magnetoresistance above 100% in magnetic fields of standard permanent magnets (about 0.1 tesla), a record magnetoresistivity at room temperature among all known materials.[130]
Magnetic substrates
In 2014 researchers magnetized graphene by placing it on an atomically smooth layer of magnetic yttrium iron garnet. The graphene's electronic properties were unaffected. Prior approaches involved doping graphene with other substances.[131] The dopant's presence negatively affected its electronic properties.[132]
Thermal conductivity
Thermal transport in graphene is an active area of research, which has attracted attention because of the potential for thermal management applications. Most experimental measurements have posted large uncertainties in the results of thermal conductivity due to limitations of the instruments used. Following predictions for graphene and related
It has been suggested that the isotopic composition, the ratio of 12C to 13C, has a significant impact on the thermal conductivity. For example, isotopically pure 12C graphene has higher thermal conductivity than either a 50:50 isotope ratio or the naturally occurring 99:1 ratio.[144] It can be shown by using the Wiedemann–Franz law, that the thermal conduction is phonon-dominated.[134] However, for a gated graphene strip, an applied gate bias causing a Fermi energy shift much larger than kBT can cause the electronic contribution to increase and dominate over the phonon contribution at low temperatures. The ballistic thermal conductance of graphene is isotropic.[145][146]
Potential for this high conductivity can be seen by considering graphite, a 3D version of graphene that has
Despite its 2-D nature, graphene has 3
Mechanical
The (two-dimensional) density of graphene is 0.763 mg per square meter.[citation needed]
Graphene is the strongest material ever tested,
Large-angle-bent graphene monolayer has been achieved with negligible strain, showing mechanical robustness of the two-dimensional carbon nanostructure. Even with extreme deformation, excellent carrier mobility in monolayer graphene can be preserved.[152]
The
2 cavities where an AFM tip was used to apply a stress to the sheet to test its mechanical properties. Its spring constant was in the range 1–5 N/m and the stiffness was 0.5 TPa, which differs from that of bulk graphite. These intrinsic properties could lead to applications such as NEMS as pressure sensors and resonators.[153] Due to its large surface energy and out of plane ductility, flat graphene sheets are unstable with respect to scrolling, i.e. bending into a cylindrical shape, which is its lower-energy state.[154]
As is true of all materials, regions of graphene are subject to thermal and quantum fluctuations in relative displacement. Although the amplitude of these fluctuations is bounded in 3D structures (even in the limit of infinite size), the Mermin–Wagner theorem shows that the amplitude of long-wavelength fluctuations grows logarithmically with the scale of a 2D structure, and would therefore be unbounded in structures of infinite size. Local deformation and elastic strain are negligibly affected by this long-range divergence in relative displacement. It is believed that a sufficiently large 2D structure, in the absence of applied lateral tension, will bend and crumple to form a fluctuating 3D structure. Researchers have observed ripples in suspended layers of graphene,[35] and it has been proposed that the ripples are caused by thermal fluctuations in the material. As a consequence of these dynamical deformations, it is debatable whether graphene is truly a 2D structure.[2][60][61][155][156] It has recently been shown that these ripples, if amplified through the introduction of vacancy defects, can impart a negative Poisson's ratio into graphene, resulting in the thinnest auxetic material known so far.[157]
Graphene nanosheets have been incorporated into a Ni matrix through a plating process to form Ni-graphene composites on a target substrate. The enhancement in mechanical properties of the composites is attributed to the high interaction between Ni and graphene and the prevention of the dislocation sliding in the Ni matrix by the graphene.[158]
Fracture toughness
In 2014, researchers from
Polycrystalline graphene
Various methods – most notably, chemical vapor deposition (CVD), as discussed in the section below - have been developed to produce large-scale graphene needed for device applications. Such methods often synthesize polycrystalline graphene.[162] The mechanical properties of polycrystalline graphene is affected by the nature of the defects, such as grain-boundaries (GB) and vacancies, present in the system and the average grain-size.
Graphene grain boundaries typically contain heptagon-pentagon pairs. The arrangement of such defects depends on whether the GB is in zig-zag or armchair direction. It further depends on the tilt-angle of the GB.[163] In 2010, researchers from Brown University computationally predicted that as the tilt-angle increases, the grain boundary strength also increases. They showed that the weakest link in the grain boundary is at the critical bonds of the heptagon rings. As the grain boundary angle increases, the strain in these heptagon rings decreases, causing the grain-boundary to be stronger than lower-angle GBs. They proposed that, in fact, for sufficiently large angle GB, the strength of the GB is similar to pristine graphene.[164] In 2012, it was further shown that the strength can increase or decrease, depending on the detailed arrangements of the defects.[165] These predictions have since been supported by experimental evidences. In a 2013 study led by James Hone's group, researchers probed the elastic stiffness and strength of CVD-grown graphene by combining nano-indentation and high-resolution TEM. They found that the elastic stiffness is identical and strength is only slightly lower than those in pristine graphene.[166] In the same year, researchers from UC Berkeley and UCLA probed bi-crystalline graphene with TEM and AFM. They found that the strength of grain-boundaries indeed tend to increase with the tilt angle.[167]
While the presence of vacancies is not only prevalent in polycrystalline graphene, vacancies can have significant effects on the strength of graphene. The general consensus is that the strength decreases along with increasing densities of vacancies. In fact, various studies have shown that for graphene with sufficiently low density of vacancies, the strength does not vary significantly from that of pristine graphene. On the other hand, high density of vacancies can severely reduce the strength of graphene.[168]
Compared to the fairly well-understood nature of the effect that grain boundary and vacancies have on the mechanical properties of graphene, there is no clear consensus on the general effect that the average grain size has on the strength of polycrystalline graphene.
