Abdominal aortic aneurysm
| Abdominal aortic aneurysm | |
|---|---|
aortic aneurysms (2015)[5] |
Abdominal aortic aneurysm (AAA) is a localized enlargement of the abdominal aorta such that the diameter is greater than 3 cm or more than 50% larger than normal.[1] An AAA usually causes no symptoms, except during rupture.[1] Occasionally, abdominal, back, or leg pain may occur.[2] Large aneurysms can sometimes be felt by pushing on the abdomen.[2] Rupture may result in pain in the abdomen or back, low blood pressure, or loss of consciousness, and often results in death.[1][6]
AAAs occur most commonly in men, those over 50 and those with a family history of the disease.
Abstinence from cigarette smoking is the single best way to prevent the disease.[1] Other methods of prevention include treating high blood pressure, treating high blood cholesterol, and avoiding being overweight.[1] Surgery is usually recommended when the diameter of an AAA grows to >5.5 cm in males and >5.0 cm in females.[1] Other reasons for repair include the presence of symptoms and a rapid increase in size, defined as more than one centimeter per year.[2] Repair may be either by open surgery or endovascular aneurysm repair (EVAR).[1] As compared to open surgery, EVAR has a lower risk of death in the short term and a shorter hospital stay, but may not always be an option.[1][9][10] There does not appear to be a difference in longer-term outcomes between the two.[11] Repeat procedures are more common with EVAR.[12]
AAAs affect 2-8% of males over the age of 65.[1] They are five times more common in men.[13] In those with an aneurysm less than 5.5 cm, the risk of rupture in the next year is below 1%.[1] Among those with an aneurysm between 5.5 and 7 cm, the risk is about 10%, while for those with an aneurysm greater than 7 cm the risk is about 33%.[1] Mortality if ruptured is 85% to 90%.[1] During 2013, aortic aneurysms resulted in 168,200 deaths, up from 100,000 in 1990.[5][14] In the United States AAAs resulted in between 10,000 and 18,000 deaths in 2009.[4]
Signs and symptoms

The vast majority of aneurysms are asymptomatic. However, as the abdominal aorta expands and/or ruptures, the aneurysm may become painful and lead to pulsating sensations in the abdomen or pain in the chest, lower back, legs, or scrotum.[15]
Complications
The complications include rupture, peripheral
The signs and symptoms of a ruptured AAA may include severe pain in the lower back, flank, abdomen or groin. A mass that pulses with the heart beat may also be felt.
Causes
The exact causes of the degenerative process remain unclear. There are, however, some hypotheses and well-defined
- Tobacco smoking: More than 90% of people who develop an AAA have smoked at some point in their lives.[21]
- Alcohol and hypertension: The inflammation caused by prolonged use of alcohol and hypertensive effects from abdominal edema which leads to hemorrhoids, esophageal varices, and other conditions, is also considered a long-term cause of AAA.[citation needed]
- Genetic influences: The influence of genetic factors is high. AAA is four to six times more common in male siblings of known patients, with a risk of 20–30%.heterozygous females. Other hypotheses of genetic causes have also been formulated.[16] Connective tissue disorders, such as Marfan syndrome and Ehlers-Danlos syndrome, have also been strongly associated with AAA.[18] Both relapsing polychondritis and pseudoxanthoma elasticum may cause abdominal aortic aneurysm.[24]
- Atherosclerosis: The AAA was long considered to be caused by atherosclerosis, because the walls of the AAA frequently carry an atherosclerotic burden. However, this hypothesis cannot explain the initial defect and the development of occlusion, which is observed in the process.[16] Another hypothesis is that plaque buildup can cause a feed-forward dysfunction in the signaling among neurons that regulate pressure in the aorta. This feed-forward process leads to an over-pressuring condition that ruptures in the aorta.[25]
- Other causes of the development of AAA include: cystic medial necrosis.[18]
Pathophysiology


The most striking
However, theDiagnosis
An abdominal aortic aneurysm is usually diagnosed by
