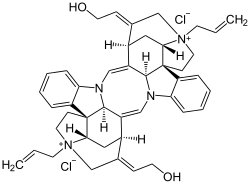
Alcuronium chloride
Appearance
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Alloferin |
| Other names | Ro 4-3816, diallylnortoxiferine |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Pharmacokinetic data | |
| Metabolism | not metabolized |
| Elimination half-life | 2–4 hours |
| Excretion | 70–90% unchanged in urine 1.3 mL/kg/min |
| Identifiers | |
| |
JSmol) | |
SMILES
| |
| |
| | |
Alcuronium chloride (formerly marketed as Alloferin) is a
gantacurium
. The replacement of both the N-methyl groups with N-allyl moieties yielded N,N-diallyl-bis-nortoxiferine, now recognized as alcuronium.
Inclusion of the allylic functions presented an enhanced potential area of biotransformation, and thus alcuronium is observed to have a much shorter duration of neuromuscular blocking action than its parent C-toxiferine I.
Effects
- Cardiovascular system: histamine release and blockage of the sympathetic ganglia including adrenal medulla could cause hypotension
- Respiratory system: apnea due to phrenic blockage but bronchoconstriction can occur from the histamine release
- Central nervous system: no effect on intraocular pressure
- gut motility
Special points
- Duration of action prolonged in states of low potassium, calcium and protein, also in states of high magnesium and acidosis.
- Pharmaceutically incompatible with thiopentone
- dilated pupils
See also
References
- PMID 13208908.
- PMID 14793878.)
{{cite journal}}: CS1 maint: untitled periodical (link - ^ Martin-Smith M (1971), In: Ariens EJ (ed.), "Drug Design". Vol. 2. Academic Press. New York and London. pp.453-530.
- ^ S2CID 20303531.
- PMID 6102875.)
{{cite journal}}: CS1 maint: untitled periodical (link - S2CID 36615570.
- PMID 130154.
Further reading
- Zahn K, Eckstein N, Tränkle C, Sadée W, Mohr K (2002). "Allosteric modulation of muscarinic receptor signaling: alcuronium-induced conversion of pilocarpine from an agonist into an antagonist". J Pharmacol Exp Ther. 301 (2): 720–8. S2CID 534003.
- Maass A, Mohr K (1996). "Opposite effects of alcuronium on agonist and on antagonist binding to muscarinic receptors". Eur J Pharmacol. 305 (1–3): 231–4. PMID 8813558.
- Jakubík J, Tucek S (1994). "Protection by alcuronium of muscarinic receptors against chemical inactivation and location of the allosteric binding site for alcuronium". J Neurochem. 63 (5): 1932–40. S2CID 23053191.
- Proska J, Tucek S (1994). "Mechanisms of steric and cooperative actions of alcuronium on cardiac muscarinic acetylcholine receptors". Mol Pharmacol. 45 (4): 709–17. PMID 8183250.