Chemical
Graphene has a theoretical specific surface area (SSA) of 2630 m2/g. This is much larger than that reported to date for carbon black (typically smaller than 900 m2/g) or for carbon nanotubes (CNTs), from ≈100 to 1000 m2/g and is similar to activated carbon.[174] Graphene is the only form of carbon (or solid material) in which every atom is available for chemical reaction from two sides (due to the 2D structure). Atoms at the edges of a graphene sheet have special chemical reactivity. Graphene has the highest ratio of edge atoms of any
In 2013, Stanford University physicists reported that single-layer graphene is a hundred times more chemically reactive than thicker multilayer sheets.[180]
Graphene can self-repair holes in its sheets, when exposed to molecules containing carbon, such as hydrocarbons. Bombarded with pure carbon atoms, the atoms perfectly align into hexagons, completely filling the holes.[181][182]
Biological
Despite the promising results in different cell studies and proof of concept studies, there is still incomplete understanding of the full biocompatibility of graphene based materials.[183] Different cell lines react differently when exposed to graphene, and it has been shown that the lateral size of the graphene flakes, the form and surface chemistry can elicit different biological responses on the same cell line.[184]
There are indications that graphene has promise as a useful material for interacting with neural cells; studies on cultured neural cells show limited success.[185][186]
Graphene also has some utility in
Graphene can be used in biosensors; in 2015, researchers demonstrated that a graphene-based sensor be can used to detect a cancer risk biomarker. In particular, by using epitaxial graphene on silicon carbide, they were repeatably able to detect 8-hydroxydeoxyguanosine (8-OHdG), a DNA damage biomarker.[188]
Support substrate
The electronics property of graphene can be significantly influenced by the supporting substrate. Studies of graphene monolayers on clean and hydrogen(H)-passivated silicon (100) (Si(100)/H) surfaces have been performed.[189] The Si(100)/H surface does not perturb the electronic properties of graphene, whereas the interaction between the clean Si(100) surface and graphene changes the electronic states of graphene significantly. This effect results from the covalent bonding between C and surface Si atoms, modifying the π-orbital network of the graphene layer. The local density of states shows that the bonded C and Si surface states are highly disturbed near the Fermi energy.
Forms
This section may be too long to read and navigate comfortably. (October 2023) |
Monolayer sheets
In 2013 a group of Polish scientists presented a production unit that allows the manufacture of continuous monolayer sheets.[190] The process is based on graphene growth on a liquid metal matrix.[191] The product of this process was called High Strength Metallurgical Graphene. In a new study published in Nature, the researchers have used a single layer graphene electrode and a novel surface sensitive non-linear spectroscopy technique to investigate the top-most water layer at the electrochemically charged surface. They found that the interfacial water response to applied electric field is asymmetric with respect to the nature of the applied field.[192]
Bilayer graphene
Bilayer graphene displays the
One way to synthesize bilayer graphene is via chemical vapor deposition, which can produce large bilayer regions that almost exclusively conform to a Bernal stack geometry.[195]
It has been shown that the two graphene layers can withstand important strain or doping mismatch[196] which ultimately should lead to their exfoliation.
Turbostratic
Turbostratic graphene exhibits weak interlayer coupling, and the spacing is increased with respect to Bernal-stacked multilayer graphene. Rotational misalignment preserves the 2D electronic structure, as confirmed by Raman spectroscopy. The D peak is very weak, whereas the 2D and G peaks remain prominent. A rather peculiar feature is that the I2D/IG ratio can exceed 10. However, most importantly, the M peak, which originates from AB stacking, is absent, whereas the TS1 and TS2 modes are visible in the Raman spectrum.[197][198] The material is formed through conversion of non-graphenic carbon into graphenic carbon without providing sufficient energy to allow for the reorganization through annealing of adjacent graphene layers into crystalline graphitic structures.
Graphene superlattices
Periodically stacked graphene and its insulating isomorph provide a fascinating structural element in implementing highly functional superlattices at the atomic scale, which offers possibilities in designing nanoelectronic and photonic devices. Various types of superlattices can be obtained by stacking graphene and its related forms.[199] The energy band in layer-stacked superlattices is found to be more sensitive to the barrier width than that in conventional III–V semiconductor superlattices. When adding more than one atomic layer to the barrier in each period, the coupling of electronic wavefunctions in neighboring potential wells can be significantly reduced, which leads to the degeneration of continuous subbands into quantized energy levels. When varying the well width, the energy levels in the potential wells along the L-M direction behave distinctly from those along the K-H direction.
A superlattice corresponds to a periodic or quasi-periodic arrangement of different materials, and can be described by a superlattice period which confers a new translational symmetry to the system, impacting their phonon dispersions and subsequently their thermal transport properties. Recently, uniform monolayer graphene-hBN structures have been successfully synthesized via lithography patterning coupled with chemical vapor deposition (CVD).[200] Furthermore, superlattices of graphene-hBN are ideal model systems for the realization and understanding of coherent (wave-like) and incoherent (particle-like) phonon thermal transport.[201][202][203][204][205]
Graphene nanoribbons



Graphene quantum dots
A graphene quantum dot (GQD) is a graphene fragment with size less than 100 nm. The properties of GQDs are different from 'bulk' graphene due to the quantum confinement effects which only becomes apparent when size is smaller than 100 nm.[206][207][208]
Graphene oxide
Graphene oxide is usually produced through chemical exfoliation of graphite. A particularly popular technique is the improved Hummer's method.
In 2022 were performed an avaluation of biological effects of graphene oxide [2]. It was shown the graphene oxide at low doses was evaluated for its biological effects on larvae and the imago of Drosophila melanogaster. Oral administration of graphene oxide at concentrations of 0.02-1% has a beneficial effect on the developmental rate and hatching ability of larvae. Long-term administration of a low dose of graphene oxide extends Drosophila lifespan and significantly enhances resistance to environmental stresses. These suggest about graphene oxide affects carbohydrate and lipid metabolism in adult Drosophila. These findings might provide a useful reference to assess the biological effects of graphene oxide, which could play an important role in a variety of graphene-based biomedical applications.[214]
Chemical modification
Refluxing single-layer graphene oxide (SLGO) in solvents leads to size reduction and folding of individual sheets as well as loss of carboxylic group functionality, by up to 20%, indicating thermal instabilities of SLGO sheets dependent on their preparation methodology. When using thionyl chloride, acyl chloride groups result, which can then form aliphatic and aromatic amides with a reactivity conversion of around 70–80%.

Hydrazine reflux is commonly used for reducing SLGO to SLG(R), but titrations show that only around 20–30% of the carboxylic groups are lost, leaving a significant number available for chemical attachment. Analysis of SLG(R) generated by this route reveals that the system is unstable and using a room temperature stirring with HCl (< 1.0 M) leads to around 60% loss of COOH functionality. Room temperature treatment of SLGO with carbodiimides leads to the collapse of the individual sheets into star-like clusters that exhibited poor subsequent reactivity with amines (c. 3–5% conversion of the intermediate to the final amide).[216] It is apparent that conventional chemical treatment of carboxylic groups on SLGO generates morphological changes of individual sheets that leads to a reduction in chemical reactivity, which may potentially limit their use in composite synthesis. Therefore, chemical reactions types have been explored. SLGO has also been grafted with polyallylamine, cross-linked through epoxy groups. When filtered into graphene oxide paper, these composites exhibit increased stiffness and strength relative to unmodified graphene oxide paper.[217]
Full hydrogenation from both sides of graphene sheet results in graphane, but partial hydrogenation leads to hydrogenated graphene.[218] Similarly, both-side fluorination of graphene (or chemical and mechanical exfoliation of graphite fluoride) leads to fluorographene (graphene fluoride),[219] while partial fluorination (generally halogenation) provides fluorinated (halogenated) graphene.