An aneurysm ruptures if the mechanical stress (tension per area) exceeds the local wall strength; consequently, peak wall stress (PWS),[30] mean wall stress (MWS),[31] and peak wall rupture risk (PWRR)[32] have been found to be more reliable parameters than diameter to assess AAA rupture risk. Medical software allows computing these rupture risk indices from standard clinical CT data and provides a patient-specific AAA rupture risk diagnosis.[33][34][35] This type of biomechanical approach has been shown to accurately predict the location of AAA rupture.[34][35][36]
-
Aortic measurement onaxial plane between the outer margins of the aortic wall[37]
-
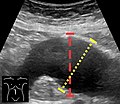
Ultrasonography in the sagittal plane, showing axial plane measure (dashed red line), as well as maximal diameter (dotted yellow line), which is preferred
-
A ruptured AAA with an open arrow marking the aneurysm and the closed arrow marking the free blood in the abdomen
-
Sagittal CT image of an AAA
-
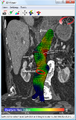
Biomechanical AAA rupture risk prediction
-
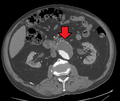
An axial contrast-enhanced CT scan demonstrating an abdominal aortic aneurysm of 4.8 by 3.8 cm
-
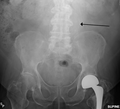
The faint outline of the calcified wall of an AAA as seen on plain X-ray
-
Abdominal aortic aneurysms (3.4 cm)
-
An aortic aneurysm as seen on CT with a small area of remaining blood flow
-
Ultrasound showing a previously repaired AAA that is leaking with flow around the graft[38]
-
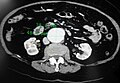
Ultrasonography of an aneurysm with a mural thrombus
Classification
| Ectatic or mild dilatation |
>2.0 cm and <3.0 cm[39] |
| Moderate | 3.0 - 5.0 cm[39] |
| Large or severe | >5.0[39] or 5.5[40] cm |
Abdominal aortic aneurysms are commonly divided according to their size and symptomatology. An aneurysm is usually defined as an outer aortic diameter over 3 cm (normal diameter of the aorta is around 2 cm),[41] or more than 50% of normal diameter.[42] If the outer diameter exceeds 5.5 cm, the aneurysm is considered to be large.[40] Ruptured AAA should be suspected in any person older than 60 who experiences collapse, unexplained low blood pressure, or sudden-onset back or abdominal pain. Abdominal pain, shock, and a pulsatile mass is only present in a minority of cases.[citation needed] Although an unstable person with a known aneurysm may undergo surgery without further imaging, the diagnosis will usually be confirmed using CT or ultrasound scanning.[citation needed]
The suprarenal aorta normally measures about 0.5 cm larger than the infrarenal aorta.[43]
Differential diagnosis
Aortic aneurysm rupture may be mistaken for the pain of kidney stones, or muscle related back pain.[6]
Prevention
- Smoking cessation
- Treatment of hypertension
Screening
The
In the United Kingdom the NHS AAA Screening Programme invites men in England for screening during the year they turn 65. Men over 65 can contact the programme to arrange to be screened.[46]
In Sweden one time screening is recommended in all males over 65 years of age.[1][8] This has been found to decrease the risk of death from AAA by 42% with a number needed to screen of just over 200.[45] In those with a close relative diagnosed with an aortic aneurysm, Swedish guidelines recommend an ultrasound at around 60 years of age.[47]
Australia has no guideline on screening.[48]
Repeat ultrasounds should be carried out in those who have an aortic size greater than 3.0 cm.[49] In those whose aorta is between 3.0 and 3.9 cm this should be every three years, if between 4.0 and 4.4 cm every two years, and if between 4.5 and 5.4 cm every year.[49]
Management
The treatment options for asymptomatic AAA are
Conservative
Conservative management is indicated in people where repair carries a high risk of mortality and in patients where repair is unlikely to improve life expectancy. The mainstay of the conservative treatment is smoking cessation.[citation needed]
Surveillance is indicated in small asymptomatic aneurysms (less than 5.5 cm) where the risk of repair exceeds the risk of rupture.[50] As an AAA grows in diameter, the risk of rupture increases. Surveillance until an aneurysm has reached a diameter of 5.5 cm has not been shown to have a higher risk as compared to early intervention.[53][54]
Medication
No medical therapy has been found to be effective at decreasing the growth rate or rupture rate of asymptomatic AAAs.
