Carbanion
In
]Formally, a carbanion is the
- R3CH + B− → R3C− + HB
where B stands for the
Carbanions have a concentration of electron density at the negatively charged carbon, which, in most cases, reacts efficiently with a variety of
Geometry
Absent π delocalization, the negative charge of a carbanion is localized in an spx hybridized orbital on carbon as a lone pair. As a consequence, localized alkyl, alkenyl/aryl, and alkynyl carbanions assume trigonal pyramidal, bent, and linear geometries, respectively. By Bent's rule, placement of the carbanionic lone pair electrons in an orbital with significant s character is favorable, accounting for the pyramidalized and bent geometries of alkyl and alkenyl carbanions, respectively. Valence shell electron pair repulsion (VSEPR) theory makes similar predictions. This contrasts with carbocations, which have a preference for unoccupied nonbonding orbitals of pure atomic p character, leading to planar and linear geometries, respectively, for alkyl and alkenyl carbocations.


However, delocalized carbanions may deviate from these geometries. Instead of residing in a hybrid orbital, the carbanionic lone pair may instead occupy a p orbital (or an orbital of high p character). A p orbital has a more suitable shape and orientation to overlap with the neighboring π system, resulting in more effective charge delocalization. As a consequence, alkyl carbanions with neighboring conjugating groups (e.g., allylic anions, enolates, nitronates, etc.) are generally planar rather than pyramidized. Likewise, delocalized alkenyl carbanions sometimes favor a linear instead of bent geometry. More often, a bent geometry is still preferred for substituted alkenyl anions, though the linear geometry is only slightly less stable, resulting in facile equilibration between the (E) and (Z) isomers of the (bent) anion through a linear transition state.[2] For instance, calculations indicate that the parent vinyl anion or ethylenide, H2C=CH−, has an inversion barrier of 27 kcal/mol (110 kJ/mol), while allenyl anion or allenide, H2C=C=CH− ↔ H2C−−C≡CH), whose negative charge is stabilized by delocalization, has an inversion barrier of only 4 kcal/mol (17 kJ/mol), reflecting stabilization of the linear transition state by better π delocalization.[3]
Trends and occurrence
Carbanions are typically
- the inductive effect. Electronegative atoms adjacent to the charge will stabilize the charge;
- the extent of conjugation of the anion. Resonance effects can stabilize the anion. This is especially true when the anion is stabilized as a result of aromaticity.
Geometry also affects the
Carbanions, especially ones derived from weak carbon acids that do not benefit sufficiently from the two stabilizing factors listed above, are generally oxygen- and water-sensitive to varying degrees. While some merely degrade and decompose over several weeks or months upon exposure to air, others may react vigorously and exothermically with air almost immediately to spontaneously ignite (pyrophoricity). Among commonly encountered carbanionic reagents in the laboratory, ionic salts of hydrogen cyanide (cyanides) are unusual in being indefinitely stable under dry air and hydrolyzing only very slowly in the presence of moisture.
Organometallic reagents like
In fact, true carbanions (i.e., a species not attached to a stabilizing covalently bound metal) without electron-withdrawing and/or conjugating substituents are not available in the condensed phase, and these species must be studied in the gas phase. For some time, it was not known whether simple alkyl anions could exist as free species; many theoretical studies predicted that even the
Simple primary, secondary and tertiary sp3 carbanions (e.g., ethanide CH3CH−2, isopropanide (CH3)2CH−, and t-butanide (CH3)3C− were subsequently determined to be unbound species (the EAs of CH3CH2•, (CH3)2CH•, (CH3)3C• are −6, −7.4, −3.6 kcal/mol, respectively) indicating that α substitution is destabilizing. However, relatively modest stabilizing effects can render them bound. For example,
In the condensed phase only carbanions that are sufficiently stabilized by delocalization have been isolated as truly ionic species. In 1984, Olmstead and
Adding
Early in 1904[10] and 1917,[11] Schlenk prepared two red-colored salts, formulated as [NMe4]+[CPh3]− and [NMe4]+[PhCH2]−, respectively, by metathesis of the corresponding organosodium reagent with tetramethylammonium chloride. Since tetramethylammonium cations cannot form a chemical bond to the carbanionic center, these species are believed to contain free carbanions. While the structure of the former was verified by X-ray crystallography almost a century later,[12] the instability of the latter has so far precluded structural verification. The reaction of the putative "[NMe4]+[PhCH2]−" with water was reported to liberate toluene and tetramethylammonium hydroxide and provides indirect evidence for the claimed formulation.