Graphene ligand/complex
Graphene can be a
Graphene fiber
In 2011, researchers reported a novel yet simple approach to fabricate graphene fibers from chemical vapor deposition grown graphene films.[222] The method was scalable and controllable, delivering tunable morphology and pore structure by controlling the evaporation of solvents with suitable surface tension. Flexible all-solid-state supercapacitors based on this graphene fibers were demonstrated in 2013.[223]
In 2015, intercalating small graphene fragments into the gaps formed by larger, coiled graphene sheets, after annealing provided pathways for conduction, while the fragments helped reinforce the fibers.[
In 2016, Kilometer-scale continuous graphene fibers with outstanding mechanical properties and excellent electrical conductivity are produced by high-throughput wet-spinning of graphene oxide liquid crystals followed by graphitization through a full-scale synergetic defect-engineering strategy.[225] The graphene fibers with superior performances promise wide applications in functional textiles, lightweight motors, microelectronic devices, etc.
Tsinghua University in Beijing, led by Wei Fei of the Department of Chemical Engineering, claims to be able to create a carbon nanotube fibre which has a tensile strength of 80 GPa (12,000,000 psi).[226]
3D graphene
In 2013, a three-dimensional honeycomb of hexagonally arranged carbon was termed 3D graphene, and self-supporting 3D graphene was also produced.[227] 3D structures of graphene can be fabricated by using either CVD or solution based methods. A 2016 review by Khurram and Xu et al. provided a summary of then-state-of-the-art techniques for fabrication of the 3D structure of graphene and other related two-dimensional materials.[228] In 2013, researchers at Stony Brook University reported a novel radical-initiated crosslinking method to fabricate porous 3D free-standing architectures of graphene and carbon nanotubes using nanomaterials as building blocks without any polymer matrix as support.[229] These 3D graphene (all-carbon) scaffolds/foams have applications in several fields such as energy storage, filtration, thermal management and biomedical devices and implants.[228][230]
Box-shaped graphene (BSG)
Three dimensional bilayer graphene has also been reported.[232][233]
Pillared graphene
Pillared graphene is a hybrid carbon, structure consisting of an oriented array of carbon nanotubes connected at each end to a sheet of graphene. It was first described theoretically by George Froudakis and colleagues of the University of Crete in Greece in 2008. Pillared graphene has not yet been synthesised in the laboratory, but it has been suggested that it may have useful electronic properties, or as a hydrogen storage material.
Reinforced graphene
Graphene reinforced with embedded carbon nanotube reinforcing bars ("rebar") is easier to manipulate, while improving the electrical and mechanical qualities of both materials.[234][235]
Functionalized single- or multiwalled carbon nanotubes are spin-coated on copper foils and then heated and cooled, using the nanotubes themselves as the carbon source. Under heating, the functional
Stacks of a few layers have been proposed as a cost-effective and physically flexible replacement for
Moulded graphene
In 2015, researchers from the
Graphene aerogel
An aerogel made of graphene layers separated by carbon nanotubes was measured at 0.16 milligrams per cubic centimeter. A solution of graphene and carbon nanotubes in a mold is freeze dried to dehydrate the solution, leaving the aerogel. The material has superior elasticity and absorption. It can recover completely after more than 90% compression, and absorb up to 900 times its weight in oil, at a rate of 68.8 grams per second.[239]
Graphene nanocoil
In 2015, a coiled form of graphene was discovered in graphitic carbon (coal). The spiraling effect is produced by defects in the material's hexagonal grid that causes it to spiral along its edge, mimicking a Riemann surface, with the graphene surface approximately perpendicular to the axis. When voltage is applied to such a coil, current flows around the spiral, producing a magnetic field. The phenomenon applies to spirals with either zigzag or armchair patterns, although with different current distributions. Computer simulations indicated that a conventional spiral inductor of 205 microns in diameter could be matched by a nanocoil just 70 nanometers wide, with a field strength reaching as much as 1 tesla.[240]
The nano-solenoids analyzed through computer models at Rice should be capable of producing powerful magnetic fields of about 1 tesla, about the same as the coils found in typical loudspeakers, according to Yakobson and his team – and about the same field strength as some MRI machines. They found the magnetic field would be strongest in the hollow, nanometer-wide cavity at the spiral's center.[240]
A solenoid made with such a coil behaves as a quantum conductor whose current distribution between the core and exterior varies with applied voltage, resulting in nonlinear inductance.[241]
Crumpled graphene
In 2016,
Production
This section may be too long to read and navigate comfortably. (October 2023) |
A rapidly increasing list of production techniques have been developed to enable graphene's use in commercial applications.[244]
Isolated 2D crystals cannot be grown via chemical synthesis beyond small sizes even in principle, because the rapid growth of phonon density with increasing lateral size forces 2D crystallites to bend into the third dimension. In all cases, graphene must bond to a substrate to retain its two-dimensional shape.[19]
Small graphene structures, such as graphene quantum dots and nanoribbons, can be produced by "bottom up" methods that assemble the lattice from organic molecule monomers (e. g. citric acid, glucose). "Top down" methods, on the other hand, cut bulk graphite and graphene materials with strong chemicals (e. g. mixed acids).
Mechanical
Mechanical exfoliation
Geim and Novoselov initially used adhesive tape to pull graphene sheets away from graphite. Achieving single layers typically requires multiple exfoliation steps. After exfoliation the flakes are deposited on a silicon wafer. Crystallites larger than 1 mm and visible to the naked eye can be obtained.[245]
As of 2014, exfoliation produced graphene with the lowest number of defects and highest electron mobility.[246]
Alternatively a
In 2014 defect-free, unoxidized graphene-containing liquids were made from graphite using mixers that produce local shear rates greater than 10×104.[248][249]
Shear exfoliation is another method which by using rotor-stator mixer the scalable production of the defect-free Graphene has become possible.[250] It has been shown that, as turbulence is not necessary for mechanical exfoliation,[251] low speed ball milling is shown to be effective in the production of High-Yield and water-soluble graphene.
Liquid phase exfoliation
Liquid phase exfoliation (LPE) is a relatively simple method which involves dispersing graphite in a liquid medium to produce graphene by sonication or high shear mixing, followed by centrifugation.[252][253][254] Restacking is an issue with this technique unless solvents with appropriate surface energy are used (e.g. NMP).