Surgery
The threshold for repair varies slightly from individual to individual, depending on the balance of risks and benefits when considering repair versus ongoing surveillance. The size of an individual's native aorta may influence this, along with the presence of comorbidities that increase operative risk or decrease life expectancy. Evidence, however, does not usually support repair if the size is less than 5.5 cm.[50]
Open repair
Open repair is indicated in young patients as an elective procedure, or in growing or large, symptomatic or ruptured aneurysms. The aorta must be clamped during the repair, denying blood to the abdominal organs and sections of the spinal cord; this can cause a range of complications. As it is essential to perform the critical part of the operation quickly, the incision is typically made large enough to facilitate the fastest repair. Recovery after open AAA surgery takes significant time. The minimums are a few days in intensive care, a week total in the hospital and a few months before full recovery.[citation needed]
Endovascular repair

Endovascular repair first became practical in the 1990s and although it is now an established alternative to open repair, its role is yet to be clearly defined. It is generally indicated in older, high-risk patients or patients unfit for open repair. However, endovascular repair is feasible for only a portion of AAAs, depending on the morphology of the aneurysm. The main advantages over open repair are that there is less peri-operative mortality, less time in
Rupture
In those with aortic rupture of the AAA, treatment is immediate surgical repair. There appear to be benefits to allowing permissive hypotension and limiting the use of intravenous fluids during transport to the operating room.[62]
Prognosis
| AAA Size (cm) | Growth rate (cm/yr)[63] | Annual rupture risk (%)[64] |
|---|---|---|
| 3.0–3.9 | 0.39 | 0 |
| 4.0–4.9 | 0.36 | 0.5–5 |
| 5.0–5.9 | 0.43 | 3–15 |
| 6.0–6.9 | 0.64 | 10–20 |
| >=7.0 | - | 20–50 |
Although the current standard of determining rupture risk is based on maximum diameter, it is known that smaller AAAs that fall below this threshold (diameter<5.5 cm) may also rupture, and larger AAAs (diameter>5.5 cm) may remain stable.[65][66] In one report, it was shown that 10–24% of ruptured AAAs were less than 5 cm in diameter.[66] It has also been reported that of 473 non-repaired AAAs examined from autopsy reports, there were 118 cases of rupture, 13% of which were less than 5 cm in diameter. This study also showed that 60% of the AAAs greater than 5 cm (including 54% of those AAAs between 7.1 and 10 cm) never experienced rupture.[67] Vorp et al. later deduced from the findings of Darling et al. that if the maximum diameter criterion were followed for the 473 subjects, only 7% (34/473) of cases would have died from rupture prior to surgical intervention as the diameter was less than 5 cm, with 25% (116/473) of cases possibly undergoing unnecessary surgery since these AAAs may never have ruptured.[67]
Alternative methods of rupture assessment have been recently reported. The majority of these approaches involve the numerical analysis of AAAs using the common engineering technique of the finite element method (FEM) to determine the wall stress distributions. Recent reports have shown that these stress distributions have been shown to correlate to the overall geometry of the AAA rather than solely to the maximum diameter.[68][69][70] It is also known that wall stress alone does not completely govern failure as an AAA will usually rupture when the wall stress exceeds the wall strength. In light of this, rupture assessment may be more accurate if both the patient-specific wall stress is coupled together with patient-specific wall strength. A noninvasive method of determining patient-dependent wall strength was recently reported,[71] with more traditional approaches to strength determination via tensile testing performed by other researchers in the field.[72][73][74] Some of the more recently proposed AAA rupture-risk assessment methods include: AAA wall stress;[30][75][76] AAA expansion rate;[77] degree of asymmetry;[70] presence of intraluminal thrombus (ILT);[78] a rupture potential index (RPI);[79][80] a finite element analysis rupture index (FEARI);[81] biomechanical factors coupled with computer analysis;[82] growth of ILT;[83] geometrical parameters of the AAA;[84] and also a method of determining AAA growth and rupture based on mathematical models.[85][86]
