Prussian blue

| |
| Names | |
|---|---|
| IUPAC name
Iron(II,III) hexacyanidoferrate(II,III)
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.034.418 |
| EC Number |
|
| 1093743 | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18Fe7N18 | |
| Molar mass | 859.239 g·mol−1 |
| Appearance | Blue opaque crystals |
| Insoluble | |
| Structure | |
| Face-centered cubic, cF43 | |
| Fm3m, No. 225[1] | |
| Pharmacology | |
| V03AB31 (WHO) | |
| Oral | |
| Hazards | |
| Safety data sheet (SDS) | MSDS Prussian blue |
| Related compounds | |
Other cations
|
Potassium ferrocyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Prussian blue (also known as Berlin blue, Brandenburg blue, Parisian and Paris blue) is a dark blue pigment produced by oxidation of ferrous ferrocyanide salts. It has the chemical formula FeIII
4[FeII
(CN)
6]
3. Turnbull's blue is chemically identical, but is made from different reagents, and its slightly different color stems from different impurities and particle sizes.
Prussian blue was created in the early 18th century and is the first modern
In medicine, orally administered Prussian blue is used as an
Prussian blue lent its name to
History

Prussian blue pigment is significant since it was the first stable and relatively
Prussian blue
The pigment readily replaced the expensive lapis lazuli derived ultramarine and was an important topic in the letters exchanged between Johann Leonhard Frisch and the president of the Prussian Academy of Sciences, Gottfried Wilhelm Leibniz, between 1708 and 1716.[8] It is first mentioned in a letter written by Frisch to Leibniz, from March 31, 1708. Not later than 1708, Frisch began to promote and sell the pigment across Europe. By August 1709, the pigment had been termed Preussisch blau; by November 1709, the German name Berlinisch Blau had been used for the first time by Frisch. Frisch himself is the author of the first known publication of Prussian blue in the paper Notitia Coerulei Berolinensis nuper inventi in 1710, as can be deduced from his letters. Diesbach had been working for Frisch since about 1701.

To date, the Entombment of Christ, dated 1709 by
In 1731, Georg Ernst Stahl published an account of the first synthesis of Prussian blue.[13] The story involves not only Diesbach, but also Johann Konrad Dippel. Diesbach was attempting to create a red lake pigment from cochineal, but obtained the blue instead as a result of the contaminated potash he was using. He borrowed the potash from Dippel, who had used it to produce his animal oil. No other known historical source mentions Dippel in this context. It is, therefore, difficult to judge the reliability of this story today. In 1724, the recipe was finally published by John Woodward.[14][15][16]
In 1752, French chemist
In the late 1800s,
Military symbol
From the beginning of the 18th century, Prussian blue was the predominant uniform coat color worn by the infantry and artillery regiments of the Prussian Army.[21] As Dunkelblau (dark blue), this shade achieved a symbolic importance and continued to be worn by most German soldiers for ceremonial and off-duty occasions until the outbreak of World War I, when it was superseded by greenish-gray field gray (Feldgrau).[22]
Synthesis
Prussian blue is produced by oxidation of ferrous ferrocyanide salts. These white solids have the formula M
2Fe[Fe(CN)
6] where M+
= Na+
or K+
. The iron in this material is all ferrous, hence the absence of deep color associated with the mixed valency. Oxidation of this white solid with hydrogen peroxide or sodium chlorate produces ferricyanide and affords Prussian blue.[23]
A "soluble" form, KFeIII[FeII(CN)
6], which is really colloidal, can be made from potassium ferrocyanide and iron(III):
- K+
+ Fe3+
+ [FeII(CN)
6]4−
→ KFeIII[FeII(CN)
6]
The similar reaction of potassium ferricyanide and iron(II) results in the same colloidal solution, because [FeIII(CN)
6]3−
is converted into ferrocyanide.
The "insoluble" Prussian blue is obtained if, in the reactions above, an excess of Fe3+
is added:
- 4Fe3+
+ 3[FeII(CN)
6]4−
→ FeIII[FeIIIFeII(CN)
6]
3 [24]
Despite the fact that it is prepared from cyanide salts, Prussian blue is not toxic because the cyanide groups are tightly bound to iron.[25] Both ferrocyanide ((FeII(CN)6)4−) and ferricyanide ((FeIII(CN)6)3−) are particularly stable and non-toxic polymeric cyanometalates due to the strong iron coordination to cyanide ions. Although cyanide bonds well with transition metals in general like chromium, these non-iron coordination compounds are not as stable as iron cyanides, therefore increasing the risk of releasing CN− ions, and subsequently comparative toxicity.[26]
Turnbull's blue
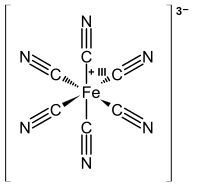
In former times, the addition of iron(II) salts to a solution of
Prussian white
Prussian white, also known as Berlin white or Everett's salt, is the
A more generic formula allowing for the substitution of Na+ cations by K+ cations is A(2−x)BxFe2(CN)6 (in which A or B = Na+ or K+). The Prussian white is closely related to the Prussian blue, but it significantly differs by its crystallographic structure, molecular framework pore size, and its color. The cubic sodium Prussian white, Na(2−x)KxFe2(CN)6·yH2O, and potassium Prussian white, K(2−x)NaxFe2(CN)6·yH2O, are candidates as cathode materials for Na-ion batteries.[30] The insertion of Na+ and K+ cations in the framework of potassium Prussian white provides favorable synergistic effects improving the long-term battery stability and increasing the number of possible recharge cycles, lengthening so its service life.[30] The large-size framework of Prussian white easily accommodating Na+ and K+ cations facilitates their intercalation and subsequent extraction during the charge/discharge cycles. The spacious and rigid host crystal structure contributes to its volumetric stability against the internal swelling stress and strain developing in sodium-batteries after many cycles.[29] The material also offers perspectives of high energy densities (Ah/kg) while providing high recharge rate, even at low temperature.[29]
Properties
Prussian blue is a microcrystalline blue powder. It is insoluble, but the crystallites tend to form a colloid. Such colloids can pass through fine filters.[2] Despite being one of the oldest known synthetic compounds, the composition of Prussian blue remained uncertain for many years. Its precise identification was complicated by three factors:
- Prussian blue is extremely insoluble, but also tends to form colloids
- Traditional syntheses tend to afford impure compositions
- Even pure Prussian blue is structurally complex, defying routine crystallographic analysis
Crystal structure