Adding a surfactant to a solvent prior to sonication prevents restacking by adsorbing to the graphene's surface.[255] This produces a higher graphene concentration, but removing the surfactant requires chemical treatments.[citation needed]
LPE results in nanosheets with a broad size distribution and thicknesses roughly in the range of 1-10 monolayers. However, liquid cascade centrifugation can be used to size select the suspensions and achieve monolayer enrichment.[256]
Sonicating graphite at the interface of two immiscible liquids, most notably heptane and water, produced macro-scale graphene films. The graphene sheets are adsorbed to the high energy interface between the materials and are kept from restacking. The sheets are up to about 95% transparent and conductive.[257]
With definite cleavage parameters, the box-shaped graphene (BSG) nanostructure can be prepared on graphite crystal.[231]
A major advantage of LPE is that it can be used to exfoliate many inorganic 2D materials beyond graphene, e.g. BN, MoS2, WS2.[258]
Splitting monolayer carbon
Graphene can be created by opening
In 2014, carbon nanotube-reinforced graphene was made via spin coating and annealing functionalized carbon nanotubes.[234]
Another approach sprays buckyballs at supersonic speeds onto a substrate. The balls cracked open upon impact, and the resulting unzipped cages then bond together to form a graphene film.[262]
Chemical
Graphite oxide reduction
P. Boehm reported producing monolayer flakes of reduced graphene oxide in 1962.[263][264] Rapid heating of graphite oxide and exfoliation yields highly dispersed carbon powder with a few percent of graphene flakes.
Another method is reduction of graphite oxide monolayer films, e.g. by
Burning a graphite oxide coated DVD produced a conductive graphene film (1,738 siemens per meter) and specific surface area (1,520 square meters per gram) that was highly resistant and malleable.[266]
A dispersed reduced graphene oxide suspension was synthesized in water by a hydrothermal dehydration method without using any surfactant. The approach is facile, industrially applicable, environmentally friendly and cost effective. Viscosity measurements confirmed that the graphene colloidal suspension (Graphene nanofluid) exhibit Newtonian behavior, with the viscosity showing close resemblance to that of water.[267]
Molten salts
Graphite particles can be corroded in molten salts to form a variety of carbon nanostructures including graphene.[268] Hydrogen cations, dissolved in molten lithium chloride, can be discharged on cathodically polarized graphite rods, which then intercalate, peeling graphene sheets. The graphene nanosheets produced displayed a single-crystalline structure with a lateral size of several hundred nanometers and a high degree of crystallinity and thermal stability.[269]
Electrochemical synthesis
Electrochemical synthesis can exfoliate graphene. Varying a pulsed voltage controls thickness, flake area, number of defects and affects its properties. The process begins by bathing the graphite in a solvent for intercalation. The process can be tracked by monitoring the solution's transparency with an LED and photodiode. [270][271]
Hydrothermal self-assembly
Graphene has been prepared by using a sugar (e.g. glucose, sugar, fructose, etc.) This substrate-free "bottom-up" synthesis is safer, simpler and more environmentally friendly than exfoliation. The method can control thickness, ranging from monolayer to multilayers, which is known as "Tang-Lau Method".[272][273][274][275]
Sodium ethoxide pyrolysis
Gram-quantities were produced by the reaction of ethanol with sodium metal, followed by pyrolysis and washing with water.[276]
Microwave-assisted oxidation
In 2012, microwave energy was reported to directly synthesize graphene in one step.[277] This approach avoids use of potassium permanganate in the reaction mixture. It was also reported that by microwave radiation assistance, graphene oxide with or without holes can be synthesized by controlling microwave time.[278] Microwave heating can dramatically shorten the reaction time from days to seconds.
Graphene can also be made by
Thermal decomposition of silicon carbide
Heating silicon carbide (SiC) to high temperatures (1100 °C) under low pressures (c. 10−6 torr, or 10−4 Pa) reduces it to graphene.[89][90][91][92][93][279]
Chemical vapor deposition
Epitaxy
Epitaxial graphene growth on silicon carbide is wafer-scale technique to produce graphene. Epitaxial graphene may be coupled to surfaces weakly enough (by the active valence electrons that create Van der Waals forces) to retain the two dimensional electronic band structure of isolated graphene.[280]
A normal
The direct synthesis of graphene on insulator TiO2 with high-dielectric-constant (high-κ). A two-step CVD process is shown to grow graphene directly on TiO2 crystals or exfoliated TiO2 nanosheets without using any metal catalyst.[283]
Metal substrates
CVD graphene can be grown on metal substrates including ruthenium,[284] iridium,[285] nickel[286] and copper.[287][288]
Roll-to-roll
In 2014, a two-step roll-to-roll manufacturing process was announced. The first roll-to-roll step produces the graphene via chemical vapor deposition. The second step binds the graphene to a substrate.[289][290]

Cold wall
Growing graphene in an industrial resistive-heating cold wall CVD system was claimed to produce graphene 100 times faster than conventional CVD systems, cut costs by 99% and produce material with enhanced electronic qualities.[291][292]
Wafer scale CVD graphene
CVD graphene is scalable and has been grown on deposited Cu thin film catalyst on 100 to 300 mm standard Si/SiO2 wafers[293][294][295] on an Axitron Black Magic system. Monolayer graphene coverage of >95% is achieved on 100 to 300 mm wafer substrates with negligible defects, confirmed by extensive Raman mapping.[294][295]
Solvent interface trapping method (SITM)
Reported by a group led by D. H. Adamson, graphene can be produced from natural graphite while preserving the integrity of the sheets using solvent interface trapping method (SITM). SITM use a high energy interface, such as oil and water, to exfoliate graphite to graphene. Stacked graphite delaminates, or spreads, at the oil/water interface to produce few-layer graphene in a thermodynamically favorable process in much the same way as small molecule surfactants spread to minimize the interfacial energy. In this way, graphene behaves like a 2D surfactant.[296][297][298] SITM has been reported for a variety of applications such conductive polymer-graphene foams,[299][300][301][302] conductive polymer-graphene microspheres,[303] conductive thin films[304] and conductive inks.[305]
Carbon dioxide reduction
A highly exothermic reaction combusts magnesium in an oxidation–reduction reaction with carbon dioxide, producing carbon nanoparticles including graphene and fullerenes.[306]
Supersonic spray
Supersonic acceleration of droplets through a
Laser
In 2014, a CO
2
Flash Joule heating
In 2019, flash Joule heating (transient high-temperature electrothermal heating) was discovered to be a method to synthesize turbostratic graphene in bulk powder form. The method involves electrothermally converting various carbon sources, such as carbon black, coal, and food waste into micron-scale flakes of graphene. The graphenization process is kinetically controlled, and the energy dose is chosen to preserve the carbon in its graphenic state (excessive energy input leads to subsequent graphitization through annealing).
