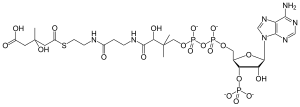
HMG-CoA
| |
| Names | |
|---|---|
| IUPAC name
(9R,21S)-1-[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)tetrahydrofuran-2-yl]-3,5,9,21-tetrahydroxy-8,8,21-trimethyl-10,14,19-trioxo-2,4,6-trioxa-18-thia-11,15-diaza-3,5-diphosphatricosan-23-oic acid 3,5-dioxide
| |
| Other names
3-hydroxy-3-methylglutaryl CoA; 3-hydroxy-3-methylglutaryl coenzyme A
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.014.820 |
IUPHAR/BPS |
|
| MeSH | HMG-CoA |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C27H44N7O20P3S | |
| Molar mass | 911.661 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Hydroxy β-methylglutaryl-CoA (HMG-CoA), also known as 3-hydroxy-3-methylglutaryl coenzyme A, is an intermediate in the
HMG-CoA is a
HMG-CoA reductase catalyzes the conversion of HMG-CoA to mevalonic acid, a necessary step in the biosynthesis of cholesterol.
Biosynthesis
Mevalonate pathway
HMG-CoA synthase.[8]
In the final step of
isoprenoid groups that are incorporated into a wide variety of end-products, including cholesterol in humans.[9]

Ketogenesis pathway
acetoacetate
.

See also
References
- ^ Sarkar DP (2015). "Classics in Indian Medicine" (PDF). The National Medical Journal of India (28): 3. Archived from the original (PDF) on 2016-05-31.
- .
- ^ "Valine, leucine and isoleucine degradation - Reference pathway". Kyoto Encyclopedia of Genes and Genomes. Kanehisa Laboratories. 27 January 2016. Retrieved 1 February 2018.
- ^ PMID 23374455.
- ^ ISBN 978-0-12-387784-0. Retrieved 6 June 2016.
Energy fuel: Eventually, most Leu is broken down, providing about 6.0kcal/g. About 60% of ingested Leu is oxidized within a few hours ... Ketogenesis: A significant proportion (40% of an ingested dose) is converted into acetyl-CoA and thereby contributes to the synthesis of ketones, steroids, fatty acids, and other compounds
Figure 8.57: Metabolism of L-leucine - ISBN 978-1-305-57720-6.
- PMID 22971202.