6 groups shown, at random, will be missing, giving on average only 18 cyanide ions (rather than the 24 shown) and three ferrous iron atoms.

The
PB has a
ions; insoluble PB has interstitial water, instead. In ideal insoluble PB crystals, the cubic framework is built from Fe(II)–C–N–Fe(III) sequences, with Fe(II)–carbon distances of 1.92 Å and Fe(III)–nitrogen distances of 2.03 Å. One-fourth of the sites of Fe(CN)
6 subunits (supposedly at random) are vacant (empty), leaving three such groups on average per unit cell.[42] The empty nitrogen sites are filled with water molecules instead, which are coordinated to Fe(III).

6 groups shown will be missing. This illustration superimposes both possibilities at each site — water molecules or cyanide ions.
The Fe(II) centers, which are
The composition is notoriously variable due to the presence of lattice defects, allowing it to be hydrated to various degrees as water molecules are incorporated into the structure to occupy
Color
Prussian blue is strongly colored and tends towards black and dark blue when mixed into
Like most high-chroma pigments, Prussian blue cannot be accurately displayed on a computer display. PB is electrochromic—changing from blue to colorless upon reduction. This change is caused by reduction of the Fe(III) to Fe(II), eliminating the intervalence charge transfer that causes Prussian blue's color.
Use
Pigment

