Carbon monoxide poisoning
| Carbon monoxide poisoning | |
|---|---|
| Other names | Carbon monoxide intoxication, carbon monoxide toxicity, carbon monoxide overdose |
hyperbaric oxygen therapy[2] | |
| Prognosis | Risk of death: 1–31%[2] |
| Frequency | >20,000 emergency visits for non-fire related cases per year (US)[1] |
| Deaths | >400 non-fire related a year (US)[1] |
Carbon monoxide poisoning typically occurs from
CO is a colorless and odorless gas which is initially non-irritating.
Diagnosis is typically based on a HbCO level of more than 3% among nonsmokers and more than 10% among smokers.
Efforts to prevent poisoning include
Carbon monoxide poisoning is relatively common, resulting in more than 20,000
Background
Carbon monoxide is not toxic to all forms of life, and the toxicity is a classical dose-dependent example of hormesis. Small amounts of carbon monoxide are naturally produced through many enzymatic and non-enzymatic reactions across phylogenetic kingdoms where it can serve as an important neurotransmitter (subcategorized as a gasotransmitter) and a potential therapeutic agent.[14] In the case of prokaryotes, some bacteria produce, consume and respond to carbon monoxide whereas certain other microbes are susceptible to its toxicity.[6] Currently, there are no known adverse effects on photosynthesizing plants.[15]
The harmful effects of carbon monoxide are generally considered to be due to tightly binding with the
As pioneered by
History
Humans have maintained a complex relationship with carbon monoxide since first learning to control fire circa 800,000 BC. Primitive
Early civilizations developed
Documented cases by Livy and Cicero allude to carbon monoxide being used as a method of suicide in ancient Rome.[9][20] Emperor Lucius Verus used smoke to execute prisoners.[9] Many deaths have been linked to carbon monoxide poisoning including Emperor Jovian, Empress Fausta, and Seneca.[9] The most high-profile death by carbon monoxide poisoning may possibly have been Cleopatra[9] or Edgar Allan Poe.[21]
In the fifteenth century, coal miners believed sudden death was caused by
Georg Ernst Stahl mentioned carbonarii halitus in 1697 in reference to toxic vapors thought to be carbon monoxide. Friedrich Hoffmann conducted the first modern scientific investigation into carbon monoxide poisoning from coal in 1716, notably rejecting villagers attributing death to demonic superstition. Herman Boerhaave conducted the first scientific experiments on the effect of carbon monoxide (coal fumes) on animals in the 1730s.[9] Joseph Priestley is credited with first synthesizing carbon monoxide in 1772 which he had called heavy inflammable air, and Carl Wilhelm Scheele isolated carbon monoxide from coal in 1773 suggesting it to be the toxic entity.[9]
The dose-dependent risk of carbon monoxide poisoning as
William Cruickshank discovered carbon monoxide as a molecule containing one carbon and one oxygen atom in 1800, thereby initiating the modern era of research exclusively focused on carbon monoxide. The mechanism for toxicity was first suggested by James Watt in 1793, followed by Adrien Chenot in 1854 and finally demonstrated by Claude Bernard after 1846 as published in 1857 and also independently published by Felix Hoppe-Seyler in the same year.[9]
The first controlled clinical trial studying the toxicity of carbon monoxide occurred in 1973.[9]
Historical detection
Carbon monoxide poisoning has plagued coal miners for many centuries. In the context of mining, carbon monoxide is widely known as
The first qualitative analytical method to detect carboxyhemoglobin emerged in 1858 with a colorimetric method developed by Felix Hoppe-Seyler, and the first quantitative analysis method emerged in 1880 with Josef von Fodor.[9]
Historical treatment
The use of oxygen emerged with anecdotal reports such as
Incidents
The worst accidental mass poisoning from carbon monoxide was the Balvano train disaster which occurred on 3 March 1944 in Italy, when a freight train with many illegal passengers stalled in a tunnel, leading to the death of over 500 people.[23]
Over 50 people are suspected to have died from smoke inhalation as a result of the Branch Davidian Massacre during the Waco siege in 1993.[24]
Weaponization
In ancient history, Hannibal executed Roman prisoners with coal fumes during the Second Punic War.[9]
The extermination of stray dogs by a carbon monoxide gas chamber was described in 1874.[9] In 1884, an article appeared in Scientific American describing the use of a carbon monoxide gas chamber for slaughterhouse operations as well as euthanizing a variety of animals.[25]
As part of the
Physiology
Carbon monoxide is produced naturally by many physiologically relevant enzymatic and non-enzymatic reactions
Therapeutics
Small amounts of CO are beneficial and enzymes exist that produce it at times of oxidative stress.
