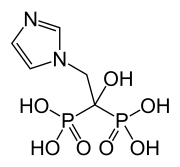
Zoledronic acid
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Reclast, Zometa, others[1] |
| Other names | zoledronate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605023 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous |
| Drug class | Bisphosphonate[3] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 22% |
| Metabolism | Nil |
| Elimination half-life | 146 hours |
| Excretion | Kidney (partial) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Zoledronic acid, also known as zoledronate and sold under the brand name Zometa
Common side effects include
Zoledronic acid was patented in 1986 and approved for medical use in the United States in 2001.[3][5] It is on the World Health Organization's List of Essential Medicines.[6][7]
Medical uses
Bone complications of cancer
Zoledronic acid is used to prevent
It can be given at home rather than in hospital. Such use has shown safety and quality-of-life benefits in people with breast cancer and bone metastases.[10]
Osteoporosis
Zoledronic acid, in brand name products Aclasta and Reclast among others,[11] may be given as a 5 mg infusion once per year for treatment of osteoporosis in men and post-menopausal women at increased risk of fracture.[12]
In 2007, the U.S. Food and Drug Administration (FDA) also approved it for the treatment of postmenopausal osteoporosis.[13][14]
Other
Zoledronic acid may be used for treatment of osteogenesis imperfecta.[15]
A single 5 mg dose of zoledronic acid is used for the treatment of Paget's disease.[medical citation needed][16]
Contraindications
- Poor kidney function (e.g. estimated glomerular filtration rate less than 30 mL/min)[17]
- Hypocalcaemia
- Pregnancy
- Paralysis
Side effects
Side effects can include
Kidneys
There is a risk of severe renal impairment. Appropriate hydration is important before administration, as is adequate
Zoledronate is rapidly processed via the
Bone
Osteonecrosis of the jaw
A rare complication that has been recently observed in cancer patients being treated with bisphosphonates is osteonecrosis of the jaw. This has mainly been seen in patients with multiple myeloma treated with zoledronic acid who have had dental extractions.[22]
Atypical fractures
After approving the drug on 8 July 2009, the
Pharmacology
As a
| Bisphosphonate | Relative potency |
|---|---|
| Etidronate | 1 |
| Tiludronate | 10 |
| Pamidronate | 100 |
| Alendronate | 100-500 |
| Ibandronate | 500-1000 |
| Risedronate | 1000 |
| Zoledronate | 5000 |
Research
Zoledronic acid has been found to have a direct antitumor effect and to synergistically augment the effects of other antitumor agents in osteosarcoma cells.[27]
Zoledronic acid has shown significant benefits versus placebo over three years, with a reduced number of
Zoledronic acid also attenuates accumulation of
With hormone therapy for breast cancer
An increase in
In a meta-analysis of trials where upfront zoledronic acid was given to prevent aromatase inhibitor-associated bone loss, active cancer recurrence appeared to be reduced.[32]
As of 2010[update] "The results of clinical studies of adjuvant treatment on early-stage hormone-receptor-positive breast-cancer patients under hormonal treatment – especially with the bisphosphonate zoledronic acid – caused excitement because they demonstrated an additive effect on decreasing disease relapses at bone or other sites. A number of clinical and in vitro and in vivo preclinical studies, which are either ongoing or have just ended, are investigating the mechanism of action and antitumoral activity of bisphosphonates."[33]
A 2010 review concluded that "adding zoledronic acid 4 mg intravenously every 6 months to endocrine therapy in premenopausal women with hormone receptor-positive early breast cancer ... is cost-effective from a US health care system perspective".[34]
References
- ^ "International trade names for zoledronic acid". Drugs.com. Retrieved 14 January 2015.
- ^ a b "Zoledronic acid Use During Pregnancy". Drugs.com. 1 June 2020. Retrieved 19 October 2020.
- ^ a b c d e f g h i j "Zoledronic Acid". The American Society of Health-System Pharmacists. Retrieved 8 December 2017.
- ^ "PRESS RELEASE: Novartis's Reclast Receives FDA Approval FOR Women With Postmenopausal Osteoporosis". FierceBiotech. 20 August 2007. Retrieved 2021-09-02.
- ISBN 9783527607495.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ National Prescribing Service (2009). "Zoledronic Acid for Osteoporosis". Medicines Update, Available at "Zoledronic acid (Aclasta) for osteoporosis: National Prescribing Service Ltd NPS". Archived from the original on April 23, 2010. Retrieved January 20, 2010.
- ^ "Zomera prescribing information" (PDF). Novartis Pharmaceuticals Corporation. U.S. Food and Drug Administration. April 2014.
- PMID 15870721.
- S2CID 22079489.
- ^ PMID 17878149.
- ^ "Biotech PRESS RELEASE: Novartis's Reclast Receives FDA Approval FOR Women With Postmenopausal Osteoporosis" (Press release). FierceBiotech, A Division of Questex A FierceMarkets Publication. August 20, 2007. Retrieved 2018-03-27.
- ^ S2CID 71443125.
- PMID 27760454.
- ^ "Paget's Disease of Bone". www.rheumatology.org. Retrieved 2015-07-09.
- PMID 20332498.
- ^ "NPS MedicineWise" (PDF). Archived from the original (PDF) on 2016-03-04. Retrieved 2014-01-25.
- ^ "Zometa 4mg/5ml Concentrate for Solution for Infusion". medicines.org.uk. Archived from the original on 2010-02-24. Retrieved 2010-02-24.
- ^ "FDA Alert: Reclast (zoledronic acid): Drug Safety Communication - New Contraindication and Updated Warning on Kidney Impairment". drugs.com.
- ^ "European Medicines Agency - Human medicines". europa.eu. Archived from the original on 2015-09-25. Retrieved 2012-04-03.
- PMID 16000365.
- ^ a b "European Medicines Agency - Human medicines". europa.eu. Archived from the original on 2013-01-19. Retrieved 2012-04-03.
- ^ "Aclasta label- Australia" (PDF). Archived from the original (PDF) on 2016-03-04. Retrieved 2014-01-25.
- ^ "Bisphosphonates". International Osteoporosis Foundation. Retrieved 30 July 2022.
- OCLC 868299888.
- PMID 20514467.
- PMID 11870242.
- PMID 26679354.
- PMID 19213681.
- PMID 20234364.
- S2CID 23710758.
- S2CID 26073229.
- PMID 20705558.
External links
- "Zoledronic acid". Drug Information Portal. U.S. National Library of Medicine.