Ion implantation
Accelerating carbon ions inside an electrical field into a semiconductor made of thin nickel films on a substrate of SiO2/Si, creates a wafer-scale (4 inches (100 mm)) wrinkle/tear/residue-free graphene layer at a relatively low temperature of 500 °C.[315][316]
CMOS-compatible graphene
Integration of graphene in the widely employed CMOS fabrication process demands its transfer-free direct synthesis on dielectric substrates at temperatures below 500 °C. At the IEDM 2018, researchers from University of California, Santa Barbara, demonstrated a novel CMOS-compatible graphene synthesis process at 300 °C suitable for back-end-of-line (BEOL) applications.[317][318][319] The process involves pressure-assisted solid-state diffusion of carbon through a thin-film of metal catalyst. The synthesized large-area graphene films were shown to exhibit high-quality (via Raman characterization) and similar resistivity values when compared with high-temperature CVD synthesized graphene films of same cross-section down to widths of 20 nm.
Simulation
In addition to experimental investigation of graphene and graphene-based devices, their numerical modeling and simulation have been an important research topic. The Kubo formula provides an analytic expression for the graphene's conductivity and shows that it is a function of several physical parameters including wavelength, temperature, and chemical potential.[320] Moreover, a surface conductivity model, which describes graphene as an infinitesimally thin (two sided) sheet with a local and isotropic conductivity, has been proposed. This model permits derivation of analytical expressions for the electromagnetic field in the presence of a graphene sheet in terms of a dyadic Green function (represented using Sommerfeld integrals) and exciting electric current.[321] Even though these analytical models and methods can provide results for several canonical problems for benchmarking purposes, many practical problems involving graphene, such as design of arbitrarily shaped electromagnetic devices, are analytically intractable. With the recent advances in the field of computational electromagnetics (CEM), various accurate and efficient numerical methods have become available for analysis of electromagnetic field/wave interactions on graphene sheets and/or graphene-based devices. A comprehensive summary of computational tools developed for analyzing graphene-based devices/systems is proposed.[322]
Graphene analogs
Graphene analogs[323] (also referred to as "artificial graphene") are two-dimensional systems which exhibit similar properties to graphene. Graphene analogs are studied intensively since the discovery of graphene in 2004. People try to develop systems in which the physics is easier to observe and to manipulate than in graphene. In those systems, electrons are not always the particles which are used. They might be optical photons,[324] microwave photons,[325] plasmons,[326] microcavity polaritons,[327] or even atoms.[328] Also, the honeycomb structure in which those particles evolve can be of a different nature than carbon atoms in graphene. It can be, respectively, a photonic crystal, an array of metallic rods, metallic nanoparticles, a lattice of coupled microcavities, or an optical lattice.
Applications
Graphene is a transparent and flexible conductor that holds great promise for various material/device applications, including solar cells,[329] light-emitting diodes (LED), integrated photonic circuit devices,[330][331] touch panels, and smart windows or phones.[332] Smartphone products with graphene touch screens are already on the market.[333]
In 2013, Head announced their new range of graphene tennis racquets.[334]
As of 2015, there is one product available for commercial use: a graphene-infused printer powder.
On August 2, 2016, BAC's new Mono model is said to be made out of graphene as a first of both a street-legal track car and a production car.[340]
In January 2018, graphene based spiral inductors exploiting kinetic inductance at room temperature were first demonstrated at the University of California, Santa Barbara, led by Kaustav Banerjee. These inductors were predicted to allow significant miniaturization in radio-frequency integrated circuit applications.[341][342][343]
The potential of epitaxial graphene on
Novel uses for graphene continue to be researched and explored. One such use is in combination with water-based epoxy resins to produce anticorrosive coatings.[345] The van der Waals nature of graphene and other two-dimensional (2D) materials also permits van der Waals heterostructures[346] and integrated circuits based on van der Waals integration of 2D materials.[347][348]
Toxicity
One review on graphene toxicity published in 2016 by Lalwani et al. summarizes the in vitro, in vivo, antimicrobial and environmental effects and highlights the various mechanisms of graphene toxicity.[349] Another review published in 2016 by Ou et al. focused on graphene-family nanomaterials (GFNs) and revealed several typical mechanisms such as physical destruction, oxidative stress, DNA damage, inflammatory response, apoptosis, autophagy, and necrosis.[350]
A 2020 study showed that the toxicity of graphene is dependent on several factors such as shape, size, purity, post-production processing steps, oxidative state, functional groups, dispersion state, synthesis methods, route and dose of administration, and exposure times.[351]
In 2014, research at Stony Brook University showed that graphene nanoribbons, graphene nanoplatelets and graphene nano–onions are non-toxic at concentrations up to 50 μg/ml. These nanoparticles do not alter the differentiation of human bone marrow stem cells towards osteoblasts (bone) or adipocytes (fat) suggesting that at low doses graphene nanoparticles are safe for biomedical applications.[352] In 2013 research at Brown University found that 10 μm few-layered graphene flakes are able to pierce cell membranes in solution. They were observed to enter initially via sharp and jagged points, allowing graphene to be internalized in the cell. The physiological effects of this remain unknown, and this remains a relatively unexplored field.[353][354]
See also
- Borophene – Allotrope of boron
- Carbon fiber– Light, strong and rigid composite material
- Penta-graphene – allotrope of carbon
- Phagraphene
- Plumbene – Material made up of a single layer of lead atoms
- Silicene – Two-dimensional allotrope of silicon
References
- ^ "graphene definition, meaning – what is graphene in the British English Dictionary & Thesaurus – Cambridge Dictionaries Online". cambridge.org.
- ^ S2CID 14647602.
- .
- ^ a b [1] "Carbon nanostructures for electromagnetic shielding applications", Mohammed Arif Poothanari, Sabu Thomas, et al., Industrial Applications of Nanomaterials, 2019. "Carbon nanostructures include various low-dimensional allotropes of carbon including carbon black (CB), carbon fiber, carbon nanotubes (CNTs), fullerene, and graphene."
- .
- ^ doi:10.3390/c4010004.
- S2CID 55246344.
- ^ S2CID 3024573.
- ^ S2CID 73626659.
- ^ S2CID 206512830.
- ^ PMID 31941941.
- .
- ^ S2CID 5729649.