Because it is easily made, cheap, nontoxic, and intensely colored, Prussian blue has attracted many applications. It was adopted as a pigment very soon after its invention and was almost immediately widely used in oil paints, watercolor, and dyeing.
Nanoparticles of Prussian blue are used as pigments in some cosmetics ingredients, according to the European Union Observatory for Nanomaterials.
| Prussian blue | ||
|---|---|---|
| (19, 30, 247°) | ||
| Source | [1] | |
| B: Normalized to [0–255] (byte) H: Normalized to [0–100] (hundred) | ||
Medicine
Prussian blue's ability to incorporate
Cs+
from the organism of those poisoned in the Goiânia accident in Brazil.[2]
Stain for iron
Prussian blue is a common
-
Histopathology of the liver, showing a Kupffer cells with significant hemosiderin deposition next to a hepatocyte with lipofuscin pigment. H&E stain.
-
Prussian blue staining, highlighting the hemosiderin pigment as blue.
-
Prussian blue stain
By machinists and toolmakers
In analytical chemistry
Prussian blue is formed in the Prussian blue assay for total phenols. Samples and phenolic standards are given acidic ferric chloride and ferricyanide, which is reduced to ferrocyanide by the phenols. The ferric chloride and ferrocyanide react to form Prussian blue. Comparing the absorbance at 700 nm of the samples to the standards allows for the determination of total phenols or polyphenols.[52][53]
Household use
Prussian blue is present in some preparations of
Research
Battery materials
Prussian blue (PB) has been studied for its applications in electrochemical energy storage since 1978.[55] Prussian Blue proper (the Fe-Fe solid) shows two well-defined reversible redox transitions in K+ solutions. Weakly solvated potassium ions (as well as Rb+ and Cs+, not shown) have the solvated radius, which fits the framework of Prussian Blue. On the other hand, the sizes of solvated Na+ and Li+ are too large for the PB cavity, and the intercalation of these ions is hindered and much slower. The low and high voltage sets of peaks in the cyclic voltammetry correspond to 1 and ⅔ electron per Fe atom, respectively.[56] The high voltage set is due to the Fe3+/Fe2+ transition at the low-spin Fe ions coordinated to C-atoms. The low-voltage set is due to high-spin Fe ion coordinated to N-atoms.[57][58][59]
It is possible to replace the Fe metal centers in PB with other metal ions such as Mn, Co, Ni, Zn, etc. to form electrochemically active Prussian blue analogues (PBAs). PB/PBAs and their derivatives have also been evaluated as electrode materials for reversible alkali-ion insertion and extraction in lithium-ion battery, sodium-ion battery, and potassium-ion battery.
See also
- Blue billy – Prussian blue deposit formed in soils contaminated by effluents of chemical industry
- Blue pigments
- Cobalt blue – Blue pigment
- Crystal violet – Triarylmethane dye used as a histological stain and in Gram's method of classifying bacteria
- Fluorescein – Synthetic organic compound used as dye and fluorescent tracer
- Han purple and Han blue – Artificial barium copper silicate pigments developed in ancient China during the Han dynasty
- List of inorganic pigments
- Midnight blue – Dark shade of blue
- Phthalocyanine Blue BN– Synthetic blue pigment from the group of phthalocyanine dyes
References
- ISBN 978-0-470-68575-4.
- ^ ISBN 9780470166468.
- ^ "WHO Model List of Essential Medicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- OCLC 936144129.
- ^ a b Bartoll, Jens. "The early use of prussian blue in paintings" (PDF). 9th International Conference on NDT of Art, Jerusalem Israel, 25–30 May 2008. Retrieved 2010-01-22.
- ^ Berger, J. E. (c.1730) Kerrn aller Fridrichs=Städtschen Begebenheiten. Staatsbibliothek zu Berlin – Preußischer Kulturbesitz, Handschriftenabteilung, Ms. Boruss. quart. 124.
- ISBN 978-1606064290.
- ^ a b Frisch, J. L. (1896) Briefwechsel mit Gottfried Wilhelm Leibniz L. H. Fischer (ed.), Berlin, Stankiewicz Buchdruck, reprint Hildesheim/New York: Georg Olms Verlag, 1976
- ^ Frisch, J. L. (1710). "Serius Exhibita. Notitia Coerulei Berolinensis nuper inventi" [Addendum. Information about the recently discovered Berlin blue.]. Miscellanea Berolinensia Ad Incrementum Scientiarum (in Latin). 1: 377–378.
