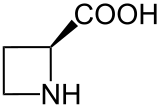
Azetidine-2-carboxylic acid
| |
| Names | |
|---|---|
| IUPAC name
Azetidine-2-carboxylic acid
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.016.693 |
| EC Number |
|
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H7NO2 | |
| Molar mass | 101.104 g/mol |
| Appearance | crystalline solid |
| Density | 1.275 g/cm3 |
| Melting point | 215 °C (419 °F; 488 K) |
| Boiling point | 242 °C (468 °F; 515 K) |
| 5.0 g/100 ml | |
| Hazards | |
| Flash point | 100.1 °C (212.2 °F; 373.2 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Azetidine-2-carboxylic acid (abbreviated Aze or Azc) is a plant
Synthesis
Optically inactive Aze was obtained in small yield from the neurotransmitter
Occurrence
Azetidine-2-carboxylic acid has been known since 1955 to be present in rhizomes and fresh foliage of certain plants. It is known to occur in two species from the
Toxicity
It has been shown that when Aze is misincorporated into proteins in place of proline, Aze deters the growth of competing vegetation and poisons predators. Other studies have shown effects of Aze resulting in a wide range of toxic and teratogenic disorders, including in a range of malformations, in various animal species including ducks, hamsters, mice, and rabbits.[2]
Misincorporation of Aze into human proteins can alter collagen, keratin, hemoglobin, and protein folding.[5] However, the lack of detailed toxicologic data and the need for more direct evidence about the damaging effects of the misincorporation of Aze on specific proteins are reasons why the toxicity of Aze to humans cannot be determined at this time.[2] Molecular studies of human prolyl- and alanyl-tRNA synthetases suggest that Aze is incorporated in proteins as proline with toxic consequences in vivo.[6] Even if Aze seems to fit into the active site of both tRNA synthetases (due to its double mimicry effect of alanine and proline), it is rejected by alanyl-tRNA synthetases post-transfer editing system.[6]
References
- ^ Merck Index, 12th Edition, 6089.
- ^ PMID 19101705.
- PMID 13363844.
- ISBN 0-412-01981-7.
- PMID 16516254.
- ^ PMID 29273753.