- ^ APS News. Series II. 18 (9): 2. 2009.
- ^ "The Nobel Prize in Physics 2010". Nobel Foundation. Retrieved 1 September 2021.
- ^ "Global Demand for Graphene after Commercial Production to be Enormous, says Report". AZONANO.com. 28 February 2014. Retrieved 24 July 2014.
- ^ Mrmak, Nebojsa (28 November 2014). "Graphene properties (A Complete Reference)". Graphene-Battery.net. Retrieved 10 November 2019.
- ISBN 978-0-9678550-9-7. Retrieved 31 March 2012.
- ^ S2CID 206513254.
- S2CID 33832203.
- .
- JSTOR 108699.
- ^ Debije, P; Scherrer, P (1916). "Interferenz an regellos orientierten Teilchen im Röntgenlicht I". Physikalische Zeitschrift (in German). 17: 277.
- ^ Friedrich, W (1913). "Eine neue Interferenzerscheinung bei Röntgenstrahlen" [A new interference phenomenon in X-rays]. Physikalische Zeitschrift (in German). 14: 317–319.
- .
- .
- JSTOR 94336.
- S2CID 121157442.
- ^ .
- .
- ^ S2CID 3470761.
- ^ S2CID 37267733.
- ^ S2CID 4424714.
- .
- ^ S2CID 3507167.
- ^ Boehm, H. P.; Clauss, A.; Fischer, G.; Hofmann, U. (1962). "Surface Properties of Extremely Thin Graphite Lamellae" (PDF). Proceedings of the Fifth Conference on Carbon. Pergamon Press. Archived from the original (PDF) on 13 April 2016. Retrieved 1 April 2016.
- S2CID 250758301.
- .
- ^ Mouras, S.; et al. (1987). "Synthesis of first stage graphite intercalation compounds with fluorides". Revue de Chimie Minérale. 24: 572. INIST 7578318.
- PMID 10003828.
- .
- ^ Geim, A. K.; Kim, P. (April 2008). "Carbon Wonderland". Scientific American.
... bits of graphene are undoubtedly present in every pencil mark
- ^ Robert B. Rutherford and Richard L. Dudman (2002): "Ultra-thin flexible expanded graphite heating element". US Patent 6667100. Filed on 2002-05-13, granted on 2003-12-23, assigned to EGC Operating Co LLC; expired.
- ^ Bor Z. Jang and Wen C. Huang (2002): "Nano-scaled graphene plates". US Patent 7071258. Filed on 2002-10-21, granted on 2006-07-04, assigned to Global Graphene Group Inc; to expire on 2024-01-06.
- ^ "Graphene edges closer to widespread production and application". www.compositesworld.com. 10 August 2016. Retrieved 25 March 2022.
- ^ a b "The Story of Graphene". www.graphene.manchester.ac.uk. The University of Manchester. 10 September 2014. Retrieved 9 October 2014.
Following discussions with colleagues, Andre and Kostya adopted a method that researchers in surface science were using – using simple Sellotape to peel away layers of graphite to expose a clean surface for study under the microscope.
- ^ "Graphene pioneers bag Nobel prize". Institute of Physics, UK. 5 October 2010. Archived from the original on 8 October 2010. Retrieved 5 October 2010.
- The Nobel Foundation. Retrieved 3 December 2013.
- ^ "New £60m Engineering Innovation Centre to be based in Manchester". The University of Manchester. 10 September 2014.
- ^ Burn-Callander, Rebecca (1 July 2014). "Graphene maker aims to build British, billion-pound venture". Daily Telegraph. Archived from the original on 11 January 2022. Retrieved 24 July 2014.
- ^ Gibson, Robert (10 June 2014). "Consett firm Thomas Swan sees export success with grapheme". The Journal. Archived from the original on 12 July 2014. Retrieved 23 July 2014.
- ^ "Global breakthrough: Irish scientists discover how to mass produce 'wonder material' graphene". The Journal.ie. 20 April 2014. Retrieved 20 December 2014.
- ^ "Cambridge Nanosystems opens new factory for commercial graphene production". Cambridge News. Archived from the original on 23 September 2015.
- ^ S2CID 78304205.
- ^ Felix, I. M. (2013). "Estudo da estrutura eletrônica do grafeno e grafeno hidratado" [Study of the electronic structure of graphene and hydrated graphene] (in Portuguese).
{{cite journal}}: Cite journal requires|journal=(help) - S2CID 196975315.
- .
- doi:10.1086/178105.
- S2CID 15066112.
- ^ PMID 17972931.
- ^ S2CID 38264967.
- ^ S2CID 13087073.
- S2CID 214615777.
- ^ S2CID 5650871. Archived from the original(PDF) on 15 November 2010.
- ^ a b c d Charlier, J.-C.; Eklund, P.C.; Zhu, J.; Ferrari, A.C. (2008). Jorio, A.; Dresselhaus, G.; Dresselhaus, M.S. (eds.). Electron and Phonon Properties of Graphene: Their Relationship with Carbon Nanotubes. Berlin/Heidelberg: Springer-Verlag.
{{cite book}}:|work=ignored (help) - S2CID 26968734.
- S2CID 17130602.
- S2CID 53633968.
- ^ PMID 18654384.
- S2CID 119213419.
- S2CID 3543049.
- ^ S2CID 12221376.
- .
- ^ Physicists Show Electrons Can Travel More Than 100 Times Faster in Graphene :: University Communications Newsdesk, University of Maryland Archived 19 September 2013 at the Wayback Machine. Newsdesk.umd.edu (24 March 2008). Retrieved on 2014-01-12.
- S2CID 24846431.
- ^ "Graphene Devices Stand the Test of Time". 22 January 2015.
- ^ "Researchers create superconducting graphene". 9 September 2015. Retrieved 22 September 2015.
- ^ a b "New form of graphene allows electrons to behave like photons". kurzweilai.net.
- S2CID 4445858.
- ^ S2CID 53419753.
- ^ Light pulses control how graphene conducts electricity. kurzweilai.net. 4 August 2014
- ^ S2CID 3518448.
- PMID 18003926.
- S2CID 14581125.
- S2CID 28441144.
- S2CID 8825103.(PDF) on 15 November 2010. Retrieved 2 September 2009.
Franz, M. (5 January 2008). "Fractionalization of charge and statistics in graphene and related structures" (PDF). University of British Columbia. Archived from the original - S2CID 118585778.
- S2CID 4349731.
- ^ S2CID 118710923.
- ^ S2CID 9546283.
- ^ S2CID 118422866.
- ^ Bibcode:2009arXiv0909.1193L.