Signs and symptoms
On average, exposures at 100
| Concentration | Symptoms |
|---|---|
| 35 ppm (0.0035%), (0.035‰) | Headache and dizziness within six to eight hours of constant exposure |
| 100 ppm (0.01%), (0.1‰) | Slight headache in two to three hours |
| 200 ppm (0.02%), (0.2‰) | Slight headache within two to three hours; loss of judgment |
| 400 ppm (0.04%), (0.4‰) | Frontal headache within one to two hours |
| 800 ppm (0.08%), (0.8‰) | Dizziness, nausea, and convulsions within 45 min; insensible within 2 hours |
| 1,600 ppm (0.16%), (1.6‰) | Headache, increased heart rate, dizziness, and nausea within 20 min; death in less than 2 hours |
| 3,200 ppm (0.32%), (3.2‰) | Headache, dizziness and nausea in five to ten minutes. Death within 30 minutes. |
| 6,400 ppm (0.64%), (6.4‰) | Headache and dizziness in one to two minutes. Convulsions, respiratory arrest, and death in less than 20 minutes. |
| 12,800 ppm (1.28%), (12.8‰) | Unconsciousness after 2–3 breaths. Death in less than three minutes. |
Acute poisoning

The main manifestations of carbon monoxide poisoning develop in the organ systems most dependent on oxygen use, the
One of the major concerns following acute carbon monoxide poisoning is the severe delayed neurological manifestations that may occur. Problems may include difficulty with higher intellectual functions,
Chronic poisoning
Chronic exposure to relatively low levels of carbon monoxide may cause persistent headaches, lightheadedness, depression, confusion, memory loss, nausea, hearing disorders and vomiting.[49][50] It is unknown whether low-level chronic exposure may cause permanent neurological damage.[28] Typically, upon removal from exposure to carbon monoxide, symptoms usually resolve themselves, unless there has been an episode of severe acute poisoning.[49] However, one case noted permanent memory loss and learning problems after a three-year exposure to relatively low levels of carbon monoxide from a faulty furnace.[51]
Chronic exposure may worsen cardiovascular symptoms in some people.
In experimental animals, carbon monoxide appears to worsen noise-induced hearing loss at noise exposure conditions that would have limited effects on hearing otherwise.[55] In humans, hearing loss has been reported following carbon monoxide poisoning.[50] Unlike the findings in animal studies, noise exposure was not a necessary factor for the auditory problems to occur.
Fatal poisoning
One classic sign of carbon monoxide poisoning is more often seen in the dead rather than the living – people have been described as looking red-cheeked and healthy. However, since this "cherry-red" appearance is more common in the dead, it is not considered a useful diagnostic sign in clinical medicine. In autopsy examinations, the appearance of carbon monoxide poisoning is notable because unembalmed dead persons are normally bluish and pale, whereas dead carbon-monoxide poisoned people may appear unusually lifelike in coloration.[56][57][58] The colorant effect of carbon monoxide in such postmortem circumstances is thus analogous to its use as a red colorant in the commercial meat-packing industry.