The postoperative mortality for an already ruptured AAA has slowly decreased over several decades but remains higher than 40%.[87] However, if the AAA is surgically repaired before rupture, the postoperative mortality rate is substantially lower, approximately 1-6%.[88]
Epidemiology
The occurrence of AAA varies by ethnicity. In the United Kingdom, the rate of AAA in Caucasian men older than 65 years is about 4.7%, while in Asian men it is 0.45%.[89] It is also less common in individuals of African, and Hispanic heritage.[1] They occur four times more often in men than in women.[1]
There are at least 13,000 deaths yearly in the U.S. secondary to AAA rupture.[1] The peak number of new cases per year among males is around 70 years of age, and the percentage of males affected over 60 years is 2–6%. The frequency is much higher in smokers than in non-smokers (8:1), and the risk decreases slowly after smoking cessation.[90] In the U.S., the incidence of AAA is 2–4% in the adult population.[16]
Rupture of the AAA occurs in 1–3% of men aged 65 or more, for whom the mortality rate is 70–95%.[40]
History
The first historical records about AAA are from Ancient Rome in the 2nd century AD, when Greek surgeon Antyllus tried to treat the AAA with proximal and distal ligature, central incision and removal of thrombotic material from the aneurysm. However, attempts to treat the AAA surgically were unsuccessful until 1923. In that year, Rudolph Matas (who also proposed the concept of endoaneurysmorrhaphy), performed the first successful aortic ligation on a human.[91] Other methods that were successful in treating the AAA included wrapping the aorta with polyethene cellophane, which induced fibrosis and restricted the growth of the aneurysm. Endovascular aneurysm repair was first performed in the late 1980s and has been widely adopted in the subsequent decades. Endovascular repair was first used for treating a ruptured aneurysm in Nottingham in 1994.[92]
Society and culture
Theoretical physicist Albert Einstein underwent an operation for an abdominal aortic aneurysm in 1949 that was performed by Rudolph Nissen, who wrapped the aorta with polyethene cellophane. Einstein's aneurysm ruptured on April 13, 1955. He declined surgery, saying, "I want to go when I want. It is tasteless to prolong life artificially. I have done my share, it is time to go. I will do it elegantly." He died five days later at age 76.[93]
Actress
Musician Conway Twitty died in June 1993 from an abdominal aortic aneurysm at the age of 59, two months before the release of what would be his final studio album, Final Touches.[citation needed]
Actor George C. Scott died in 1999 from a ruptured abdominal aortic aneurysm at age 71.[citation needed]
In 2001, former presidential candidate Bob Dole underwent surgery for an abdominal aortic aneurysm in which a team led by vascular surgeon Kenneth Ouriel inserted a stent graft:[95]
Ouriel said that the team inserted a Y-shaped tube through an incision in Dole's leg and placed it inside the weakened portion of the aorta. The aneurysm will eventually contract around the stent, which will remain in place for the rest of Dole's life.[95]
Actor
Actor Tommy Ford died of abdominal aneurysm in October 2016 at 52 years old.[96]
Gary Gygax, co-creator of Dungeons & Dragons, died in 2008 from an abdominal aortic aneurysm at the age of 69.
