Acetoacetic acid

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-Oxobutanoic acid[1] | |
| Systematic IUPAC name
3-Oxobutyric acid | |
| Other names
Acetoacetic acid
Diacetic acid Acetylacetic acid Acetonecarboxylic acid | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H6O3 | |
| Molar mass | 102.089 g·mol−1 |
| Appearance | Colorless, oily liquid |
| Melting point | 36.5 °C (97.7 °F; 309.6 K) |
| Boiling point | Decomposes |
| Soluble | |
| Solubility in organic solvents | Soluble in ethanol, ether |
| Acidity (pKa) | 3.58[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Acetoacetic acid (
Biochemistry
Under typical physiological conditions, acetoacetic acid exists as its
- AcCH2CO2H → AcCH2CO−2 + H+
Unbound acetoacetate is primarily produced by
- AcCH2C(O)−CoA + OH− → AcCH2CO−2 + H−CoA
The acetoacetate-CoA itself is formed by three routes:
- 3-hydroxy-3-methylglutaryl CoA releases acetyl CoA and acetoacetate:
- −O2CCH2−C(Me)(OH)−CH2C(O)−CoA → −O2CCH2−Ac + Ac−CoA
- −O2CCH2−C(
- Acetoacetyl-CoA can come from beta oxidation of butyryl-CoA:
- Et−CH2C(O)−CoA + 2NAD+ + H2O + FAD → Ac−CH2C(O)−CoA + 2NADH + FADH2
- Condensation of pair of acetyl CoA molecules as catalyzed by thiolase.[4]: 393
- 2Ac−CoA → AcCH2C(O)−CoA + H−CoA
In mammals, acetoacetate produced in the liver (along with the other two "
Synthesis and properties
Acetoacetic acid may be prepared by the hydrolysis of diketene. Its esters are produced analogously via reactions between diketene and alcohols,[3] and acetoacetic acid can be prepared by the hydrolysis of these species.[6] In general, acetoacetic acid is generated at 0 °C and used in situ immediately.[7]
It decomposes at a moderate reaction rate into acetone and carbon dioxide:
- CH3C(O)CH2CO2H → CH3C(O)CH3 + CO2
The acid form has a half-life of 140 minutes at 37 °C in water, whereas the basic form (the anion) has a half-life of 130 hours. That is, it reacts about 55 times more slowly.[8] The corresponding decarboxylation of trifluoroacetoacetate is used to prepare trifluoroacetone:
- CF3C(O)CH2CO2H → CF3C(O)CH3 + CO2
It is a
Acetoacetic acid displays keto-enol
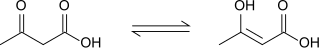
Applications

Acetoacetic esters are used for the
Detection
Acetoacetic acid is measured in the urine of people with diabetes to test for
Similar tests are used in
See also
References
- ISBN 978-0-85404-182-4.
- ^ Dawson, R. M. C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ^ ISBN 978-3527306732.
- ^ Biochemistry(2nd ed.).
- ISBN 0-7167-2009-4.
- .
- ; Collected Volumes, vol. 4, p. 633.
- .
- ISSN 0022-3263.
- PMID 17999519.
- ^ PMID 26975543.
- S2CID 21195842.
- PMID 24351673.
- S2CID 2819002.
- PMID 27435643.