- ^ S2CID 118388086.
- S2CID 203913300.
- ^ S2CID 16445720.
- ^ S2CID 2927627.
- ^ S2CID 4392125.
- S2CID 118398377.
- S2CID 118473227.
- S2CID 31092090.
- ^ S2CID 26403885.
- )
- S2CID 17595181.
- ^ "Graphene Gazing Gives Glimpse Of Foundations Of Universe". ScienceDaily. 4 April 2008.
- .
- hdl:10220/17639.
- S2CID 205217165.
- .
- ^ .
- PMID 23071901.
- S2CID 59070301.
- S2CID 207313024.
- S2CID 119284608.
- S2CID 119233725.
- .
- PMID 23188285.
- S2CID 119237334.
- S2CID 118338133.
- hdl:10261/98472.
- S2CID 36067301.
- PMID 18999843.
- .
- S2CID 31835103.
- S2CID 119254414.
- PMID 21939213.
- ^ S2CID 4411466.
- ^ S2CID 119145153.
- S2CID 119608880.
- ^ Hashimoto, T.; Kamikawa, S.; Yagi, Y.; Haruyama, J.; Yang, H.; Chshiev, M. (2014). "Graphene edge spins: spintronics and magnetism in graphene nanomeshes" (PDF). Nanosystems: Physics, Chemistry, Mathematics. 5 (1): 25–38.
- PMID 37045919.
- ^ T. Hashimoto, S. Kamikawa, Y. Yagi, J. Haruyama, H. Yang, M. Chshiev, "Graphene edge spins: spintronics and magnetism in graphene nanomeshes", February 2014, Volume 5, Issue 1, pp 25
- ^ Coxworth, Ben (27 January 2015). "Scientists give graphene one more quality – magnetism". Gizmag. Retrieved 6 October 2016.
- S2CID 9006722.
- ^ S2CID 9310741.
- ISBN 978-0-306-67020-6.
- S2CID 207664146.
- S2CID 207558462.
- S2CID 10617464.
- S2CID 118664500.
- S2CID 213783.
- S2CID 115849714.
- S2CID 45253497.
- PMID 21314164.
- Advantage Business Media. scientificcomputing.com.
- .
- PMID 21384860.
- ISBN 978-90-5699-228-6.
- ^ PMID 16197233.
- S2CID 119461729.
- ^ Lifshitz, I.M. (1952). Journal of Experimental and Theoretical Physics (in Russian). Vol. 22. p. 475.
- ^ Class for Physics of the Royal Swedish Academy of Sciences (5 October 2010). "Scientific Background on the Nobel Prize in Physics 2010 GRAPHENE" (PDF). Nobel Prize. Archived from the original (PDF) on 1 July 2018.
- .
- .
- .
- S2CID 119223424.
- S2CID 118596336.
- S2CID 19738771.
- S2CID 9501340. Archived from the original(PDF) on 27 October 2020. Retrieved 7 January 2020.
- PMID 24777167.
- ^ Dorrieron, Jason (4 December 2014). "Graphene Armor Would Be Light, Flexible and Far Stronger Than Steel". Singularity Hub. Retrieved 6 October 2016.
- ^ Coxworth, Ben (1 December 2014). "Graphene could find use in lightweight ballistic body armor". Gizmag. Retrieved 6 October 2016.
- .
- .
- S2CID 12301209.
- PMID 22751178.
- S2CID 35277622.
- .
- ^ S2CID 135899138.
- S2CID 286118.
- ^ S2CID 118840850.
- ^ S2CID 118835225.
- ^ PMID 23528068.
- ^ PMID 25103818.
- S2CID 6655234.
- .
- S2CID 12628957.
- .
- S2CID 96586004.
- .
- ^ "Thinnest graphene sheets react strongly with hydrogen atoms; thicker sheets are relatively unaffected". Phys.org. 1 February 2013.
- S2CID 11008306.
- S2CID 6655234.
- .
- PMID 21650218.
- PMID 26700626.
- ^ "Graphene shown to safely interact with neurons in the brain". University of Cambridge. 29 January 2016. Retrieved 16 February 2016.
- S2CID 20794090.
- S2CID 55035225.
- S2CID 207573621.
- S2CID 93345920.
- ^ "Polish scientists find way to make super-strong graphene sheets | Graphene-Info". www.graphene-info.com. Retrieved 1 July 2015.
- S2CID 235321882.
- S2CID 119443126.
- S2CID 33249360.
- ^ S2CID 896422.
- S2CID 219011027.
- ^ S2CID 210926149.
- S2CID 221623214.
- .
- PMID 23353677.
- PMID 29426893.
- S2CID 210116531.
- S2CID 245712349.
- ^ Félix, Isaac de Macêdo (29 March 2016). Transporte térmico em nanofitas de grafeno-nitreto de boro (masterThesis). Brasil.
- ^ Félix, Isaac de Macêdo (4 August 2020). Condução de calor em nanofitas quase-periódicas de grafeno-hBN (doctoralThesis) (in Brazilian Portuguese). Universidade Federal do Rio Grande do Norte.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ S2CID 9055313.
- ^ PMID 24848545.
- S2CID 96410135.
- PMID 20731455.
- ^ "Graphene Oxide Paper". Northwestern University. Archived from the original on 2 June 2016. Retrieved 28 February 2011.
- .
- S2CID 24385952.
- S2CID 15204080.
- S2CID 248823106.
- PMID 16771469.
- .
- S2CID 55033112.
- S2CID 3536592.
- S2CID 98682200.
- .
- .
- PMID 21875131.
- S2CID 22426420.
- PMID 26339027.
- S2CID 31988847.
- S2CID 46890587.
- hdl:2027.42/99684. Retrieved 24 August 2013.
- ^ PMID 27459895.
- PMID 23436939.
- PMID 25788440.
- ^ S2CID 119369379.
- .
- S2CID 12995375.
- ^ a b c d "Carbon nanotubes as reinforcing bars to strengthen graphene and increase conductivity". Kurzweil Library. 9 April 2014. Retrieved 23 April 2014.
- PMID 24694285.
- ^ "Robust new process forms 3D shapes from flat sheets of graphene". grainger.illinois.edu. 23 June 2015. Archived from the original on 12 May 2020. Retrieved 31 May 2020.
- ^ Jeffrey, Colin (28 June 2015). "Graphene takes on a new dimension". New Atlas. Retrieved 10 November 2019.
- ^ "How to form 3-D shapes from flat sheets of graphene". Kurzweil Library. 30 June 2015. Retrieved 10 November 2019.