- PMID 21612121.
- ^ Bartoll, J.; Jackisch, B.; Most, M.; Wenders de Calisse, E.; Vogtherr, C. M. (2007). "Early Prussian Blue. Blue and green pigments in the paintings by Watteau, Lancret and Pater in the collection of Frederick II of Prussia". Techné. 25: 39–46.
- ^ Mulherron, Jamie (2001). "Prussian Blue, Boucher and Newton: the Material, Practice and Theory of Rococo painting". Object (3): 68–93.
- ^ Stahl, Georg Ernst (1731). Georgii Ernesti Stahlii, Experimenta, Observationes, Animadversiones, CCC Numero, Chymicae Et Physicae: Qualium alibi vel nulla, vel rara, nusquam autem satis ampla, ad debitos nexus, & veros usus, deducta mentio, commemeratio, aut explicatio, invenitur. Qualium partim, in aliis Autoris scriptis, varia mentio facta habetur; partim autem nova commemoratio hoc Tractatu exhibetur: utrimque vero, universa res uberius explicatur atque confirmatur (in Latin). Haude.
- .
- JSTOR 103734.. The recipe was subsequently published in Geoffroy, Étienne-François (1727) "Observations sur la Preparation de Bleu de Prusse ou Bleu de Berlin," Mémoires de l'Académie royale des Sciences année 1725. Paris. pp. 153–172.
- ^ "The Creation of Color in Eighteenth-Century Europe: Prussian Blue". www.gutenberg-e.org. Retrieved 2022-07-28.
- ^ Macquer, Pierre-Joseph (1752) "Éxamen chymique de bleu de Prusse," Mémoires de l'Académie royale des Sciences année 1752 ... (Paris, 1756), pp. 60–77. This article was reviewed in "Sur le bleu de Prusse," Histoire de l'Académie royale des Sciences... (1752), (Paris, 1756), pp. 79–85.
- ^ Scheele, Carl W. (1782) "Försök, beträffande det färgande ämnet uti Berlinerblå" (Experiment concerning the coloring substance in Berlin blue), Kungliga Svenska Vetenskapsakademiens handlingar (Royal Swedish Academy of Science's Proceedings), 3: 264–275 (in Swedish). Reprinted in Latin as: "De materia tingente caerulei berolinensis" in: Carl Wilhelm Scheele with Ernst Benjamin Gottlieb Hebenstreit (ed.) and Gottfried Heinrich Schäfer (trans.), Opuscula Chemica et Physica (Leipzig ("Lipsiae"), (Germany): Johann Godfried Müller, 1789), vol. 2, pages 148–174.
- ^ see Tekhelet#Sepia officinalis
- ^ "Has the long lost chilazon, source of the biblical blue techeiles dye been rediscovered?". 8 April 2008. Archived from the original on 8 April 2008. Retrieved 12 May 2020.
- ISBN 1855321602
- ISBN 1-85753-271-6
- ^ .
- ^ Egon Wiberg, Nils Wiberg, Arnold Frederick Holleman: Inorganic chemistry, p.1444. Academic Press, 2001; Google books
- ^ Journal of Toxicology, Suicide Attempt by Ingestion of Potassium Ferricyanide
- S2CID 236672995.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
- ^ a b c d "Prussian White". Macsen Labs. 2023-10-28. Retrieved 2024-03-16.
- ^ ISSN 0378-7753.
- .
- .
- .
- .
- .
- .
- .
- .
- .
- .
- .
- ^ .
- .
- ^ "Turning Big Ben's clock dials blue". UK Parliament. Retrieved 21 October 2023.
- ISBN 0894682563.
- ^ "Questions and Answers on Prussian Blue". Food and Drug Administration. Archived from the original on 2009-07-10. Retrieved 2020-03-20.
- ^ "Questions and Answers on Calcium-DTPA and Zinc-DTPA (Updated)". U.S. Food & Drug Administration. 3 November 2018. Retrieved 21 March 2020.
- ^ Radiogardase: Package insert with formula Archived 2011-03-20 at the Wayback Machine
- ^ Heyltex Corporation – Toxicology Archived 2007-11-12 at the Wayback Machine
- ^ Formula for Perls Prussian blue stain. Accessed April 2, 2009.
- ^ Hagerman, Ann E. (18 March 2011). "Tannin Chemistry" (PDF). Archived from the original (PDF) on 2013-08-26. Retrieved 2009-12-19. (1.41 MB)
- ISSN 0021-8561.
- Montreal Gazette. Retrieved February 28, 2017.
- ISSN 1945-7111.
- ISSN 0013-4651.
- ISSN 0001-4842.
- ISSN 1944-8244.
- ISSN 0025-5408.
External links
- The FDA's page on Prussian blue
- The CDC's page on Prussian blue
- National Pollutant Inventory – Cyanide compounds fact sheet
- Heyltex Corporation distributors of Radiogardase (Prussian blue insoluble capsules) Archived 2020-02-21 at the Wayback Machine
- Sarah Lowengard, "Prussian Blue" in The Creation of Color in Eighteenth Century Europe Columbia University Press, 2006
- Prussian blue, ColourLex
- Kraft, Alexander (2008). "On the discovery and history of Prussian blue" (PDF). Bull. Hist. Chem.33 (2): 61–67.