Research
Risk assessment
There have been many calls for alternative approaches to rupture risk assessment over the past number of years, with many believing that a biomechanics-based approach may be more suitable than the current diameter approach. Numerical modeling is a valuable tool to researchers allowing approximate wall stresses to be calculated, thus revealing the rupture potential of a particular aneurysm. Experimental models are required to validate these numerical results and provide a further insight into the biomechanical behavior of the AAA. In vivo, AAAs exhibit a varying range of material strengths[97] from localised weak hypoxic regions[98] to much stronger regions and areas of calcifications.[99]
Finding ways to predict future AAA growth is seen as a research priority.[100]
Another related line of research is utilizing mathematical decision modeling (e.g.,
Experimental models
Experimental models can now be manufactured using a novel technique involving the injection-moulding lost-wax manufacturing process to create patient-specific anatomically correct AAA replicas.[104] Work has also focused on developing more realistic material analogues to those in vivo, and recently a novel range of silicone-rubbers was created allowing the varying material properties of the AAA to be more accurately represented.[105] These rubber models can also be used in a variety of experimental situations, from stress analysis using the photoelastic method[106] New endovascular devices are being developed that are able to treat more complex and tortuous anatomies.[107]
Prevention and treatment
An animal study showed that removing a single protein prevents early damage in blood vessels from triggering a later-stage, complications. By eliminating the gene for a signaling protein called cyclophilin A (CypA) from a strain of mice, researchers were able to provide complete protection against abdominal aortic aneurysm.[108]
Other recent research identified Granzyme B (GZMB) (a protein-degrading enzyme) to be a potential target in the treatment of abdominal aortic aneurysms. Elimination of this enzyme in mice models both slowed the progression of aneurysms and improved survival.[109][110]
Preclinical Research
The mechanisms that lead to AAA development are still not completely understood at a cellular and molecular level. In order to better understand the pathophysiology of AAA, it is often necessary to use experimental animal models. It is often questioned how well do these models translate to human disease. Even though there is no animal model that exactly represents the human condition, all the existing ones focus on one different pathophysiological aspect of the disease. Combining the results from different animal models with clinical research can provide a better overview of the AAA pathophysiology. The most common animal models are rodents (mice and rats), although for certain studies, such as testing preclinical devices or surgical procedures, large animal models (pig, sheep) are more frequently used. The rodent models of AAA can be classified according to different aspects. There are dissecting models vs non-dissecting models and genetically determined models vs chemically induced models. The most commonly used models are the angiotensin-II infusion into ApoE knockout mice (dissecting model, chemically induced), the calcium chloride model (non-dissecting, chemically induced) and the elastase model (non-dissecting, chemically induced model).[111][112] A recent study has shown that β-Aminopropionitrile plus elastase application to abdominal aorta causes more severe aneurysm in mice as compared to elastase alone.[113]
References
- ^ PMID 25427112.
- ^ PMID 16623206.
- ^ PMID 22040707.
- ^ a b c d "Aortic Aneurysm Fact Sheet". cdc.gov. July 22, 2014. Archived from the original on 3 February 2015. Retrieved 3 February 2015.
- ^ PMID 27733281.
- ^ PMID 25635203.
- ^ S2CID 265875637.
- ^ PMID 25443524.
- PMID 25006502.
- PMID 21820922.
- PMID 24453068.
- PMID 25443774.
- ISBN 978-0-7020-7870-5.
- PMID 25530442.
- ISBN 978-0-07-146633-2.
- ^ a b c d e f Abdominal Aortic Aneurysm at eMedicine
- PMID 10493476.
- ^ ISBN 80-7169-724-9
- ISBN 978-1-4377-2788-3.
- PMID 25884861.
- PMID 18234753.
- S2CID 27523622.
- S2CID 40560333.
- ISBN 978-1-4160-2999-1.
- PMID 15922098.
- PMID 20035050.
- PMID 21900960.
- S2CID 21272785.
- PMID 21471188.
- ^ PMID 12663969.
- PMID 36354512.
- PMID 20447844.
- ^ "VASCOPS". Archived from the original on 2011-07-17. Retrieved 2011-01-15.
- ^ S2CID 204813645.
- ^ PMID 34318314.
- S2CID 35797072.
