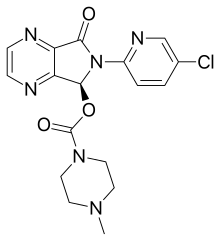
Eszopiclone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Lunesta, Eszop, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605009 |
| License data |
|
By mouth | |
| ATC code | |
| Legal status | |
| Legal status | |
| Elimination half-life | 6 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Eszopiclone, sold under the brand name Lunesta among others, is a
Common side effects include headache,
Approved for medical use in the United States in 2004,
Medical uses
A 2018 Cochrane review found that it produced moderate improvement in sleep onset and maintenance. The authors suggest that where preferred non-pharmacological treatment strategies have been exhausted, eszopiclone provides an efficient treatment for insomnia.
Eszopiclone is slightly effective in the treatment of insomnia where difficulty in falling asleep is the primary complaint.[4] The benefit over placebo was found to be of questionable clinical significance.[4] Although the drug effect and the placebo response were rather small and of questionable clinical importance, the two together produce a reasonably large clinical response.[4]
Elderly
Sedative hypnotic drugs including eszopiclone are more commonly prescribed to the elderly than to younger patients despite benefits of medication being generally unimpressive.[16]
In 2015, the American Geriatrics Society reviewed the safety information about eszopiclone and similar drugs and concluded that the "nonbenzodiazepine, benzodiazepine receptor agonist hypnotics (eszopiclone, zaleplon, zolpidem) are to be avoided without consideration of duration of use because of their association with harms balanced with their minimal efficacy in treating insomnia."
The review made this determination both because of the relatively large dangers to elderly individuals from zolpidem and other "z-drugs" together with the fact the drugs have "minimal efficacy in treating insomnia." This was a change from the 2012 AGS recommendation, which suggested limiting use to 90 days or less. The review stated: "the 90‐day‐use caveat [was] removed from nonbenzodiazepine, benzodiazepine receptor agonist hypnotics, resulting in an unambiguous 'avoid' statement (without caveats) because of the increase in the evidence of harm in this area since the 2012 update."[17]
An extensive review of the medical literature regarding the management of insomnia and the elderly found that there is considerable evidence of the effectiveness and durability of non-drug treatments for insomnia in adults of all ages and that these interventions are underutilized. Compared with the benzodiazepines, the
A 2009 meta-analysis found a higher rate of infections.[19]
Adverse effects
Sleeping pills, including eszopiclone, have been associated with an increased risk of death.[20]
Hypersensitivity to eszopiclone is a contraindication to its use. The presence of liver impairment, lactation and activities requiring mental alertness (e.g., driving) may be considered when determining frequency and dosage.[7]
- unpleasant taste[7]
- headache[7]
- peripheral edema[7][21]
- chest pain[7]
- abnormal thinking[7]
- behavior changes[7]
- depression[7][21]
- sleep driving[7] and sleepwalking
- dry mouth[7]
- rash[7][21]
- altered sleep patterns[7]
- impaired coordination[7]
- dizziness[7]
- daytime drowsiness[7]
- itching[21]
- painful or frequent urination[21]
- back pain[21]
- aggressive behavior[21]
- confusion[21]
- agitation[21]
- suicidal thoughts[21]
- depersonalization[21]
- amnesia[21]
A 2009 meta-analysis found a 44% higher rate of mild infections, such as pharyngitis or sinusitis, in people taking eszopiclone or other hypnotic drugs compared to those taking a placebo.[22]
Dependence
In the United States, eszopiclone is a schedule IV controlled substance under the
A study funded and carried out by Sepracor, the manufacturer of eszopiclone, found no signs of tolerance or dependence in a group of patients followed for up to six months.[23]
Non-medical use
A study of non-medical use potential of eszopiclone found that in persons with a known history of non-medical
Overdose
Overdoses of eszopiclone up to 90 times the recommended dose have been reported in which the patient fully recovered.[3] Fatalities have been reported only in cases in which eszopiclone was combined with other drugs or alcohol.[3] Overdose may be successfully treated with flumazenil, a GABAA receptor antagonist used also for benzodiazepine overdose.[24]
Poison control centers reported that between 2005 and 2006 there were 525 total eszopiclone overdoses recorded in the state of Texas, the majority of which were intentional suicide attempts.[25]
If consumed within the last hour, eszopiclone overdose can be treated with the administration of
Interactions
There is an increased risk of
Pharmacology
Eszopiclone acts on
History
In a controversial 2009 article in the
Availability in Europe
On September 11, 2007, Sepracor signed a marketing deal with British pharmaceutical company
References
- FDA. Retrieved 22 Oct 2023.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d e f "Lunesta- eszopiclone tablet, coated". DailyMed. 24 May 2023. Retrieved 7 July 2023.
- ^ PMID 23248080.
- ^ a b c d e f g "Eszopiclone Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 6 April 2019.
- PMID 30303519.
- ^ a b c d e f g h i j k l m n o p q r s t u v "Eszopiclone" (PDF). F.A. Davis. 2017. Archived from the original (PDF) on October 8, 2017. Retrieved April 15, 2017.
- PMID 30303519.
- ^ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ^ "Eszopiclone - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ^ "Lunivia: Withdrawn application". European Medicines Agency. 17 September 2018. Retrieved 19 February 2023.
- ^ Edwards J (13 June 2009). "End of Sepracor-GSK Deal Raises Question in Lunesta Patent Fight". www.cbsnews.com. Retrieved 7 April 2019.
- ^ "Sepracor Pharmaceuticals Ltd withdraws its marketing authorisation application for Lunivia (eszopiclone)". European Medicines Agency. 15 June 2009. Archived from the original on 1 December 2017. Retrieved 7 April 2019.
- PMID 30303519.
- ^ "FDA warns of next-day impairment with sleep aid Lunesta". U.S. Food and Drug Administration (FDA). 15 January 2016. Retrieved 7 July 2023.
- PMID 18035234.
- S2CID 38797655.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - PMID 16860264.
- PMID 19968019.
- S2CID 7946506.
- ^ a b c d e f g h i j k l m n Rxlist (26 October 2016). "Lunesta". Archived from the original on 5 December 2008. Retrieved 15 April 2017.
- PMID 19968019.
- ^ PMID 16424933.
- ISBN 978-0-07-147914-1.
- S2CID 25102558.
- ^ "Zopiclone overdose". MHRA. Medicines and Healthcare Products Regulatory Agency. Archived from the original on December 6, 2014.
- PMID 18265473.
- PMID 16373464.
- PMID 16750462.
- PMID 21143957.
- ^ Ashton CH (April 2007). "Benzodiazepine Equivalence Table". benzo.org.uk. Retrieved 21 March 2008.
- PMID 19846841.
- ^ a b "GlaxoSmithKline and Sepracor Inc. announce international alliance for commercialisation of Lunivia". Archived from the original on December 27, 2010.
- ^ Committee for Medicinal Products for Human Use – Summary of Positive Opinion for Lunivia[permanent dead link] – European Medicines Agency/Committee for Medicinal Products for Human Use, 23 Oct 2010
- ^ "Sepracor Pharmaceuticals Ltd withdraws its marketing authorisation application for Lunivia (eszopiclone)". European Medicines Agency. 15 May 2009. Archived from the original on 1 December 2017.
- ^ Smillie M (23 April 2010). "Data exclusivity and definition of a new active substance: suspension of generic escitalopram-containing medicines by CHMP". Bird and Bird Commercial Law. Archived from the original on 23 May 2010.
- ^ "Almorexant for Treatment of Primary Insomnia". Clinical Trials Arena. Retrieved 2021-08-04.