Epidemiology
The true number of cases of carbon monoxide poisoning is unknown, since many non-lethal exposures go undetected.[36][59] From the available data, carbon monoxide poisoning is the most common cause of injury and death due to poisoning worldwide.[60] Poisoning is typically more common during the winter months.[31][61][62][63] This is due to increased domestic use of gas furnaces, gas or kerosene space heaters, and kitchen stoves during the winter months, which if faulty and/or used without adequate ventilation, may produce excessive carbon monoxide.[31][64] Carbon monoxide detection and poisoning also increases during power outages, when electric heating and cooking appliances become inoperative and residents may temporarily resort to fuel-burning space heaters, stoves, and grills (some of which are safe only for outdoor use but nonetheless are errantly burned indoors).[65][66][67]
It has been estimated that more than 40,000 people per year seek medical attention for carbon monoxide poisoning in the United States.[68] 95% of carbon monoxide poisoning deaths in the United States are due to gas space heaters.[69][70] In many industrialized countries, carbon monoxide is the cause of more than 50% of fatal poisonings.[11] In the United States, approximately 200 people die each year from carbon monoxide poisoning associated with home fuel-burning heating equipment.[71] Carbon monoxide poisoning contributes to the approximately 5613 smoke inhalation deaths each year in the United States.[72] The CDC reports, "Each year, more than 500 Americans die from unintentional carbon monoxide poisoning, and more than 2,000 commit suicide by intentionally poisoning themselves."[73] For the 10-year period from 1979 to 1988, 56,133 deaths from carbon monoxide poisoning occurred in the United States, with 25,889 of those being suicides, leaving 30,244 unintentional deaths.[72] A report from New Zealand showed that 206 people died from carbon monoxide poisoning in the years of 2001 and 2002. In total carbon monoxide poisoning was responsible for 43.9% of deaths by poisoning in that country.[74] In South Korea, 1,950 people had been poisoned by carbon monoxide with 254 deaths from 2001 through 2003.[75] A report from Jerusalem showed 3.53 per 100,000 people were poisoned annually from 2001 through 2006.[76] In Hubei, China, 218 deaths from poisoning were reported over a 10-year period with 16.5% being from carbon monoxide exposure.[77]
Causes
| Concentration | Source |
|---|---|
| 0.1 ppm | Natural atmosphere level (MOPITT)[78] |
| 0.5 to 5 ppm | Average level in homes[79] |
| 5 to 15 ppm | Near properly adjusted gas stoves in homes[79] |
| 100 to 200 ppm | Exhaust from automobiles in the Mexico City central area[80] |
| 5,000 ppm | Exhaust from a home wood fire[81] |
| 7,000 ppm | Undiluted warm car exhaust without a catalytic converter[81] |
| 30,000 ppm | Afterdamp following an explosion in a coal mine[82] |
Carbon monoxide is a product of combustion of organic matter under conditions of restricted oxygen supply, which prevents complete
Riding in the back of
Poisoning may also occur following the use of a self-contained underwater breathing apparatus (SCUBA) due to faulty diving air compressors.[93]
In caves carbon monoxide can build up in enclosed chambers due to the presence of decomposing organic matter.[94] In coal mines incomplete combustion may occur during explosions resulting in the production of afterdamp. The gas is up to 3% CO and may be fatal after just a single breath.[82] Following an explosion in a colliery, adjacent interconnected mines may become dangerous due to the afterdamp leaking from mine to mine. Such an incident followed the Trimdon Grange explosion which killed men in the Kelloe mine.[95]
Another source of poisoning is exposure to the organic solvent
Prevention
Detectors

Prevention remains a vital public health issue, requiring public education on the safe operation of appliances, heaters, fireplaces, and internal-combustion engines, as well as increased emphasis on the installation of carbon monoxide detectors.[61] Carbon monoxide is tasteless, odourless, and colourless, and therefore can not be detected by visual cues or smell.[100]
The United States
The use of carbon monoxide detectors has been standardized in many areas. In the US, NFPA 720–2009,[104] the carbon monoxide detector guidelines published by the National Fire Protection Association, mandates the placement of carbon monoxide detectors/alarms on every level of the residence, including the basement, in addition to outside sleeping areas. In new homes, AC-powered detectors must have battery backup and be interconnected to ensure early warning of occupants at all levels.[104] NFPA 720-2009 is the first national carbon monoxide standard to address devices in non-residential buildings. These guidelines, which now pertain to schools, healthcare centers, nursing homes, and other non-residential buildings, include three main points:[104]
- 1. A secondary power supply (battery backup) must operate all carbon monoxide notification appliances for at least 12 hours,
- 2. Detectors must be on the ceiling in the same room as permanently installed fuel-burning appliances, and
- 3. Detectors must be located on every habitable level and in every HVACzone of the building.