- ^ Timothy Jang (2017-08-28). "Bedside Ultrasonography Evaluation of Abdominal Aortic Aneurysm - Technique". Medscape. Archived from the original on 2018-01-25.
- ^ "UOTW #35 - Ultrasound of the Week". Ultrasound of the Week. 27 January 2015. Archived from the original on 9 May 2017. Retrieved 27 May 2017.
- ^ ISBN 978-0-323-27817-1.
- ^ PMID 15757960.
- ^ PMID 16990459.
- PMID 25427112.
- ^ Jeffrey Jim, Robert W Thompson (2018-03-05). "Clinical features and diagnosis of abdominal aortic aneurysm". UpToDate. Archived from the original on 2018-03-30.
- ^ PMID 31821437.
- ^ PMID 27871502.
- ^ "NHS Abdominal Aortic Aneurysm Screening Programme" (PDF).
- ^ "Aortascreening av nära släkting (Aortic screening of close relative)" (PDF). Västra Götaland Regional Council. Archived from the original (PDF) on 2019-01-04. Retrieved 2019-01-03.
- PMID 23781541.
- ^ PMID 23443444.
- ^ PMID 32609382.
- S2CID 9019195.
- S2CID 231760989.
- S2CID 25392801.
- PMID 12000813.
- PMID 16782510.
- S2CID 20273503.
- S2CID 54288202.
- PMID 15944424.
- S2CID 26174408.
- .
- PMID 28548204.
- PMID 24670823.
- PMID 6465980.
- PMID 12756363.
- PMID 884821.
- ^ PMID 9808857.
- ^ PMID 17254589.
- PMID 9576075.
- S2CID 21394944.
- ^ PMID 19028061.
- S2CID 25411800.
- PMID 16337949.
- S2CID 21970395.
- S2CID 218868284.
- PMID 12218986.
- PMID 15234698.
- PMID 9537528.
- PMID 12218961.
- S2CID 23649698.
- S2CID 40000934.
- hdl:10344/184.
- PMID 16529648.
- PMID 11112467.
- S2CID 22127197.
- S2CID 10539114.
- PMID 18255074.
- S2CID 30298923.
- S2CID 26394236.
- PMID 19666232.
- PMID 10587395.
- PMID 16107099.
- S2CID 45648419.
- ^ Lowenfels AB (14 June 2002). "Famous Patients, Famous Operations, Part 3". Medscape.
- ^ Ball L (April 27, 1989). "Ball dies of ruptured aorta". Los Angeles Times. Retrieved May 12, 2013.
- ^ a b "Bob Dole has surgery to treat aneurysm". USA Today via Associated Press. 2001-06-27. Archived from the original on 2009-05-07. Retrieved 2009-09-22.
- ^ Mele C (12 October 2016). "Thomas Mikal Ford, Known for His Role in '90s Sitcom 'Martin,' Dies at 52". The New York Times. Retrieved 8 March 2019.
- PMID 16337949.
- PMID 11496282.
- PMID 17227104.
- PMID 28874296.
- S2CID 5632830.
- PMID 25924187.
- PMID 38336915.
- PMID 18532870.
- PMID 19595622.
- PMID 19642790.
- ProQuest 224419745.
- ^ "Study establishes major new treatment target in diseased arteries". U.S. News & World Report. May 10, 2009. Archived from the original on June 6, 2011.
- PMID 20035050.
- ^ "Discovery points way for new treatment for aneurysms". University of British Columbia. January 27, 2010. Archived from the original on February 19, 2010.
- PMID 28062500.
- S2CID 28971998.
- PMID 34997075.

![Aortic measurement on abdominal ultrasonography in the axial plane between the outer margins of the aortic wall[37]](http://upload.wikimedia.org/wikipedia/commons/thumb/7/7b/Ultrasonographic_measurement_of_aortic_diameter_at_the_navel.svg/120px-Ultrasonographic_measurement_of_aortic_diameter_at_the_navel.svg.png)