One tool for the detection of carbanions in solution is
Carbon acids
Any compound containing hydrogen can, in principle, undergo deprotonation to form its conjugate base. A compound is a carbon acid if deprotonation results in loss of a proton from a carbon atom. Compared to compounds typically considered to be acids (e.g.,
These values below are pKa values determined in
Carbon acid acidities by
These values may differ significantly from aqueous pKa values.Name Formula Structural formula pKa in DMSO Cyclohexane C6H12 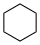
~60 Methane CH4 
~56 Benzene C6H6 
~49[17] PropeneC3H6 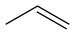
~44 Toluene C6H5CH3 
~43 Ammonia (N–H) NH3 
~41 Dithiane C4H8S2 
~39 Dimethyl sulfoxide (CH3)2SO 
35.1 Diphenylmethane C13H12 
32.3 Acetonitrile CH3CN 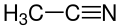
31.3 Aniline (N–H) C6H5NH2 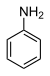
30.6 Triphenylmethane C19H16 
30.6 Fluoroform CHF3 
30.5[18] Xanthene C13H10O 
30.0 Ethanol (O–H) C2H5OH 
29.8 Phenylacetylene C8H6 
28.8 Thioxanthene C13H10S 
28.6 Acetone C3H6O 
26.5 Chloroform CHCl3 
24.4[18] Benzoxazole C7H5NO 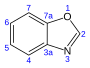
24.4 Fluorene C13H10 
22.6 Indene C9H8 
20.1 Cyclopentadiene C5H6 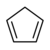
18.0 Nitromethane CH3NO2 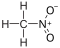
17.2 Diethyl malonate C7H12O4 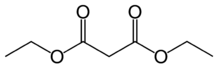
16.4 Acetylacetone (H3CCO)2CH2 
13.3 Hydrogen cyanide HCN 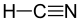
12.9 Acetic acid (O–H) CH3COOH 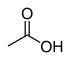
12.6 Malononitrile C3H2N2 
11.1 Dimedone C8H12O2 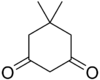
10.3 Meldrum's acid C6H8O4 
7.3 Hexafluoroacetylacetone (F3CCO)2CH2 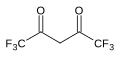
2.3 Hydrogen chloride (Cl–H) HCl HCl (g) −2.0[19] Triflidic acid HC(SO2CF3)3 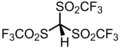
~ −16[a]
- Note that acetic acid, ammonia, aniline, ethanol, and hydrogen chloride are not carbon acids, but are common acids shown for comparison.
- ^ The reported pKa in acetonitrile (MeCN) is −3.7.[20] The pKa in DMSO was estimated by the correlation pKaMeCN = 0.98 × pKaDMSO + 11.6.[21]
As indicated by the examples above, acidity increases (pKa decreases) when the negative charge is delocalized. This effect occurs when the substituents on the carbanion are unsaturated and/or electronegative. Although carbon acids are generally thought of as acids that are much weaker than "classical" Brønsted acids like acetic acid or phenol, the cumulative (additive) effect of several electron accepting substituents can lead to acids that are as strong or stronger than the inorganic mineral acids. For example,
The acidity of the α-hydrogen in
Chiral carbanions
With the
The first ever evidence for the existence of chiral organolithium compounds was obtained in 1950. Reaction of chiral 2-iodooctane with
On heating the reaction to 0 °C the optical activity is lost. More evidence followed in the 1960s. A reaction of the cis isomer of 2-methylcyclopropyl bromide with s-butyllithium again followed by carboxylation with dry ice yielded cis-2-methylcyclopropylcarboxylic acid. The formation of the trans isomer would have indicated that the intermediate carbanion was unstable.[23]
In the same manner the reaction of (+)-(S)-l-bromo-l-methyl-2,2-diphenylcyclopropane with n-butyllithium followed by quenching with
Of recent date are chiral methyllithium compounds:[25]
The
History
A carbanionic structure first made an appearance in the reaction mechanism for the benzoin condensation as correctly proposed by Clarke and Arthur Lapworth in 1907.[26] In 1904 Wilhelm Schlenk prepared [Ph3C]−[NMe4]+ in a quest for tetramethylammonium (from tetramethylammonium chloride and Ph3CNa)[10] and in 1914 he demonstrated how triarylmethyl radicals could be reduced to carbanions by alkali metals [27] The phrase carbanion was introduced by Wallis and Adams in 1933 as the negatively charged counterpart of the carbonium ion[28][29]
See also
References
- ISSN 0040-4039.
- ISBN 978-1-118-90637-8.
- PMID 16592384.
- ISSN 0002-7863.
- OCLC 55939535.
- .
- PMID 12203352.
- .
- ^ .
- ISSN 1099-0682.
- PMID 12203352.
- .
- .
- ^ a b Evans, D. A.; Ripin, D. H. (2005). "Chem 206 pKa Table" (PDF). Archived from the original (PDF) on 2019-07-02.
- .
- .
- ^ ISSN 0040-4020.
- S2CID 29697201.
- PMID 21166439.
- PMID 19275192.
- .
- .
- .
- PMID 17243828.
- .
- .
- .
- S2CID 98171271.


























![Chiral oxy[2H1]methyllithiums. Bu stands for butyl, i-Pr stands for isopropyl.](http://upload.wikimedia.org/wikipedia/commons/thumb/1/1d/PhosphatePhosphonateRearrangement.png/500px-PhosphatePhosphonateRearrangement.png)