Gas organizations will often recommend getting gas appliances serviced at least once a year.[105]
Legal requirements
The NFPA standard is not necessarily enforced by law. As of April 2006, the US state of Massachusetts requires detectors to be present in all residences with potential CO sources, regardless of building age and whether they are owner-occupied or rented.[106] This is enforced by municipal inspectors and was inspired by the death of 7-year-old Nicole Garofalo in 2005 due to snow blocking a home heating vent.[107] Other jurisdictions may have no requirement or only mandate detectors for new construction or at time of sale.
Despite similar deaths in vehicles with clogged exhaust pipes (for example in the
World Health Organization recommendations
The following guideline values (ppm values rounded) and periods of time-weighted average exposures have been determined in such a way that the carboxyhaemoglobin (COHb) level of 2.5% is not exceeded, even when a normal subject engages in light or moderate exercise:
- 100 mg/m3 (87 ppm) for 15 min
- 60 mg/m3 (52 ppm) for 30 min
- 30 mg/m3 (26 ppm) for 1 h
- 10 mg/m3 (9 ppm) for 8 h
- 7 mg/m3 (6 ppm) for 24 h (for indoor air quality, so as not to exceed 2% COHb for chronic exposure)[108]
Diagnosis



As many symptoms of carbon monoxide poisoning also occur with many other types of poisonings and infections (such as the flu), the diagnosis is often difficult.[59][109] A history of potential carbon monoxide exposure, such as being exposed to a residential fire, may suggest poisoning, but the diagnosis is confirmed by measuring the levels of carbon monoxide in the blood. This can be determined by measuring the amount of carboxyhemoglobin compared to the amount of hemoglobin in the blood.[28]
The ratio of carboxyhemoglobin to hemoglobin molecules in an average person may be up to 5%, although cigarette smokers who smoke two packs per day may have levels up to 9%.[110] In symptomatic poisoned people they are often in the 10–30% range, while persons who die may have postmortem blood levels of 30–90%.[111][112]
As people may continue to experience significant symptoms of CO poisoning long after their blood carboxyhemoglobin concentration has returned to normal, presenting to examination with a normal carboxyhemoglobin level (which may happen in late states of poisoning) does not rule out poisoning.[113]
Measuring
Carbon monoxide may be quantitated in blood using spectrophotometric methods or chromatographic techniques in order to confirm a diagnosis of poisoning in a person or to assist in the forensic investigation of a case of fatal exposure.
A
Differential diagnosis
There are many conditions to be considered in the differential diagnosis of carbon monoxide poisoning.
Treatment
| Oxygen pressure О2 | Time |
|---|---|
| 21% oxygen at normal atmospheric pressure (fresh air) | 5 hours 20 min |
| 100% oxygen at normal atmospheric pressure (non-rebreather oxygen mask) | 1 hours 20 min |
| 100% hyperbaric oxygen (3 atmospheres absolute) | 23 min |
Initial treatment for carbon monoxide poisoning is to immediately remove the person from the exposure without endangering further people. Those who are unconscious may require CPR on site.[56] Administering oxygen via non-rebreather mask shortens the half-life of carbon monoxide from 320 minutes, when breathing normal air, to only 80 minutes.[40] Oxygen hastens the dissociation of carbon monoxide from carboxyhemoglobin, thus turning it back into hemoglobin.[16][124] Due to the possible severe effects in the baby, pregnant women are treated with oxygen for longer periods of time than non-pregnant people.[125]
Hyperbaric oxygen
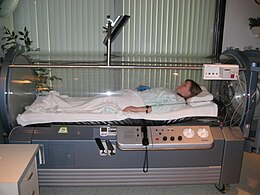
Other
Further treatment for other complications such as
Pathophysiology
The precise mechanisms by which the effects of carbon monoxide are induced upon bodily systems are complex and not yet fully understood.