- ^ Anthony, Sebastian (10 April 2013). "Graphene aerogel is seven times lighter than air, can balance on a blade of grass - Slideshow | ExtremeTech". ExtremeTech. Retrieved 11 October 2015.
- ^ a b "Graphene nano-coils discovered to be powerful natural electromagnets". Kurzweil Library. 16 October 2015. Retrieved 10 November 2019.
- PMID 26452145.
- ^ Stacey, Kevin (21 March 2016). "Wrinkles and crumples make graphene better | News from Brown". news.brown.edu. Brown University. Archived from the original on 8 April 2016. Retrieved 23 June 2016.
- S2CID 19544549.
- hdl:2262/91730.
- S2CID 123480416.
- arXiv:1406.0809 [cond-mat.mtrl-sci].
- PMID 21711598.
- ^ "A new method of producing large volumes of high-quality graphene". KurzweilAI. 2 May 2014. Retrieved 3 August 2014.
- S2CID 43256835.
- S2CID 216233251.
- S2CID 43256835.
- .
- S2CID 205443620.
- S2CID 27531863.
- S2CID 16624132.
- S2CID 189813507.
- S2CID 27816586.
- S2CID 23576676.
- .
- S2CID 2920478.
- S2CID 205216466.
- ^ "How to Make Graphene Using Supersonic Buckyballs | MIT Technology Review". MIT Technology Review. 13 August 2015. Retrieved 11 October 2015.
- ^ "Boehm's 1961 isolation of graphene". Graphene Times. 7 December 2009. Archived from the original on 8 October 2010.
- ^ Geim, Andre (January 2010). "Many Pioneers in Graphene Discovery". Letters to the Editor. American Physical Society. Retrieved 10 November 2019.
- S2CID 26172029.
- S2CID 18958488.on 16 June 2013. Retrieved 20 March 2012.
Marcus, Jennifer (15 March 2012). "Researchers develop graphene supercapacitor holding promise for portable electronics / UCLA Newsroom". Newsroom.ucla.edu. Archived from the original - S2CID 53349683.
- .
- PMID 26053881.
- ^ "How to tune graphene properties by introducing defects | KurzweilAI". www.kurzweilai.net. 30 July 2015. Retrieved 11 October 2015.
- S2CID 206072084.
- hdl:10397/15682.
- S2CID 137213724.
- S2CID 205874900.
- S2CID 23688312.
- PMID 19119279.
- S2CID 11991071.
- S2CID 14567874.
- PMID 19229263.
- .
- ^ "Samsung's graphene breakthrough could finally put the wonder material into real-world devices". ExtremeTech. 7 April 2014. Retrieved 13 April 2014.
- S2CID 206556123.
- .
- ^ "A smarter way to grow graphene". PhysOrg.com. May 2008.
- S2CID 43507175.
- ^ "New process could lead to more widespread use of graphene". Gizmag.com. 28 May 2014. Retrieved 14 June 2014.
- .
- S2CID 213144.
- ^ Martin, Steve (18 September 2014). "Purdue-based startup scales up graphene production, develops biosensors and supercapacitors". Purdue University. Retrieved 4 October 2014.
- ^ "Startup scales up graphene production, develops biosensors and supercapacitors". R&D Magazine. 19 September 2014. Retrieved 4 October 2014.
- ^ Quick, Darren (26 June 2015). "New process could usher in "graphene-driven industrial revolution"". www.gizmag.com. Retrieved 5 October 2015.
- PMID 26053564.
- S2CID 30130350.
- ^ S2CID 55726071.
- ^ S2CID 5077855.
- ISSN 0008-6223.
- OSTI 1265313.
- S2CID 225385130.
- S2CID 102395615.
- S2CID 205274548.
- S2CID 219316507.
- S2CID 220798190.
- S2CID 204883163.
- PMID 23879536.
- PMID 33093555.
- S2CID 96850993.
- S2CID 96283118.
- S2CID 96283118. Retrieved 14 June 2014.
- PMID 25493446.
- ISSN 0008-6223.
- S2CID 221623214.
- S2CID 226203667.
- S2CID 232864412.
- S2CID 233573678.
- ^ "Korean researchers grow wafer-scale graphene on a silicon substrate | KurzweilAI". www.kurzweilai.net. 21 July 2015. Retrieved 11 October 2015.
- .
- S2CID 116643404.
- S2CID 58675631.
- ^ "Graphene goes mainstream". The Current, UC Santa Barbara. 23 July 2019.
- S2CID 119638159.
- S2CID 32535262.
- S2CID 216262889.
- PMID 24002076.
- S2CID 26962706.
- S2CID 8335461.
- .
- S2CID 31526933.
- S2CID 118519844.
- .
- S2CID 117786282.
- S2CID 25707442.
- S2CID 26541191.
- S2CID 203653990.
- ^ "Racquet Review: Head Graphene XT Speed Pro". Tennis.com. Retrieved 15 October 2016.
- ^ "GRAPHENITE – GRAPHENE INFUSED 3D PRINTER POWDER – 30 Lbs – $499.95". noble3dprinters.com. Noble3DPrinters. Retrieved 16 July 2015.[permanent dead link]
- ^ "Graphene Uses & Applications". Graphenea. Retrieved 13 April 2014.
- PMID 23405887.
- S2CID 18266151.
- ^ "Applied Graphene Materials plc :: Graphene dispersions". appliedgraphenematerials.com. Archived from the original on 27 May 2014. Retrieved 26 May 2014.
- ^ "BAC Debuts First Ever Graphene Constructed Vehicle". 2 August 2016. Retrieved 4 August 2016.
- S2CID 139420526.
- ^ Siegel, E. (2018). "The Last Barrier to Ultra-Miniaturized Electronics is Broken, Thanks To A New Type Of Inductor". Forbes.com.
- ^ "Engineers reinvent the inductor after two centuries". physicsworld. 2018.
- S2CID 203653976.
- S2CID 139956928.
- S2CID 251175507.
- S2CID 258279195.
- S2CID 256768556.
- PMID 27154267.
- PMID 27799056.
- PMID 32523022.
- PMID 24674462.
- ^ Stacey, Kevin (10 July 2013). "Jagged graphene edges can slice and dice cell membranes - News from Brown". brown.edu.
- PMID 23840061.
External links
- Manchester's Revolutionary 2D Material at The University of Manchester
- Graphene at The Periodic Table of Videos(University of Nottingham)
- Graphene: Patent surge reveals global race
- 'Engineering Controls for Nano-scale Graphene Platelets During Manufacturing and Handling Processes' (PDF)
- Band structure of graphene (PDF).
