Hemoglobin
Carbon monoxide has a higher diffusion coefficient compared to oxygen, and the main enzyme in the human body that produces carbon monoxide is heme oxygenase, which is located in nearly all cells and platelets.[6] Most endogenously produced CO is stored bound to hemoglobin as carboxyhemoglobin. The simplistic understanding for the mechanism of carbon monoxide toxicity is based on excess carboxyhemoglobin decreasing the oxygen-delivery capacity of the blood to tissues throughout the body. In humans, the affinity between hemoglobin and carbon monoxide is approximately 240 times stronger than the affinity between hemoglobin and oxygen.[56][151][152] However, certain mutations such as the Hb-Kirklareli mutation has a relative 80,000 times greater affinity for carbon monoxide than oxygen resulting in systemic carboxyhemoglobin reaching a sustained level of 16% COHb.[9]
Hemoglobin is a
At toxic concentrations, carbon monoxide as carboxyhemoglobin significantly interferes with respiration and gas exchange by simultaneously inhibiting acquisition and delivery of oxygen to cells and preventing formation of
) in addition to the toxicities of excess carbon monoxide inhibiting numerous hemoproteins, metallic and non-metallic targets which affect cellular machinery.Myoglobin
Carbon monoxide also binds to the
Cytochrome oxidase
Another mechanism involves effects on the mitochondrial respiratory enzyme chain that is responsible for effective tissue utilization of oxygen. Carbon monoxide binds to
Central nervous system effects
The mechanism that is thought to have a significant influence on delayed effects involves formed blood cells and chemical mediators, which cause brain
Pregnancy
Carbon monoxide poisoning in pregnant women may cause severe adverse
References
- ^ a b c d e f g h i j k National Center for Environmental Health (30 December 2015). "Carbon Monoxide Poisoning – Frequently Asked Questions". www.cdc.gov. Archived from the original on 5 July 2017. Retrieved 2 July 2017.
- ^ PMID 22998990.
- ^ ISBN 978-1284107272. Archivedfrom the original on 10 September 2017. Retrieved 2 July 2017.
- ISBN 978-1405103572. Retrieved 2 July 2017.
- ^ PMID 26563790.
- ^ S2CID 224824871.
- PMID 28077358.
- PMID 31820474.
- ^ .
- ^ ISBN 978-0849384189. Archivedfrom the original on 10 September 2017. Retrieved 2 July 2017.
- ^ PMID 12324190.
- ISBN 978-0323448383. Archivedfrom the original on 10 September 2017. Retrieved 2 July 2017.
- PMID 11387414.
- ^ S2CID 205477130.
- ^ "Carbon monoxide". ernet.in. Archived from the original on 2014-04-14.
- ^ PMID 10771127.
- ^ "Carbon Monoxide". American Lung Association. Archived from the original on 2008-05-28. Retrieved 2009-09-14.
- PMID 16805152.
- ISBN 978-9241572132.
- ^ ISBN 978-0849384189. Archivedfrom the original on 2017-09-10.
- ^ Geiling N. "The (Still) Mysterious Death of Edgar Allan Poe". Smithsonian Magazine. Retrieved 2021-05-03.
- ^ A scientific approach to the paranormal | Carrie Poppy, pp. 4:01, retrieved 2021-05-27
- ISBN 0-8453-4844-2.
- ^ "10 Things You May Not Know About Waco". FRONTLINE. Retrieved 2021-05-28.
- ^ Scientific American. Munn & Company. 1884. p. 148.
- ISBN 978-0313346422. Archivedfrom the original on 2013-05-26.
- ^ S2CID 231762031.
- ^ ISBN 978-0071360012.
- S2CID 6982787.
- ^ S2CID 23892477.
- ^ PMID 16567227.
- ^ "OSHA Fact Sheet: Carbon Monoxide" (PDF). United States National Institute for Occupational Safety and Health. Archived (PDF) from the original on 2009-08-26. Retrieved 2009-09-14.
- ^ "Carbon monoxide – 29 CFR Part 1917 Section 1917.24". United States Department of Labor: Occupational Safety and Health Administration. Archived from the original on 2013-05-23. Retrieved 2013-05-27.
- PMID 19022078.
- PMID 9876005.
- ^ PMID 7966524.
- S2CID 8773611.
- ^ PMID 11410684.
- PMID 9559621.
- ^ PMID 19297574.
- ^ emedicine. Archivedfrom the original on 9 March 2009. Retrieved 2009-04-27.
- PMID 2298080.
- S2CID 46035819.
- S2CID 35144795.
- PMID 16434630.
- PMID 6860181.
- S2CID 1280793.
- PMID 4061987.
- ^ PMID 1552375.
- ^ OCLC 939229378.
- PMID 14589544.
- PMID 19851048.
- ^ Shephard R (1983). Carbon Monoxide The Silent Killer. Springfield Illinois: Charles C Thomas. pp. 93–96.
- PMID 2682242.
- S2CID 5731842.
- ^ .
- S2CID 40041894.
- PMID 11858933.
- ^ PMID 9950394.
- PMID 12362013.
- ^ PMID 9828249.
- from the original on 2017-06-24.
- PMID 19860138.
- PMID 3580051.
- ^ "Department of Public Health Warns of Dangers of Carbon Monoxide Poisoning During Power Outages". Tower Generator. Archived from the original on 2012-04-26. Retrieved 2011-11-23.
- ^ "Avoiding Carbon Monoxide poisoning during a power outage". CDC. Archived from the original on 2011-12-12. Retrieved 2011-11-23.
- S2CID 29853571.
- PMID 9752939.
- ^ "2004 Addendum to Overseas and Australian Statistics and Benchmarks for Customer Gas Safety Incidents" (PDF). Office of Gas Safety, Victoria. 2004. Archived from the original (PDF) on 2015-09-24.
- ^ "The risk of carbon monoxide poisoning from domestic gas appliances" (PDF). Report to the Department of Resources, Energy and Tourism. Feb 2012. Archived from the original (PDF) on 2015-03-12.
- ^ a b "Carbon Monoxide Detectors Can Save Lives: CPSC Document #5010". US Consumer Product Safety Commission. Archived from the original on 2009-04-09. Retrieved 2009-04-30.
- ^ PMID 1712865.
- ^ "Carbon Monoxide poisoning fact sheet" (PDF). Centers for Disease Control and Prevention. July 2006. Archived (PDF) from the original on 2008-12-18. Retrieved 2008-12-16.
- PMID 16286939. Archived from the original(Free full text) on 2011-07-14.
- S2CID 22203132.
- S2CID 44624059.
- PMID 19854011.
- ISBN 978-0309026314.
- ^ a b Green W. "An Introduction to Indoor Air Quality: Carbon Monoxide (CO)". United States Environmental Protection Agency. Archived from the original on 2008-12-18. Retrieved 2008-12-16.
- ^ Singer SF. The Changing Global Environment. Dordrecht: D. Reidel Publishing Company. p. 90.
- ^ a b Gosink T (1983-01-28). "What Do Carbon Monoxide Levels Mean?". Alaska Science Forum. Geophysical Institute, University of Alaska Fairbanks. Archived from the original on 2008-12-25. Retrieved 2008-12-16.
- ^ hdl:1842/5365
- ^ "Man died from carbon monoxide poisoning after using 'heat beads' in Greystanes home". The Sydney Morning Herald. 2015-07-18. Archived from the original on 2015-07-19.
- PMID 16083675.
- PMID 1163706.
- ^ "NIOSH Carbon Monoxide Hazards from Small Gasoline Powered Engines". United States National Institute for Occupational Safety and Health. Archived from the original on 2007-10-29. Retrieved 2007-10-15.
- S2CID 6375808.
- ^ Emmerich SJ (July 2011). Measured CO Concentrations at NIST IAQ Test House from Operation of Portable Electric Generators in Attached Garage – Interim Report (Report). United States National Institute of Standards and Technology. Archived from the original on 2013-02-24. Retrieved 2012-04-18.
- PMID 1370334.
- from the original on 2017-06-24.
- PMID 11917924.
- ^ "NIOSH Carbon Monoxide Dangers in Boating". United States National Institute for Occupational Safety and Health. Archived from the original on 2007-10-13. Retrieved 2007-10-15.
- PMID 9388533.
- ISBN 978-0781728454.
- ^ Trimdon Grange, Durham, 16 February 1882, archived from the original on 30 March 2017, retrieved 22 May 2012
- S2CID 13941392.
- PMID 236156.
- PMID 10674554.
- ^ US EPA, OCSPP (22 August 2019). "Final Rule on Regulation of Methylene Chloride in Paint and Coating Removal for Consumer Use". US EPA.
- PMID 22692708. Archived from the original on 2010-12-25. Retrieved 2013-04-14.)
{{cite journal}}: CS1 maint: unfit URL (link - ^ PMID 8765117.
- ^ Lipinski ER (February 14, 1999). "Keeping Watch on Carbon Monoxide". The New York Times. Archived from the original on June 23, 2011. Retrieved 2009-09-09.
- PMID 9496987.
- ^ a b c NFPA 720: Standard for the Installation of Carbon Monoxide (CO) Detection and Warning Equipment. Quincy, MA: National Fire Protection Agency. 2009.
- ^ "Gas Safety in the Home". UK Gas Safe Register. Archived from the original on 2013-05-02. Retrieved 2013-05-27.
- ^ "MGL Ch. 148 §28F1/2 – Nicole's Law, effective March 31, 2006". malegislature.gov. Archived from the original on December 30, 2012.
- ^ "Massachusetts Law About Carbon Monoxide Detectors". Trial Court Law Libraries. Commonwealth of Massachusetts. Archived from the original on 2013-03-05. Retrieved 2013-02-22.
- ^ "Carbon monoxide". WHO Guidelines for Indoor Air Quality: Selected Pollutants. WHO. 2010.
- PMID 18430228.
- ISBN 978-0721654850.
- ^ Sato K, Tamaki K, Hattori H, et al. Determination of total hemoglobin in forensic blood samples with special reference to carboxyhemoglobin analysis. For. Sci. Int. 48: 89–96, 1990.
- ^ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 237–41.
- S2CID 20998393.
- PMID 455674. Archived from the originalon 2019-12-16. Retrieved 2009-07-17.
- PMID 7361284.
- PMID 18500075. Archived from the original on 2011-06-25. Retrieved 2009-07-17.)
{{cite journal}}: CS1 maint: unfit URL (link - PMID 20646785.
- PMID 2031826.
- PMID 3578881.
- PMID 3824275.
- PMID 7314006.
- PMID 2403894.
- ISBN 978-1402043765.
- ^ PMID 6491241.
- PMID 3778597.
- PMID 1445151.
- ^ S2CID 30011914.
- ^ PMID 21491385.
- S2CID 70992548.
- S2CID 41578807.
- S2CID 40639134.
- S2CID 71227659.
- from the original on 2008-09-05.
- PMID 7710151.
- S2CID 26710636.
- PMID 7742714. Archived from the original on 2011-08-11. Retrieved 2007-10-05.)
{{cite journal}}: CS1 maint: unfit URL (link - ^ Mathieu D, Mathieu-Nolf M, Durak C, Wattel F, Tempe JP, Bouachour G, Sainty JM (1996). "Randomized prospective study comparing the effect of HBO vs 12 hours NBO in non-comatose CO-poisoned patients: results of the preliminary analysis". Undersea & Hyperbaric Medicine. 23: 7. Archived from the original on 2011-07-02. Retrieved 2008-05-16.
{{cite journal}}: CS1 maint: unfit URL (link) - PMID 12362006.
- S2CID 28464628.
- PMID 15239731.
- PMID 12572577.
- PMID 23767905.
- PMID 9924492.
- ^ Peirce EC (1986). "Treating acidemia in carbon monoxide poisoning may be dangerous". Journal of Hyperbaric Medicine. 1 (2): 87–97. Archived from the original on 2011-07-03. Retrieved 2009-10-29.
{{cite journal}}: CS1 maint: unfit URL (link) - PMID 12361932. Archived from the originalon 2012-07-07.
- PMID 10767186.
- PMID 8037390.
- PMID 21958445.
- ^ "Press Announcements - FDA allows marketing of new device to help treat carbon monoxide poisoning". www.fda.gov. Retrieved 21 March 2019.
- ^ PMID 12679050.
- PMID 12356933.
- PMID 16992272. Archived from the original(PDF) on 2011-07-08. Retrieved 2009-09-15.
- ISBN 978-1-319-11467-1.
- PMID 1750629.
- PMID 12969439.
- ^ PMID 11387414.
- PMID 19751650.
- PMID 8722431.
- PMID 11559621.
- PMID 2262778.
External links
- Centers for Disease Control and Prevention (CDC) – Carbon Monoxide – NIOSH Workplace Safety and Health Topic
- International Programme on Chemical Safety (1999). Carbon Monoxide, Environmental Health Criteria 213, Geneva: WHO
